
| Size | Price | Stock | Qty |
|---|---|---|---|
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g | |||
| 25g | |||
| Other Sizes |
Purity: ≥98%
Lamotrigine (Lamictal, BW 430C; BW430C; Crisomet, Lamictin, Lamitor), an approved anti-convulsant drug used in the treatment of epilepsy and bipolar disorder, is an inhibitor of 5-HT with IC50 of 240 μM and 474 μM in human platelets and rat brain synaptosomes. Moreover, lamotrigine blocks sodium channels.
| Targets |
Sodium channel; 5-HT (human platelets) ( IC50 = 240 μM ); 5-HT (rat brain synaptosomes) ( IC50 = 474 μM )
Lamotrigine (BW430C; LTG) is a voltage-gated sodium channel blocker, primarily inhibiting persistent sodium currents. In rat cortical neurons, it exhibits an IC₅₀ of 6.5 μM for persistent sodium currents and 25 μM for transient sodium currents [3,6] - Lamotrigine (BW430C; LTG) modulates glutamate release (a major excitatory neurotransmitter) from presynaptic terminals, with an EC₅₀ of 10 μM for reducing depolarization-induced glutamate release in rat hippocampal synaptosomes [1,6] - Lamotrigine (BW430C; LTG) has no significant affinity for GABA-A, GABA-B, or dopamine D₂ receptors (Ki > 1000 μM) in human brain membranes [1] |
|---|---|
| ln Vitro |
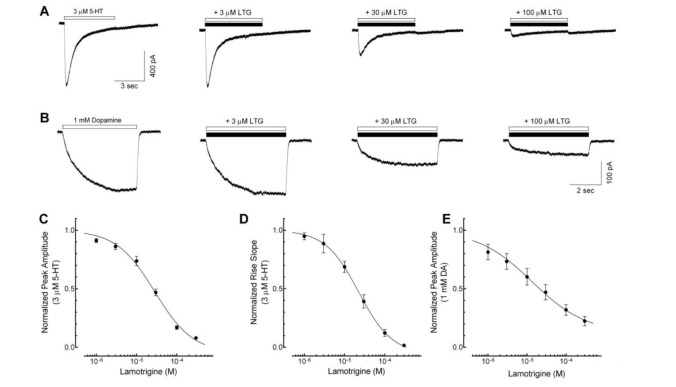
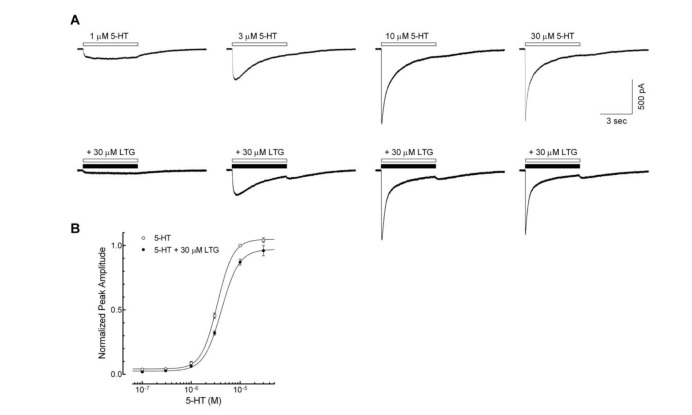
In vitro activity: Lamotrigine prevents the release of excitatory neurotransmitters, especially glutamate and aspartate, by blocking voltage-dependent sodium channels, stabilizing presynaptic neuronal membranes.[1] Lamotrigine inhibits glutamate and aspartate release (ED 50 = 5.38 mg/L for each) in rat cerebral cortex tissue incubated with veratrine 10 mg/L twice as well as GABA release (ED 50 = 11.2 mg/L). However, it is much less effective in inhibiting acetylcholine release (ED 50 = 25.6 mg/L) in cortical slices exposed to veratrine 75 mg/L. There is no change in basal glutamate release.[2] Lamotrigine directly affects voltage-activated sodium channels by inhibiting the high-frequency sustained repetitive firing of sodium-dependent action potentials. [3] Lamotrigine acts by direct inhibition at the NMDA receptor, does not produce effects on the central nervous system (CNS) similar to those of PCP, and is anticipated to be free of side effects related to NMDA blockade. [4]
Sodium Channel Inhibition: In primary cultures of rat cortical neurons, Lamotrigine (BW430C; LTG) (1–30 μM) concentration-dependently inhibits persistent sodium currents (measured via whole-cell patch-clamp): 10 μM reduces persistent current by 60%, 25 μM reduces transient sodium currents by 35%, and 30 μM completely blocks repetitive neuronal firing [3] - Glutamate Release Reduction: In rat hippocampal synaptosomes stimulated with 4-aminopyridine (4-AP, 100 μM), Lamotrigine (BW430C; LTG) (1–20 μM) dose-dependently decreases glutamate release (HPLC detection): 10 μM reduces release by 45%, 20 μM reduces release by 70%, with no effect on GABA release [1] - Neuronal Excitability Modulation: In rat hippocampal CA1 pyramidal neurons, Lamotrigine (BW430C; LTG) (5, 10, 20 μM) increases the threshold for action potential generation: 10 μM increases threshold from -45 mV to -35 mV and shortens action potential duration by 20% [6] |
| ln Vivo |
Lamotrigine inhibits the hindlimb extension induced by MES and pentetrazol in rats and mice, indicating an antiepileptic effect in these animals. These effects last for longer than 24 hours, peaking one hour after lamotrigine is administered. [4] The electrically evoked EEG after-discharge test shows that lamotrigine is active, which may suggest that it is acting against simple and complex partial seizures. Lamotrigine at intravenous doses greater than 5 mg/kg reduces the after-discharge duration in rats in a dose-dependent manner. [5]
Mouse Maximal Electroshock Seizure (MES) Model: In male CD-1 mice, oral administration of Lamotrigine (BW430C; LTG) (1, 3, 10 mg/kg) 60 min before MES (50 mA, 0.2 s) dose-dependently prevents tonic hindlimb extension (THLE): 3 mg/kg reduces THLE incidence by 50%, 10 mg/kg achieves 100% protection (ED₅₀ = 5 mg/kg) [2] - Rat Pentylenetetrazol (PTZ)-Induced Seizure Model: In male Sprague-Dawley rats, intraperitoneal (i.p.) Lamotrigine (BW430C; LTG) (2, 5, 10 mg/kg) 30 min before PTZ (80 mg/kg, i.p.) delays seizure onset: 5 mg/kg increases onset latency from 45 s (vehicle) to 120 s and reduces seizure severity (score 0–4) from 3.5 to 1.0 [4] - Chronic Epilepsy Model (Kindled Rats): In amygdala-kindled rats (chronic seizure model), daily oral Lamotrigine (BW430C; LTG) (10, 20 mg/kg) for 14 days reduces kindled seizure duration: 20 mg/kg decreases duration from 60 s (vehicle) to 25 s and reduces afterdischarge duration by 40% [5] |
| Enzyme Assay |
Rat Cortical Neuron Patch-Clamp Assay: Primary rat cortical neurons (E18-E20 embryos) were cultured for 7–10 days in DMEM + 10% FBS. Whole-cell patch-clamp recordings were performed at room temperature using an intracellular solution containing (in mM): KCl 140, MgCl₂ 1, EGTA 10, HEPES 10 (pH 7.2). The extracellular solution contained (in mM): NaCl 145, KCl 5, CaCl₂ 2, MgCl₂ 1, HEPES 10 (pH 7.4). Lamotrigine (BW430C; LTG) (1–30 μM) was added to the extracellular solution. Persistent sodium currents were evoked by a 500-ms depolarizing step from -70 mV to -30 mV; current amplitude was measured and normalized to vehicle control. IC₅₀ values were calculated via nonlinear regression [3,6]
|
| Cell Assay |
Rat Hippocampal Synaptosome Glutamate Release Assay: Rat hippocampi were homogenized in ice-cold sucrose buffer (0.32 M) and centrifuged at 1000 × g for 10 min. The supernatant was centrifuged at 17,000 × g for 20 min to isolate synaptosomes. Synaptosomes (0.5 mg protein/mL) were resuspended in Krebs-Ringer buffer (KRB: 125 mM NaCl, 4.8 mM KCl, 25 mM HEPES, pH 7.4) and pre-incubated with Lamotrigine (BW430C; LTG) (1–20 μM) for 10 min. 4-AP (100 μM) was added to stimulate glutamate release. After 5 min, reactions were terminated by centrifugation (10,000 × g for 5 min), and glutamate in supernatants was quantified via reverse-phase HPLC with fluorescence detection [1]
- Primary Cortical Neuron Excitability Assay: Rat cortical neurons were seeded on poly-L-lysine-coated coverslips (1×10⁴ cells/coverslip) and cultured for 7 days. Medium was replaced with KRB containing Lamotrigine (BW430C; LTG) (5–20 μM) for 15 min. Neuronal excitability was assessed via current-clamp recordings: action potentials were evoked by injecting depolarizing current steps (10–100 pA, 500 ms), and threshold voltage and action potential duration were measured [6] |
| Animal Protocol |
Dissolved in 0.25% methylcellulose solution; 4.5 (dogs) and 11.7 (rats) mg /kg; i.v. injection
Dog/Rat Mouse MES Model Protocol: Male CD-1 mice (20–22 g) were randomized into 4 groups (n=10/group): Vehicle (0.5% methylcellulose, p.o.), Lamotrigine 1 mg/kg (p.o.), 3 mg/kg (p.o.), 10 mg/kg (p.o.). Lamotrigine (BW430C; LTG) was dissolved in 0.5% methylcellulose (injection volume: 10 mL/kg). Sixty minutes post-drug, mice received MES via ear electrodes (50 mA, 0.2 s). Tonic hindlimb extension (THLE) was scored as a positive seizure; protection was defined as absence of THLE [2] - Rat PTZ Model Protocol: Male Sprague-Dawley rats (250–280 g) were fasted for 8 h and divided into 4 groups (n=8/group): Vehicle (normal saline, i.p.), Lamotrigine 2 mg/kg (i.p.), 5 mg/kg (i.p.), 10 mg/kg (i.p.). Thirty minutes post-injection, rats received PTZ (80 mg/kg, i.p.) dissolved in normal saline. Seizure onset latency (time to first myoclonic jerk) and severity (0=no seizure, 4=tonic-clonic seizure) were recorded for 30 min [4] - Kindled Rat Model Protocol: Male Wistar rats (220–250 g) were implanted with a bipolar electrode in the right amygdala. Kindling was induced by daily electrical stimulation (100 μA, 1 ms pulse, 50 Hz for 1 s) until 5 consecutive stage 5 seizures (Racine scale) were achieved. Rats were then randomized into 3 groups (n=7/group): Vehicle (0.5% methylcellulose, p.o.), Lamotrigine 10 mg/kg (p.o.), 20 mg/kg (p.o.). Drug was administered daily for 14 days; kindled seizures were induced on day 15, and duration and afterdischarge duration were recorded [5] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Lamotrigine is rapidly and completely absorbed with minimal first-pass metabolism; its bioavailability is estimated at 98%. Peak plasma concentration (Cmax) is reached 1.4 to 4.8 hours after administration, but this depends on the dose, concomitant medications, and epileptic status. The absorption rate and extent are considered comparable between compressed tablets taken with water and chewable dispersible tablets taken with or without water. Lamotrigine is primarily excreted in urine and feces. After oral administration of 240 mg of radiolabeled lamotrigine, approximately 94% of the drug and its metabolites are recovered in urine, and 2% in feces. A pharmacokinetic study showed that 43% to 87% of the lamotrigine dose recovered in urine, primarily as glucuronidated metabolites. 2-N-glucuronide is also primarily excreted in urine. After oral administration, the mean apparent volume of distribution (Vd/F) of lamotrigine ranges from 0.9 to 1.3 L/kg, regardless of the dose. Lamotrigine accumulates in the kidneys of male rats and may exhibit similar properties in humans. Lamotrigine also binds to melanin-containing tissues, such as the eyes and pigmented skin. The mean apparent plasma clearance (Cl/F) ranges from 0.18 to 1.21 mL/min/kg. This value varies depending on the dosing regimen, concomitant antiepileptic drugs, and individual disease status. In one study, the clearance in healthy volunteers receiving lamotrigine monotherapy was approximately 0.44 mL/min/kg after a single dose. /Breast Milk/ Lamotrigine is distributed into breast milk. Because lamotrigine can cause serious adverse reactions in nursing infants, the importance of the drug to the mother should be weighed when deciding whether to discontinue breastfeeding or discontinue the medication. /MILK/ This study aimed to investigate the pharmacokinetics of lamotrigine (LTG) during labor, the neonatal period, and lactation. High-performance liquid chromatography (HPLC) was used to determine the plasma and breast milk concentrations of LTG in 9 pregnant women with epilepsy treated with LTG and their 10 infants. Samples were collected at delivery, during the first 3 days postpartum, and during lactation 2–3 weeks postpartum. At delivery, maternal plasma LTG concentrations were similar to those in cord blood, indicating extensive transplacental transport of LTG. Neonatal plasma LTG concentrations decreased slowly. At 72 hours postpartum, the median infant plasma LTG concentration was 75% of the cord blood concentration (range: 50–100%). At 2–3 weeks postpartum, the median breast milk/maternal plasma concentration ratio was 0.61 (range: 0.47–0.77), and the LTG plasma concentration in lactating infants remained at approximately 30% of the maternal plasma level (median, range: 23–50%). In the first two weeks postpartum, maternal plasma lamotrigine (LTG) concentrations significantly increased, with a median increase of 170% in the plasma concentration/dose ratio. Lamotrigine can bind to melanin-rich eye tissues in pigmented rats and cynomolgus monkeys, but this has not been reported in humans… Long-term use may lead to drug accumulation in melanin-rich tissues (including eye tissues) and may produce toxic effects; therefore, clinicians should be aware of potential ocular adverse reactions caused by drug binding to melanin. This study aimed to determine the relative bioavailability of lamotrigine (LTG) chewable dispersible tablets after rectal administration. A two-period crossover design was used, with a 2-week washout period between administration periods. Twelve healthy adult volunteers were included. 100 mg of LTG chewable dispersible tablets were administered orally and rectally, respectively. Plasma samples were collected before and within 120 hours after administration. Lamotrigine (LTG) concentrations in the samples were analyzed using high-performance liquid chromatography (HPLC), and their relative bioavailability was determined. Drug concentrations after rectal administration were lower than those after oral administration. The relative bioavailability (F = AUC(rectal)/AUC(oral)) was 0.52 ± 0.23 (SD). Lamotrigine chewable dispersible tablets are absorbed rectally, but to a lesser extent than orally. Rectal administration of these tablet suspensions may be an acceptable route of administration. For more complete data on absorption, distribution, and excretion of lamotrigines (9 in total), please visit the HSDB record page. Metabolisms/Metabolites Lamotrigine undergoes glucuronidation primarily to form 2-N-glucuronide conjugates, a pharmacologically inactive metabolite. In clinical trials, the total radioactivity detected after administration of 240 mg of radiolabeled lamotrigine was as follows: parent lamotrigine (10%), 2-N-glucuronide (76%), 5-N-glucuronide (10%), 2-N-methyl metabolite (0.14%), and various other minor metabolites (4%). Metabolites of [(14)C]lamotrigine (78 μmol/kg) administered intravenously to adult male Wistar rats were characterized, with particular attention paid to the thioether derivatives of the epoxide intermediate. Radioactivity recovered in bile from anesthetized and intubated animals within 4 hours was 7.3 ± 3.0% (mean ± standard deviation, n = 4) of the administered dose; radioactivity recovered in bladder urine after 4 hours was 5.5 ± 0.5% of the administered dose. The bile contained [(14)C]lamotrigine (1.4 ± 0.3%), the glutathione adduct of [(14)C]dihydrohydroxylamotrigine (1.8 ± 0.3%) (i.e., the aromatic oxide adduct), and the glutathione adduct of [(14)C]lamotrigine (1.5 ± 0.7%), cysteylglycine adduct (1.9 ± 0.5%), and N-acetylcysteine adduct (0.4 ± 0.2%). The formation of thioether metabolites can be partially blocked by the cytochrome P450 inhibitor ketoconazole. Urine contained [(14)C]lamotrigine (4.5±0.5%) and [(14)C]lamotrigine N-oxide (0.9±0.2%). Almost all of the radiolabeled material in the skin (15.6±1.4%) was [(14)C]lamotrigine. … Lamotrigine is primarily metabolized via glucuronide conjugation. The main metabolite is the inactive 2-N-glucuronide conjugate. Lamotrigine is mainly excreted in urine and feces, primarily as unchanged lamotrigine (10%), 2-N-glucuronide (76%), 5-N-glucuronide (10%), 2-N-methyl metabolite (0.14%), and other unidentified minor metabolites (4%). (A308) Half-life: 25 ± 10 hours (healthy individuals); 42.9 hours (chronic renal failure) Biological half-life The mean elimination half-life of lamotrigine is approximately 14–59 hours. This value depends on the dose, concomitant medications, and disease state. A pharmacokinetic study showed a half-life of 22.8–37.4 hours in healthy volunteers. The study also noted that enzyme-inducing antiepileptic drugs such as phenobarbital, phenytoin, or carbamazepine shorten the half-life of lamotrigine. On the other hand, valproic acid prolongs the half-life of lamotrigine (48–59 hours). Researchers described the findings of a patient who intentionally ingested a large dose of lamotrigine (labeled dose 4.5 g, estimated absorption 2.9 g)… The peak concentration of lamotrigine was measured at 35.8 mg/L, with a half-life of 19.5 hours, indicating linear kinetics in the event of overdose. The plasma half-life of a single dose is 24 to 35 hours. Administration of phenytoin sodium, carbamazepine, phenobarbital, or primidone can shorten the half-life of lamotrigine to approximately 15 hours and reduce its plasma concentration. Oral Absorption: In healthy volunteers (n=6), the peak plasma concentration (Cmax) of oral lamotrigine (BW430C;LTG) (100 mg) was 2.5 μg/mL, with a time to peak concentration of 2–3 hours (Tmax), and an absolute oral bioavailability of 80% (no significant first-pass metabolism) [1,6] -Distribution and Half-Life: The volume of distribution (Vd) of lamotrigine (BW430C;LTG) in humans is 0.9–1.3 L/kg, and it exhibits moderate blood-brain barrier penetration (brain-plasma ratio of 0.8 in rats). In humans, the terminal elimination half-life (t₁/₂) of lamotrigine (BW430C; LTG) is 24–30 hours (monotherapy), which can be extended to 40–50 hours when used in combination with sodium valproate (a glucuronidation inhibitor) [1,6]. Metabolism and excretion: Lamotrigine is primarily metabolized in the liver by UDP-glucuronyl transferases (UGT1A4, UGT2B7) to produce inactive glucuronide conjugates. Approximately 70% of the dose is excreted in the urine (as metabolites) within 72 hours, 10% in the feces, and <10% is excreted unchanged [1,6]. |
| Toxicity/Toxicokinetics |
Toxicity Summary
Identification and Uses: Lamotrigine is a white to pale off-white powder. Lamotrigine is an anticonvulsant and may also be used to treat bipolar disorder. Human Exposure and Toxicity: Lamotrigine is associated with a variety of side effects, including rashes that may develop into Stevens-Johnson syndrome or toxic epidermal necrolysis. It is also associated with the development of motor tic disorders, most commonly on the head, neck, and shoulders. Life-threatening rashes associated with lamotrigine almost always occur within 2–8 weeks of starting treatment; however, severe rashes are rare after prolonged treatment (e.g., 6 months). Lamotrigine-related rashes appear to have no distinct characteristics. Because it is difficult to distinguish between benign rashes and rashes that may develop into severe or even life-threatening ones, lamotrigine should generally be discontinued immediately upon the appearance of a rash (unless it is determined that the rash is unrelated to the drug). However, even after discontinuation, the rash may still develop into a life-threatening, permanent disability, or disfigurement. In controlled clinical trials, 3% of adult patients receiving lamotrigine as adjunctive therapy discontinued treatment due to rash; this proportion was 4.5% in adult patients receiving lamotrigine as monotherapy; and 4.4% of pediatric patients receiving lamotrigine in controlled clinical trials discontinued treatment due to rash. Rarely, multi-organ failure and varying degrees of hepatic failure have been reported with lamotrigine as adjunctive therapy, with some cases even being fatal. The possibility of such potentially fatal adverse reactions should be considered in patients who develop signs and symptoms related to multi-organ and/or hepatic impairment after initiating lamotrigine as adjunctive therapy. During the premarketing development phase of lamotrigine, a cohort study involving 4,700 patients with epilepsy receiving adjunctive therapy with this drug (5,747 person-years of exposure) reported 20 cases of sudden death of unknown cause. Although these mortality rates were higher than the expected mortality rate in age- and sex-matched healthy (non-epileptic) individuals, they were similar to the mortality rates in similar epilepsy patients receiving antiepileptic drugs with different chemical structures. Of the 414 cases of patients receiving lamotrigine monotherapy in early pregnancy, 12 cases of severe birth defects were reported. Of the 88 cases of patients receiving lamotrigine combination therapy, including sodium valproate, in early pregnancy, 11 cases of severe birth defects were reported. Of the 182 cases of patients exposed to lamotrigine combination therapy (excluding sodium valproate) in early pregnancy, 5 cases of severe birth defects were reported. No clear pattern of severe birth defects was observed in offspring exposed to lamotrigine monotherapy or combination therapy. The risk of all severe birth defects following lamotrigine monotherapy in early pregnancy (2.9%) was similar to that in the general population and in women included in other registry studies exposed to antiepileptic drug monotherapy (3.3% to 4.5%). Lamotrigine also did not increase the incidence of structural or numerical abnormalities in in vitro human lymphocyte assays. Animal studies: In animal studies, no evidence of carcinogenicity was observed in oral administration of lamotrigine for up to 2 years at the maximum tolerated dose (30 mg/kg body weight daily in mice and 10–15 mg/kg body weight daily in rats). High-dose intraperitoneal injection of lamotrigine caused intrauterine growth retardation in mice, while repeated low-dose administration led to a dose-dependent increase in embryonic uptake, causing craniofacial and tail malformations and maternal toxicity. A study on the teratogenic activity of lamotrigine was conducted on the brain tissue of rat fetuses treated with the drug. Results showed that the experimental group of fetuses had lower birth weight, increased brain volume and diameter, increased subcortical density, and ventricular dilation. A behavioral teratogenicity study was conducted on rats during organogenesis. On day 21 postpartum, pups born to mothers treated with 5 mg/kg or higher doses of lamotrigine daily exhibited significantly prolonged open field exploration latency and decreased standing frequency. In swimming maze tests conducted between days 39 and 44 postpartum, offspring from mothers receiving 25 mg/kg lamotrigine daily completed the maze with prolonged maze completion time. In vitro Salmonella mutagenesis assays and mammalian mouse lymphoma assays did not demonstrate mutagenicity of lamotrigine. In in vivo rat bone marrow assays, lamotrigine did not increase the incidence of chromosomal structural or numerical abnormalities. One mechanism of action of lamotrigine (its relevance in humans remains to be confirmed) involves its effects on sodium channels. In vitro pharmacological studies have shown that lamotrigine inhibits voltage-sensitive sodium and/or calcium channels, thereby stabilizing neuronal membranes and ultimately regulating the release of presynaptic excitatory amino acids such as glutamate and aspartate. Studies have shown that lamotrigine binds to sodium channels in a manner similar to that of local anesthetics. Toxicity Data LD50: 250 (mg/kg) (mice) LD50: 250 (mg/kg) (rat) LD50 > 640 (mg/kg) (oral, rat) (Sawyer) LD50 > 640 (mg/kg) (oral, mouse) (Sawyer) Interactions Rash, including severe and potentially life-threatening rashes, appears to be more likely to occur in patients concurrently taking valproic acid. Valproic acid reduces the clearance of lamotrigine and increases its plasma concentration by more than twofold; starting with a dose higher than the recommended dose reduction or subsequent dose escalation regimen, especially in patients concurrently taking valproic acid, may increase the incidence of rashes (including severe rashes) in patients treated with lamotrigine. Clinical trials showed that 1% of adult patients and 1.2% of pediatric patients receiving treatment regimens containing immediate-release lamotrigine and valproic acid developed rashes requiring hospitalization; while 0.16% of adult patients and 0.6% of pediatric patients receiving lamotrigine-free regimens were hospitalized for rashes. Concomitant use of valproic acid and/or enzyme-inducing anticonvulsants (e.g., phenobarbital, primidone, carbamazepine, phenytoin sodium) can increase or decrease lamotrigine metabolism and clearance, thus requiring dose adjustments to maintain efficacy and/or avoid toxicity. Adding valproic acid to lamotrigine treatment reduces lamotrigine clearance and increases steady-state plasma lamotrigine concentrations by slightly more than 50%, regardless of whether enzyme-inducing antiepileptic drugs are administered concurrently. Conversely, adding phenobarbital, primidone, or carbamazepine to lamotrigine therapy can reduce steady-state plasma lamotrigine concentrations by approximately 40%, while adding phenytoin sodium can reduce steady-state plasma lamotrigine concentrations by approximately 45-54%; the effect of phenytoin sodium depends on its total daily dose (100-400 mg). Discontinuation of enzyme-inducing antiepileptic drugs is expected to prolong the elimination half-life and plasma concentrations of lamotrigine, while discontinuation of valproic acid is expected to decrease the elimination half-life and plasma concentrations of lamotrigine. Although the manufacturer states that the therapeutic plasma concentration range of lamotrigine has not been determined and that the dosage should be determined based on treatment response, changes in lamotrigine plasma concentrations caused by enzyme-inducing antiepileptic drugs or valproic acid should be considered when adding to or discontinuing existing antiepileptic drug regimens containing lamotrigine. Adding lamotrigine to existing treatment regimens with phenytoin sodium or carbamazepine generally does not significantly alter the steady-state plasma concentrations of these concomitant medications. It has been reported that adding lamotrigine to carbamazepine treatment leads to increased plasma concentrations of the pharmacologically active metabolite of carbamazepine (carbamazepine-10,11-epoxide) and increases the incidence of certain adverse reactions (e.g., dizziness, headache, diplopia, blurred vision, ataxia, nausea, nystagmus). However, no sustained increase in carbamazepine-10,11-epoxide plasma concentrations and/or increased toxicity has been observed with the co-administration of lamotrigine and carbamazepine, and the interaction mechanism between these drugs remains unclear. In healthy individuals, adding lamotrigine to valproic acid treatment resulted in a 25% decrease in valproic acid trough concentrations over 3 weeks, after which the concentrations stabilized. For more complete data on interactions of lamotrigines (11 in total), please visit the HSDB record page. Plasma protein binding: In human plasma (as determined by ultrafiltration), the protein binding of lamotrigine (BW430C; LTG) at concentrations of 0.5–10 μg/mL was 55%, independent of concentration [1,6] -Acute toxicity: The oral LD₅₀ of lamotrigine (BW430C; LTG) in male Sprague-Dawley rats was >500 mg/kg; the oral LD₅₀ in mice was >300 mg/kg. No death or serious toxicity (convulsions, hepatotoxicity) was observed at rat doses up to 200 mg/kg [1] -Chronic toxicity: In a 90-day rat study (dose: 10, 50, 200 mg/kg/day), the no adverse effect dose (NOAEL) was 50 mg/kg/day. Mild ataxia and increased liver weight (15%) were observed at a daily dose of 200 mg/kg, but no histopathological changes were observed [6] - Drug interactions and safety: Lamotrigine (BW430C; LTG) in combination with carbamazepine (CYP3A4 inducer) shortens its half-life (t₁/₂) to 12-15 hours. Rare but serious side effects include Stevens-Johnson syndrome (occurring in approximately 0.1% of adults), especially at higher initial doses [6] |
| References | |
| Additional Infomation |
Therapeutic Uses
Anticonvulsant; Calcium channel blocker; Excitatory amino acid antagonist; Voltage-gated sodium channel blocker. ClinicalTrials.gov is a registry and results database that indexes human clinical studies funded by public and private institutions worldwide. The website is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each record on ClinicalTrials.gov includes a summary of the study protocol, including: the disease or condition; the intervention (e.g., the medical product, behavior, or procedure under investigation); the title, description, and design of the study; participation requirements (eligibility criteria); the location of the study; contact information for the study location; and links to relevant information from other health websites, such as the NLM's MedlinePlus (for patient health information) and PubMed (for citations and abstracts of academic articles in the medical field). Lamotrigine is indexed in the database. Lamoxysine is indicated as adjunctive therapy in patients aged 2 years and older for the following seizure types: partial seizures, primary generalized tonic-clonic seizures (PGTC), and generalized seizures of Lennox-Gastaut syndrome. /Included in US Product Label/ Lamioxysine is indicated for adult patients (16 years and older) with partial seizures who are switching from monotherapy to carbamazepine, phenytoin sodium, phenobarbital, primidone, or sodium valproate as an antiepileptic drug (AED). /Included in US Product Label/ For more complete data on the therapeutic uses of lamotrigine (6 types), please visit the HSDB record page. Drug Warning /Black Box Warning/ Warning: Severe Rash. Lamotrigine may cause a severe rash requiring hospitalization and discontinuation of treatment. These rashes (including Stevens-Johnson syndrome) occurred in approximately 0.3% to 0.8% of pediatric patients (2 to 17 years old) treated with lamotrigine, and in approximately 0.08% to 0.3% of adult patients treated with lamotrigine. In a prospective cohort study of 1,983 children (2 to 16 years old) with epilepsy receiving adjunctive lamotrigine therapy, one case of rash-related death was reported. Global post-marketing experience suggests rare reports of toxic epidermal necrolysis and/or rash-related deaths in both adult and pediatric patients, but the number of cases is too small to accurately estimate the incidence. Apart from age, no other known factors currently predict the risk or severity of lamotrigine-induced rashes. Studies suggest (but have not yet confirmed) that the following factors may also increase the risk of rash: (1) co-administration of lamotrigine with valproate (including valproic acid and divalproate sodium); (2) an initial dose of lamotrigine exceeding the recommended dose; or (3) a dose escalation of lamotrigine exceeding the recommended dose. However, rash cases may occur even in the absence of the above factors. Almost all life-threatening rash cases caused by lamotrigine occur within 2 to 8 weeks after the start of treatment. However, there are also isolated cases that occur after prolonged treatment (e.g., 6 months). Therefore, the duration of treatment cannot be relied upon to predict the potential risk indicated by the first appearance of a rash. Although lamotrigine can also cause benign rashes, it is not reliable to predict which rashes will develop into severe or life-threatening ones. Therefore, lamotrigine should generally be discontinued immediately upon the appearance of a rash unless the rash is clearly unrelated to the drug. Discontinuation of the drug may not prevent the rash from developing into a life-threatening or permanent disability or disfigurement. In controlled trials of lamotrigine as adjunctive therapy, dizziness, headache, and ataxia were the most common neurological adverse reactions, occurring in 38%, 29%, and 22% of adult patients, respectively. Clinical trials have shown that the incidence of dizziness and ataxia, and the proportion of patients discontinuing lamotrigine due to these adverse reactions, are dose-related. In a dose-response study, 54%, 31%, and 27% of patients, respectively, experienced dizziness after receiving lamotrigine 500 mg/day, lamotrigine 300 mg/day, or placebo, while 28%, 10%, and 10% of patients, respectively, experienced ataxia after receiving the corresponding treatment regimen. In controlled clinical trials, 14% and 6% of adult patients receiving lamotrigine as adjunctive therapy experienced somnolence or insomnia, respectively. Among adult patients receiving lamotrigine treatment, 6% and 4%, respectively, reported motor incoordination or tremor… In controlled clinical trials, among adult patients receiving lamotrigine as adjunctive therapy, 4% experienced depression, 4% experienced anxiety, 3% experienced irritability, 3% experienced speech disorders, and 2% experienced inattention. In controlled trials, among adult patients receiving lamotrigine as adjunctive therapy, 3% and 2%, respectively, reported seizures or exacerbations of epilepsy; lamotrigine treatment also reported an increase in seizure frequency. In clinical trials, 7 out of 2343 adult patients receiving lamotrigine as adjunctive therapy reported treatment-induced seizures and were definitively diagnosed with status epilepticus; however, the manufacturer notes that due to differences in the definition of treatment-induced status epilepticus among different researchers, it is difficult to obtain a valid estimate of such cases. In controlled trials, 7%, 7%, 5%, and 5% of adult patients receiving lamotrigine monotherapy experienced coordination disorders, dizziness, anxiety, and insomnia, respectively; amnesia, ataxia, fatigue, depression, hypoesthesia, hypersexuality, decreased or increased reflexes, nystagmus, and irritability occurred in 2% of patients. In controlled clinical trials, more than 1% of adult patients receiving lamotrigine adjunctive therapy experienced sensory abnormalities or fatigue, but the incidence was comparable to or higher in the placebo group. For more complete data on lamotrigine (42 total warnings), please visit the HSDB record page. Pharmacodynamics: Lamotrigine may prevent seizures and mood symptoms by stabilizing the presynaptic neuronal membrane and blocking the release of excitatory neurotransmitters such as glutamate, which is a contributing factor to seizures. Regarding cardiovascular effects: Lamotrigine's metabolite, 2-N-methyl metabolite (formed by glucuronidation), has been reported to cause dose-dependent prolongation of the PR interval, widening of the QRS complex, and, at high doses, complete atrioventricular block. Although this harmful metabolite exists in trace amounts in the human body, its plasma concentration may be elevated in conditions that reduce drug glucuronidation, such as liver disease. Lamotrigine (BW430C; LTG) is a second-generation antiepileptic drug (AED) approved by the FDA in 1994 for the treatment of partial seizures, primary generalized tonic-clonic seizures, and type I bipolar disorder (maintenance therapy) [1,6] - Mechanism of action: Its antiepileptic and mood-stabilizing effects involve two key mechanisms: 1) blocking voltage-gated sodium channels to inhibit persistent neuronal overexcitation; 2) reducing the release of presynaptic glutamate (a major excitatory neurotransmitter) to prevent seizure spread and mood disturbances [1,3] - Clinical efficacy: In a 12-week trial (n=400 patients with partial epilepsy), lamotrigine (BW430C; LTG) (200 mg/day, orally) reduced seizure frequency by 50% or more in 60% of patients, compared to only 25% in the placebo group. It can also improve quality of life by reducing epilepsy-related disability[5] - Dosage Precautions: Lamotrigine (BW430C; LTG) requires gradual dose increases (starting at 25 mg/day) to minimize the risk of rash. Due to slowed metabolism, it is not recommended for patients with severe hepatic impairment[6] |
| Molecular Formula |
C9H7CL2N5
|
|
|---|---|---|
| Molecular Weight |
256.09
|
|
| Exact Mass |
255.007
|
|
| Elemental Analysis |
C, 42.21; H, 2.76; Cl, 27.69; N, 27.35
|
|
| CAS # |
84057-84-1
|
|
| Related CAS # |
Lamotrigine-13C3,d3; 1246815-13-3; Lamotrigine-13C3; 1188265-38-4; Lamotrigine hydrate; 375347-20-9; Lamotrigine-13C,d3; 2517756-06-6; Lamotrigine-13C2,15N; 2483830-10-8; Lamotrigine-d3; 1132746-94-1
|
|
| PubChem CID |
3878
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.6±0.1 g/cm3
|
|
| Boiling Point |
503.1±60.0 °C at 760 mmHg
|
|
| Melting Point |
177-181°C
|
|
| Flash Point |
258.1±32.9 °C
|
|
| Vapour Pressure |
0.0±1.3 mmHg at 25°C
|
|
| Index of Refraction |
1.706
|
|
| LogP |
-0.19
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
5
|
|
| Rotatable Bond Count |
1
|
|
| Heavy Atom Count |
16
|
|
| Complexity |
242
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
ClC1C(Cl)=C(C2C(N)=NC(N)=NN=2)C=CC=1
|
|
| InChi Key |
PYZRQGJRPPTADH-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C9H7Cl2N5/c10-5-3-1-2-4(6(5)11)7-8(12)14-9(13)16-15-7/h1-3H,(H4,12,13,14,16)
|
|
| Chemical Name |
6-(2,3-dichlorophenyl)-1,2,4-triazine-3,5-diamine
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (9.76 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (9.76 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (9.76 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 0.5% methylcellulose: 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.9049 mL | 19.5244 mL | 39.0488 mL | |
| 5 mM | 0.7810 mL | 3.9049 mL | 7.8098 mL | |
| 10 mM | 0.3905 mL | 1.9524 mL | 3.9049 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Lithium Versus Lamotrigine in Bipolar Disorder, Type II
CTID: NCT06184581
Phase: Phase 4 Status: Recruiting
Date: 2024-06-07
 |
|---|
 |
 |