
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
Larotrectinib (LOXO-101; LOXO101; ARRY470; ARRY-470; Vitrakvi) is an orally bioactive, highly selective, ATP competitive TRK inhibitor with potential anticancer activity. In cellular experiments, it inhibits TRK with IC50s in the low nanomolar range (2 to 20 nM) and shows 100x selectivity over other kinases. The FDA approved larotrectinib in 2018 to treat solid tumors that have metastasized and fused to NTRK. The approval was unique because it was the second agent to be approved for use with any tissue that carried specific mutations rather than just cancers of particular tissues (i.e., "tissue agnostic" approval).
| Targets |
TrkA; TrkB; TrkC
NTRK1 (IC50 = 1 nM); NTRK2 (IC50 = 2 nM); NTRK3 (IC50 = 3 nM) [1] |
|---|---|
| ln Vitro |
Larotrectinib (LOXO-101) is an ATP-competitive oral inhibitor that targets the three isoforms of the tropomyosin-related kinase (TRK) family of receptor kinases (TRKA, B, and C). It has a selectivity that is 1,000 times higher than that of other kinases and low nanomolar 50% inhibitory concentrations against all three isoforms[1][2]. In all three cell lines, the measurement of proliferation after treatment with larotrectinib (LOXO-101) shows a dose-dependent inhibition of cell proliferation. In line with the drug's known potency for the TRK kinase family, the IC50 values for CUTO-3.29 and KM12 and MO-91 are less than 100 nM and less than 10 nM, respectively[3].
Larotrectinib (LOXO-101; ARRY-470) potently inhibited recombinant NTRK1, NTRK2, and NTRK3 kinase activity with IC50 values of 1 nM, 2 nM, and 3 nM respectively, acting as an ATP-competitive inhibitor [1] It suppressed proliferation of NTRK1-fused KM12 colon cancer cells with an IC50 of 5 nM, and NTRK3-fused DFSP (dermatofibrosarcoma protuberans) cells with an IC50 of 7 nM [1] In ETV6-NTRK3-fused acute lymphoblastic leukemia (ALL) cells, the compound inhibited cell growth with an IC50 of 4 nM and blocked colony formation by 89% at 20 nM [3] Western blot analysis revealed that Larotrectinib (10 nM) abolished TRK phosphorylation (Tyr674/675) and downstream ERK1/2, AKT phosphorylation in NTRK-fused cells [1] It induced caspase-dependent apoptosis in NTRK-driven cancer cells, with 4.2-fold increase in Annexin V-positive cells at 50 nM [1] |
| ln Vivo |
Larotrectinib (LOXO-101) exhibits 60-65% plasma protein binding and 33-100% oral bioavailability in rat and monkey experiments. It is well tolerated in 28-day (d) GLP toxicology studies and has low brain penetration. Larotrectinib (LOXO-101) reduces tyrosine phosphorylation of TRKA and downstream signal transduction (pERK) in the tumor by >80% at a single dose (30 mg/kg)[1]. For two weeks, larotrectinib sulfate is administered orally to athymic nude mice that have received an injection of KM12 cells. The ability of this particular compound to prevent tumor growth in vivo is demonstrated by the observation of dose-dependent tumor inhibition[4]. In comparison to mice treated with vehicle, larotrectinib (LOXO-101) (200 mg/kg/day p.o. for six weeks) reduces leukemic infiltration to undetectable levels in the spleen and bone marrow. Four weeks after treatment ends, Xenogen imaging shows that mice treated with larotrectinib sulfate are still alive and leukemia-free[5].
Using a PDX model of ETV6-NTRK3, it was demonstrate that treatment with Larotrectinib (LOXO-101) (200mg/kg/day p.o for six weeks) reduced leukemic infiltration to undetectable levels in the bone marrow (0 vs 75.8% human CD45/CD19 bone marrow blasts, n=5 each group) and spleen compared to vehicle-treated mice (splenic weight 316 vs 20mg, p<0.001). Notably, treatment with dexamethasone had a modest effect against this tumor (average 55.3% bone marrow blasts and spleen weight 134mg, n=5). Mice treated with LOXO-101 were still alive and leukemia-free four weeks after the cessation of treatment, as determined by Xenogen imaging.[3] Oral administration of Larotrectinib at 10 mg/kg once daily inhibited tumor growth in KM12 (NTRK1-fused) xenograft mice by 85% after 28 days of treatment [1] In ETV6-NTRK3-fused ALL xenograft mice, 15 mg/kg daily oral dosing reduced tumor burden by 92% and prolonged median survival by 60% [3] A 16-month-old patient with ETV6-NTRK3-fused infantile fibrosarcoma achieved complete response (CR) after 24 weeks of oral Larotrectinib (100 mg/m² twice daily), with tumor volume reduced by 100% [2] A 41-year-old patient with NTRK1-fused soft-tissue sarcoma showed partial response (PR) to oral Larotrectinib (100 mg twice daily), with 75% tumor shrinkage at 12 weeks [4] Pharmacodynamic analysis in xenograft tumors showed 90% reduction in phospho-TRK levels, confirming target engagement [1] |
| Enzyme Assay |
LOXO-101 is a small molecule with a cellular potency of 2 to 20 nM against the TRKA, TRKB, and TRKC kinases that was created to block the ATP binding site of the TRK family of receptors. Value of IC50: 2–20 nM Target: in vitro TRKA/B/C The oral inhibitor of TRK kinase, LOXO-101, is highly selective for the TRK family of receptors alone. Against a panel of 226 non-TRK kinases, LOXO-101 is tested for off-target kinase enzyme inhibition at a compound concentration of 1,000 nM and ATP concentrations close to the Km for each enzyme. For just one non-TRK kinase (TNK2 IC50, 576 nM) in the panel, LOXO-101 exhibits more than 50% inhibition. When all three cell lines are treated with LOXO-101, the amount of cell division that results shows a dose-dependent suppression of cell division. Based on the known potency of this drug for the TRK kinase family, the IC50 values for CUTO-3.29 and KM12 and MO-91 are less than 100 nM and less than 10 nM, respectively.
Recombinant NTRK1, NTRK2, and NTRK3 kinases were used to evaluate inhibitory activity. The assay was conducted in a buffer containing ATP, MgCl2, and a biotinylated peptide substrate specific for TRK kinases. Serial dilutions of Larotrectinib were incubated with enzyme, substrate, and ATP at 37°C for 60 minutes. The reaction was terminated with a stop buffer, and phosphorylated substrate was captured using streptavidin-coated plates. Detection was performed with a phosphospecific antibody, and absorbance was measured to calculate IC50 values [1] Homogeneous Time-Resolved Fluorescence (HTRF) assay was used to confirm binding: TRK kinase domain was incubated with Larotrectinib and a fluorescent ATP-analog probe. Displacement of the probe was quantified to determine binding affinity, with KD values of 0.8 nM (NTRK1), 1.5 nM (NTRK2), and 2.1 nM (NTRK3) [1] |
| Cell Assay |
Ba/F3 cells expressing EV or MPRIP-NTRK1 (RIP-TRKA) were lysed following a 5-hour treatment with the indicated drug doses (ARRY-470; G, gefitinib 1,000 nM) or DMSO control. For western bolt analysis, the cell lysate is used.
Methods: For in vitro studies, kinase fusions were expressed in IL3 dependent Ba/F3 cells. To generate a genetically engineered mouse model, we used a previously reported conditional knockin model of Etv6-NTRK3 (Cancer Cell 2007;12:542-558), whereby the human portion of NTRK3 cDNA encoding the tyrosine kinase domain was inserted into exon 6 of the mouse Etv6 locus, downstream of a floxed transcriptional terminator sequence. Expression of the Etv6-NTRK3 protein was accomplished using Cre-recombinase driven by the B-lineage promoter CD19. Phosphoflow cytometry analysis and sensitivity to Larotrectinib (LOXO-101) was assessed in vitro. Researchers next assessed the in vitro efficacy of the TRK inhibitors crizotinib, which also inhibits ALK, and a more specific inhibitor,Larotrectinib (LOXO-101). Compared to crizotinib (IC50 205 nM), Larotrectinib (LOXO-101) was 10 times more potent against BaF3-ETV6-NTRK3 cells (IC5017 nM), and had no effect on other kinase fusions (ABL1, ABL2, CSF1R, FLT3, JAK2) up to 10µM. In addition, LOXO-101 was remarkably selective for TRK A, B and C in a cytotoxicity screen of 77 human cancer cell lines as compared to crizotinib. Using a PDX model of ETV6-NTRK3, we demonstrate that treatment with LOXO-101 (200mg/kg/day p.o for six weeks) reduced leukemic infiltration to undetectable levels in the bone marrow (0 vs 75.8% human CD45/CD19 bone marrow blasts, n=5 each group) and spleen compared to vehicle-treated mice (splenic weight 316 vs 20mg, p<0.001). Notably, treatment with dexamethasone had a modest effect against this tumor (average 55.3% bone marrow blasts and spleen weight 134mg, n=5). Mice treated with LOXO-101 were still alive and leukemia-free four weeks after the cessation of treatment, as determined by Xenogen imaging. NTRK-fused cancer cells (KM12, DFSP, ETV6-NTRK3-ALL) were seeded in 96-well plates at 3×103 cells/well and allowed to adhere overnight. Serial dilutions of Larotrectinib were added, and cells were incubated for 72 hours at 37°C in 5% CO2. Cell viability was measured using a colorimetric assay to determine antiproliferative IC50 [1][3] TRK signaling inhibition assay: NTRK-fused cells were pretreated with Larotrectinib (0.1–50 nM) for 1 hour, then lysed. Cell lysates were analyzed by Western blot using anti-phospho-TRK, anti-phospho-ERK1/2, anti-phospho-AKT, and total protein antibodies [1] Colony formation assay: ETV6-NTRK3-ALL cells were seeded in 6-well plates at 500 cells/well, treated with Larotrectinib (0–50 nM), and incubated for 14 days. Colonies were stained with crystal violet and counted to assess inhibitory effects [3] Apoptosis assay: NTRK-fused cells were treated with Larotrectinib (0–100 nM) for 48 hours, stained with Annexin V-FITC/PI, and analyzed by flow cytometry [1] |
| Animal Protocol |
Mice: Throughout the investigation, arthymic nude mice are employed. The mice are given a subcutaneous injection of 5x105 KM12 cells into the dorsal flank region. Tumor volume is measured directly with calipers and is computed using the following formula: length × (width5)/2. Mice are randomly chosen to receive either diluent, 60 mg/kg/dose, or 200 mg/kg/dose of Larotrectinib (LOXO-101) after the tumor has established and reached a size of 150–200 mm5. For 14 days, larotrectinib (LOXO-101) is given orally via gavage once a day. Three, six, and twenty-four hours after the final dosage, tissue and blood are extracted[4].
A patient derived xenograft (PDX) model of ETV6-NTRK3 was established by engrafting primary human ALL cells expressing luciferase into NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) mice. Phosphoflow cytometry analysis and sensitivity to LOXO-101 was assessed in vivo.[3] KM12 xenograft model: Female nude mice were subcutaneously implanted with 5×106 KM12 cells. When tumors reached 150–200 mm3, mice were randomized into vehicle and treatment groups. Larotrectinib was formulated in 0.5% hydroxypropyl cellulose + 0.1% Tween 80 and administered orally at 10 mg/kg once daily for 28 days. Tumor volume and body weight were measured twice weekly [1] ETV6-NTRK3-ALL xenograft model: Female NOD/SCID mice were intravenously injected with 2×106 ETV6-NTRK3-fused ALL cells. Larotrectinib (15 mg/kg) was administered orally once daily starting 7 days post-cell injection. Tumor burden was monitored by bioluminescence imaging, and survival was recorded for 60 days [3] Clinical dosing (infantile fibrosarcoma): A 16-month-old patient received oral Larotrectinib at 100 mg/m² twice daily (approximately 12 mg/kg/day) for 24 weeks, with doses adjusted based on body surface area [2] Clinical dosing (soft-tissue sarcoma): A 41-year-old patient received oral Larotrectinib at 100 mg twice daily (200 mg/day) for 12 weeks, with continuous administration until disease progression [4] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
The mean absolute bioavailability of larotrectinib capsules is approximately 34% (range: 32-37%). In adult patients receiving 100 mg larotrectinib capsules twice daily, peak plasma concentration (Cmax) is reached approximately 1 hour after administration and steady state is reached within 3 days. The mean steady-state peak plasma concentration (Cmax) and area under the curve (AUC0-24h) of larotrectinib capsules are 788 ng/mL and 4351 ngh/mL, respectively. In healthy subjects, the AUC of larotrectinib oral solution is similar to that of the capsules, but the peak plasma concentration (Cmax) of the oral solution is 36% higher than that of the capsules. Compared to the fasting state, healthy subjects taking larotrectinib after a high-fat meal showed similar AUC values, but the Cmax was reduced by 35%. After a single oral administration of 100 mg of radiolabeled larotrectinib to healthy subjects, 58% (5% unchanged) of the radioactive material was recovered from feces and 39% (20% unchanged) from urine. The mean volume of distribution of larotrectinib at steady state after intravenous administration to healthy subjects was approximately 48 L. The mean clearance CL/F of larotrectinib was 98 L/h. Metabolism/Metabolites Larotrectinib is primarily metabolized via CYP3A4. After a single oral administration of 100 mg of radiolabeled larotrectinib to healthy subjects, the main circulating drug components in plasma were unmetabolized larotrectinib (19%) and O-linked glucuronide (26%). Biological half-life The half-life of larotrectinib after oral administration in healthy subjects was 2.9 hours. The bioavailability of a single oral administration of 10 mg/kg larotrectinib in mice was 73%[1]. The plasma half-life (t1/2) of this compound in humans is 24 hours, and in mice it is 12 hours [1]. In humans, after oral administration of 100 mg twice daily, the peak plasma concentration (Cmax) reached 832 ng/mL 1 hour later [1]. The drug is widely distributed in tissues, with a tumor-to-plasma concentration ratio of 5.1. Four hours after oral administration to KM12 xenograft mice [1], the half-life of the drug was 360 minutes, and CYP3A4 was identified as the major metabolic enzyme [1]. |
| Toxicity/Toxicokinetics |
Hepatotoxicity
In early clinical trials, 176 patients with various solid tumors carrying NTRK gene fusions were enrolled. Among patients treated with larotrectinib, 45% experienced elevated serum transaminase levels. 6% of patients had serum transaminase levels exceeding 5 times the upper limit of normal, with 2% discontinuing treatment prematurely as a result. Elevated serum transaminase levels typically appear 4 to 12 weeks after treatment but are usually not accompanied by jaundice or elevated alkaline phosphatase. Most transaminase elevations return to normal within 4 to 8 weeks, and discontinuation is uncommon. Re-initiating larotrectinib at a lower dose after transaminase abnormalities are generally well-tolerated and do not lead to recurrence of liver injury. There are no reported cases of jaundice or other symptoms during larotrectinib treatment; however, clinical experience with this kinase inhibitor is limited, and pre-marketing clinical trials were conducted under close clinical monitoring. Probability Score: E (Unproven but suspected cause of clinically significant liver injury). Effects during pregnancy and lactation ◉ Overview of use during lactation There is currently no information on the clinical use of larotrectinib during lactation. The manufacturer recommends discontinuing breastfeeding during larotrectinib treatment and for one week after the last dose. ◉ Effects on breastfed infants No relevant published information was found as of the revision date. ◉ Effects on lactation and breast milk No relevant published information was found as of the revision date. Protein binding Larotrectinib binds to human plasma proteins in vitro at a rate of 70%, and the binding rate is independent of drug concentration. The plasma concentration ratio is 0.9. In clinical studies, adverse events associated with larotrectinib were mild to moderate, including fatigue (35%), nausea (28%), and dizziness (15%) [2][4]. In a 28-day repeated-dose toxicity study in rats, oral doses up to 100 mg/kg/day did not cause significant changes in body weight, hematological or hepatic and renal function [1]. Larotrectinib has a plasma protein binding rate of 95% in human plasma, 93% in mouse plasma, and 91% in rat plasma [1]. No significant drug interactions with CYP3A4 substrates or inhibitors were observed [1]. |
| References | |
| Additional Infomation |
Pharmacodynamics
In a series of purified enzyme activity assays, larotrectinib showed IC50 values of 5–11 nM for inhibition of TRKA, TRKB, and TRKC. The inhibitory concentration of another kinase, TNK2, was approximately 100 times that of the three kinases mentioned above. At a dose nine times the recommended adult dose, larotrectinib did not cause any clinically significant QTc interval prolongation. No dose adjustment is recommended for patients with any degree of renal impairment; however, a dose reduction should be given for patients with moderate (Child-Pugh B) to severe (Child-Pugh C) hepatic impairment. Larotrectinib (LOXO-101; ARRY-470) is a selective pan-TRK inhibitor used to treat NTRK fusion-driven cancers of all tumor types [1][2][3][4]. Its mechanism of action involves binding to the ATP-binding pocket of NTRK kinases, inhibiting their catalytic activity and downstream signaling pathways involved in cell proliferation and survival (ERK/AKT pathway) [1]. It is the first FDA-approved tissue-nonspecific cancer therapy targeting NTRK gene fusions in solid tumors and hematologic malignancies [1]. Clinical efficacy has been observed in a variety of NTRK fusion cancers, including infantile fibrosarcoma, soft tissue sarcoma, acute lymphoblastic leukemia, and colon cancer [2][3][4]. |
| Molecular Formula |
C21H22F2N6O2
|
|---|---|
| Molecular Weight |
428.44
|
| Exact Mass |
428.177
|
| Elemental Analysis |
C, 58.87; H, 5.18; F, 8.87; N, 19.62; O, 7.47
|
| CAS # |
1223403-58-4
|
| Related CAS # |
Larotrectinib sulfate;1223405-08-0;(R)-Larotrectinib;1223404-68-9
|
| PubChem CID |
46188928
|
| Appearance |
White to yellow solid powder
|
| Density |
1.6±0.1 g/cm3
|
| Index of Refraction |
1.725
|
| LogP |
1.48
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
7
|
| Rotatable Bond Count |
3
|
| Heavy Atom Count |
31
|
| Complexity |
659
|
| Defined Atom Stereocenter Count |
2
|
| SMILES |
FC1C([H])=C([H])C(=C([H])C=1[C@@]1([H])C([H])([H])C([H])([H])C([H])([H])N1C1C([H])=C([H])N2C(=C(C([H])=N2)N([H])C(N2C([H])([H])C([H])([H])[C@@]([H])(C2([H])[H])O[H])=O)N=1)F
|
| InChi Key |
NYNZQNWKBKUAII-KBXCAEBGSA-N
|
| InChi Code |
InChI=1S/C21H22F2N6O2/c22-13-3-4-16(23)15(10-13)18-2-1-7-28(18)19-6-9-29-20(26-19)17(11-24-29)25-21(31)27-8-5-14(30)12-27/h3-4,6,9-11,14,18,30H,1-2,5,7-8,12H2,(H,25,31)/t14-,18+/m0/s1
|
| Chemical Name |
(3S)-N-[5-[(2R)-2-(2,5-difluorophenyl)pyrrolidin-1-yl]pyrazolo[1,5-a]pyrimidin-3-yl]-3-hydroxypyrrolidine-1-carboxamide
|
| Synonyms |
ARRY-470; Larotrectinib; LOXO-101; LOXO 101; ARRY-470; ARRY470; LOXO101; ARRY 470; Larotrectinib; 1223403-58-4; (S)-N-(5-((R)-2-(2,5-difluorophenyl)pyrrolidin-1-yl)pyrazolo[1,5-a]pyrimidin-3-yl)-3-hydroxypyrrolidine-1-carboxamide; LOXO 101; Vitrakvi; trade name: Vitrakvi
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.84 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.84 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3340 mL | 11.6702 mL | 23.3405 mL | |
| 5 mM | 0.4668 mL | 2.3340 mL | 4.6681 mL | |
| 10 mM | 0.2334 mL | 1.1670 mL | 2.3340 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03834961 | Active Recruiting |
Drug: Larotrectinib Sulfate | Solid Neoplasm Infantile Fibrosarcoma |
Children's Oncology Group | September 18, 2019 | Phase 2 |
| NCT02637687 | Active Recruiting |
Drug: Larotrectinib (Vitrakvi, BAY2757556) |
Solid Tumors Harboring NTRK Fusion |
Bayer | December 16, 2015 | Phase 1 Phase 2 |
| NCT04655404 | Recruiting | Procedure: Larotrectinib surgical Drug: Larotrectinib |
Diffuse Intrinsic Pontine Glioma High Grade Glioma |
Nationwide Children's Hospital | April 8, 2021 | Early Phase 1 |
| NCT05783323 | Not yet recruiting | Drug: Larotrectinib monotherapy Radiation: 131I therapy |
Cancer Cancer, Thyroid |
Children's Hospital of Philadelphia |
November 2023 | Phase 2 |
| NCT02576431 | Recruiting | Drug: BAY2757556 (Larotrectinib, Vitrakvi) |
Solid Tumors Harboring NTRK Fusion |
Bayer | September 30, 2015 | Phase 2 |
|
|---|
|
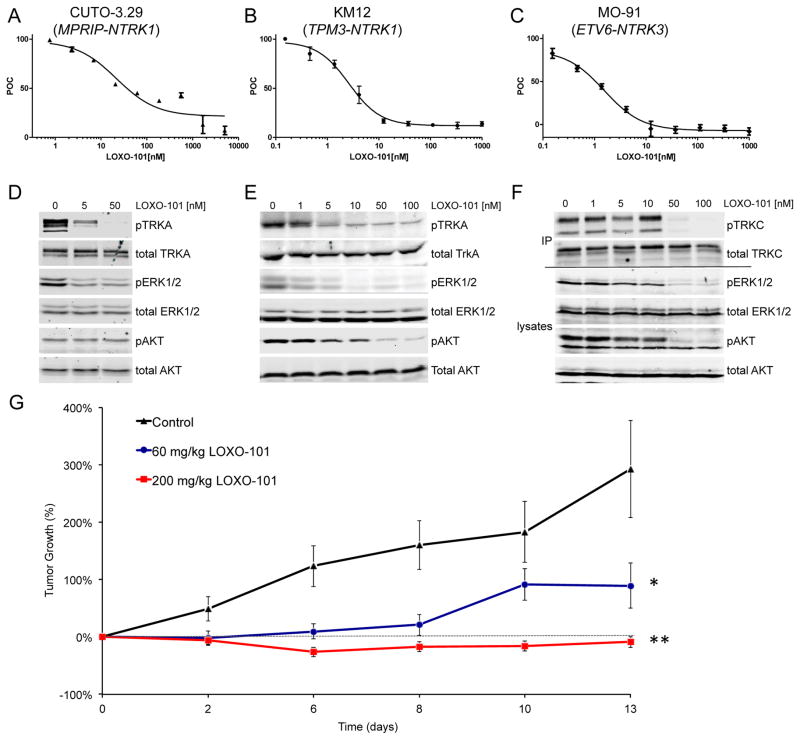 LOXO-101 inhibition of cancer cells harboring oncogenic TRK.Cancer Discov.2015 Oct;5(10):1049-57. |
 Molecular characterization of tumor sample. Cancer Discov.2015 Oct;5(10):1049-57. |
|---|
 Radiologic response to LOXO-101.Cancer Discov.2015 Oct;5(10):1049-57. |