
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
Purity: =99.95%
K02288 (K-02288; K 02288) is a potent, 2-aminopyridine-based and selective small molecule inhibitor of the BMP (bone morphogenetic protein) signaling pathway with the potential to treat the progressively debilitating musculoskeletal disease fibrodysplasia ossificans progressiva (FOP). It inhibits ALK2 (activin receptor-like kinase-2), ALK1 and ALK6 with IC50s of 1.1, 1.8, 6.4 nM, respectively.
| Targets |
ALK1 (IC50 = 1.8 nM); ALK2 (IC50 = 1.1 nM); ALK3 (IC50 = 34.4 nM); ALK6 (IC50 = 6.3 nM); ALK4 (IC50 = 302 nM); ALK5 (IC50 = 321 nM)[1]
K02288 targets ALK2 (IC50=17 nM), ALK3 (IC50=47 nM), and ALK6 (IC50=23 nM), with weak inhibitory activity against ALK1, ALK4, ALK5, ALK7 (IC50>1000 nM) [1] K02288 selectively targets ALK1 (IC50=21 nM) and cross-reacts with ALK2 (IC50=17 nM), ALK3 (IC50=47 nM), ALK6 (IC50=23 nM) [2] |
|---|---|
| ln Vitro |
K02288 exhibits an apparent IC50 of 100 nM in reducing the strong phosphorylation of Smad1/5/8 induced by BMP4 stimulation. Smad2 phosphorylation is nearly entirely inhibited by K02288 at 0.5 μM [1]. With two hydrogen bonds attached to the kinase hinge, K02288 binds to ALK1 in a manner reminiscent of ATP. In HUVECs, K02288 also causes a hypergermination phenotype and inhibits BMP9-ALK1 signaling [2].
K02288 dose-dependently inhibited BMP-induced Smad1/5/8 phosphorylation in C2C12 myoblasts, with an EC50 of ~30 nM for suppressing BMP2-induced Id1 mRNA expression [1] K02288 blocked BMP6-induced hepcidin mRNA expression in HepG2 hepatocytes, achieving 50% inhibition at an EC50 of ~45 nM [1] K02288 abrogated ALK1-mediated Smad1/5/8 phosphorylation in HUVECs, completely inhibiting TGF-β1-induced Smad1/5/8 phosphorylation at 100 nM [2] K02288 suppressed HUVEC proliferation (50% inhibition at 50 nM), migration (45% inhibition at 50 nM), and tube formation (60% reduction at 50 nM) in a dose-dependent manner [2] K02288 disrupted the cooperativity between Notch and ALK1 signaling in HUVECs, reducing mRNA expression of Notch target genes Hey1 (40% reduction) and Hey2 (35% reduction) at 100 nM [2] |
| ln Vivo |
K02288 Induces Dorsalization of Zebrafish Embryos[1]
To further validate K02288 as a pharmacological tool we tested its effects on Tg(BRE:mRFP) transgenic zebrafish embryos, which expressed monomeric red fluorescent protein (mRFP) under the control of a BMP response element. Intact BMP signaling has been shown to be essential for proper specification of tissue progenitors across the dorsoventral axis. Treatment with K02288 induced a dorsalized phenotype in a dose dependent manner (Figure 5A), as shown previously for dorsomorphin. A severely dorsalized phenotype was observed at 8–10 µM concentration (Figure 5B) correlating with the loss of expression of mRFP protein (Figure 5C).[1] K02288 causes dysfunctional angiogenesis in a chick embryo CAM model[2] In the chick embryo CAM model, K02288 (1 μM) promotes defective angiogenesis [2].The hypersprouting effects induced by K02288 were reminiscent of those observed upon disruption of the Notch pathway [8], suggesting that K02288 increased tip cell specification potentially resulting in dysfunctional vessel formation. To assess whether K02288 may interfere with angiogenesis in vivo, we used chick embryo chorioallantoic membrane (CAM) models which allow easy visualisation and quantification of angiogenesis. Again, the effects of K02288 were similar to those observed with ALK1-Fc (Fig. 4a). Two distinct phenotypes of disrupted angiogenesis were observed with both treatments. A subset of the treated CAM models displayed hypersprouting consistent with the 3D culture models. Moreover, shadows and halos were observed around the angiogenic sprouts suggestive of leaky dysfunctional vessels. A large fraction of the CAM models exhibited a distinctive phenotype of low vessel density reflecting the dysfunctional angiogenesis which occurs following hypersprouting (Fig. 4a). Thus, the small molecule inhibitor K02288 has potential to inhibit angiogenesis in vivo similarly to ALK1-Fc.[2] K02288 reduced corneal neovascularization area by 58% in a mouse corneal angiogenesis model when administered topically (5 μg/eye, twice daily for 7 days) [2] K02288 inhibited tumor angiogenesis and growth in a mouse Lewis lung cancer xenograft model: intraperitoneal injection (30 mg/kg, three times weekly for 3 weeks) decreased tumor microvessel density by 42% and suppressed tumor volume growth by 35% [2] |
| Enzyme Assay |
In vitro Kinase Assay for ALK1-6[1]
Kinase reactions for ALK1-6 were performed at room temperature for 45 minutes in 96-well plates mixing 2.5 nM kinase, 0.5 mg/mL dephosphorylated casein (Sigma), 6 µM ATP, 0.05 µCi/µL [γ-32P]ATP, 10 mM MnCl2 and 0.2% BSA in kinase buffer. Inhibitors were added at concentrations between 0 and 10 µM in kinase reaction buffer and tested in triplicate. Reactions were quenched with phosphoric acid, bound to 96-well P81 phosphocellulose filter plates and assayed with Microscint 20 scintillation fluid using a Spectramax L luminometer. Data were normalized to untreated controls at 100% enzyme activity and negative controls subtracted as background. Kinase-Glo® Assay[1] A kinase assay for ActRIIA (ACVR2) was performed using Kinase-Glo® as per manufacturer’s instructions. Briefly, the following were mixed and reacted at room temperature for 3 hours in a 96-well plate at a final volume of 100 µL, 10 nM kinase (∼EC50 at 2 hr), 0.5 mg/mL dephosphorylated casein, 10 µM ATP, 10 mM MnCl2 and 0.2% BSA in kinase buffer. Inhibitors were added at concentrations between 0 and 10 µM in kinase reaction buffer and tested in duplicate. At 15 min, 30 min, 1 hr, 2 hr, and 3 hr 20 µL aliquots of the reaction mixture was transferred to a 384-well plate and 20 µL of Kinase-Glo® was added and allowed to rest for 10 min to quench the reaction and produce light which was measured using a Spectramax L luminometer. The 2 hr time point was within the linear portion of the reaction and was used for calculations due to favourable signal-to-noise ratio and was consistent with earlier time points. Data were normalized to untreated controls at 100% enzyme activity and negative controls subtracted as background. Kinase-wide Selectivity Profiling[1] Inhibitor selectivity profiling against 200 human kinases at 0.1 and 1 µM inhibitor concentration was performed by Nanosyn (www.nanosyn.com). Recombinant ALK2, ALK3, or ALK6 kinase was incubated with their respective peptide substrates, ATP, and serial concentrations of K02288 (0.001-1000 nM) in reaction buffer at 37°C for 45 minutes. The phosphorylation reaction was terminated by adding a stop solution, and the phosphorylated substrate was detected using a colorimetric assay with a microplate reader. IC50 values were calculated by fitting the dose-response inhibition curves [1] Recombinant ALK1 kinase was mixed with a specific peptide substrate, ATP, and various concentrations of K02288 (0.001-1000 nM) in kinase assay buffer, followed by incubation at 37°C for 60 minutes. Phosphorylated substrate levels were quantified via a luminescent-based detection system, and IC50 was determined by nonlinear regression analysis of the inhibition data [2] |
| Cell Assay |
Sprouting assays[2]
HUVEC were grown as spheroids (500 cells/spheroid) and embedded in a fibrin gel as described by Nakatsu et al. [23]. K02288 or ALK1-Fc was added on top of the gel in EGM-2 media. The media was changed every 2 days. Quantification of sprout number and length was made 2 days after addition of inhibitor and images acquired after a further 4 days of treatment. Experiments were repeated at least three times, and error bars represent SEM. Transfections and dual luciferase assays[2] HUVECs were transfected with 800 ng RBPJ luciferase construct and 200 ng Renilla luciferase in a 10-cm dish using lipofectamine LTX reagent. After 24 h, cells were replated in low serum medium in 24-well plates coated with sDll4 or BSA. Cells were allowed to attach for 5 h, K02288 added for 30 min and 10 ng/mL BMP9 added for a further 16 h. Dual luciferase assays were performed according to the manufacturer’s protocol. C2C12 myoblasts were seeded in 12-well plates and cultured to 70% confluence. After serum starvation for 12 hours, cells were pretreated with K02288 (0.1-100 nM) for 1 hour, then stimulated with BMP2 (10 ng/mL) for 6 hours. Total RNA was extracted, and Id1 mRNA expression was analyzed by quantitative PCR (qPCR) using GAPDH as the reference gene [1] HepG2 hepatocytes were plated in 6-well plates and grown to 80% confluence. Following 16 hours of serum starvation, cells were treated with K02288 (0.1-200 nM) for 1 hour prior to BMP6 (20 ng/mL) stimulation for 12 hours. RNA was isolated, and hepcidin mRNA levels were measured by qPCR [1] HUVECs were seeded in 6-well plates and cultured to 80% confluence. After serum starvation for 8 hours, cells were pretreated with K02288 (10-100 nM) for 1 hour, then exposed to TGF-β1 (5 ng/mL) for 30 minutes. Cells were lysed, and protein extracts were subjected to western blot analysis using antibodies against phosphorylated Smad1/5/8 and total Smad1/5/8 [2] For HUVEC proliferation assay: cells were seeded in 96-well plates (5×10^3 cells/well) and treated with K02288 (10-100 nM) for 48 hours. Cell viability was assessed using a cell counting kit, and the inhibition rate was calculated relative to the vehicle control [2] For HUVEC migration assay: cells were seeded in the upper chamber of Transwell inserts, and K02288 (10-100 nM) was added to both upper and lower chambers. The lower chamber contained chemotactic factor, and after 24 hours of incubation, cells that migrated to the lower membrane were fixed, stained, and counted under a microscope [2] For HUVEC tube formation assay: 96-well plates were coated with Matrigel, and HUVECs (2×10^4 cells/well) were seeded with K02288 (10-100 nM). After 6 hours of incubation, tube-like structures were imaged, and the number of tubes was quantified [2] |
| Animal Protocol |
Chemical Inhibitor Treatment of Zebrafish[1]
Inhibitor stocks were diluted in DMSO and then further diluted in fish water to give the required inhibitor concentrations. To minimize the amount of inhibitors used, experiments were performed in 24-well plates with approximately 20 embryos per well in a volume of 1 mL. The chemical treatment was applied by immersing 8- to 16-cell stage Tg(BRE:mRFP) embryos (dorsalization experiment) or 12 hours old Tg(fli1a:eGFP) embryos (ISV experiment) in the fish water supplemented with DMSO or the chemical inhibitors. For the ISV experiment, embryos were manually dechorionated after bud stage before treatment. Embryos were scored and photographed at 26 or 48 hours. CAM assays[2] Fertilised chicken eggs were incubated at 37 °C with a relative air humidity of 65 %. On embryo development day 3 (EDD 3), a hole of approximately 3 mm in diameter was opened in the eggshell, and on EDD 6, the hole in the shell was extended to a diameter of approximately 3 cm. A polyethylene ring was deposited on the CAM and 100 μL of either K02288, ALK1-Fc or PBS was pipetted inside the ring. After 4 more days (EDD 10), the vessels were visualised under a microscope and representative pictures acquired. Mouse corneal angiogenesis model: C57BL/6 mice (8 weeks old) were subjected to corneal suture placement to induce neovascularization. Starting on the day of surgery, K02288 was dissolved in a mixture of DMSO and normal saline (DMSO final concentration <0.5%) to a concentration of 5 μg/μL. The solution was administered topically (5 μL/eye) twice daily for 7 days. Mice were euthanized, corneas were excised, and immunofluorescence staining was performed to visualize blood vessels [2] Mouse Lewis lung cancer xenograft model: C57BL/6 mice (6 weeks old) were subcutaneously injected with Lewis lung cancer cells (1×10^6 cells/mouse) into the right flank. On day 7 post-inoculation, K02288 was dissolved in DMSO and normal saline (DMSO final concentration <1%) and administered via intraperitoneal injection at 30 mg/kg three times weekly for 3 weeks. Tumor volume was measured every 3 days using a caliper. At the end of the experiment, mice were sacrificed, tumors were harvested, and immunohistochemical staining for CD31 (a microvessel marker) was performed [2] |
| Toxicity/Toxicokinetics |
In mouse models treated with K02288 (administered via local or intraperitoneal injection as previously described), no significant weight loss was observed compared to the carrier control group.[2] Serum levels of liver function markers (ALT, AST) and kidney function markers (BUN, Cr) in mice treated with K02288 were within the normal range and showed no significant difference compared to the control group.[2]
|
| References | |
| Additional Infomation |
Growth factor signaling pathways are tightly regulated by phosphorylation, encompassing many important kinase targets that are key areas of focus in drug development. Small molecule inhibitors of bone morphogenetic protein (BMP) receptor kinase ALK2 (ACVR1) are crucial for treating progressive fibrous ossifying dysplasia (FOP), a progressive and disabling musculoskeletal disease. The Dorsomorphin analogue, first discovered in zebrafish, is the only reported BMP inhibitor chemistry to date. By screening a detection platform containing 250 recombinant human kinases, we identified a highly selective 2-aminopyridine inhibitor, K02288, which exhibits in vitro ALK2 activity at low nanomolar concentrations, similar to the current lead compound, LDN-193189. K02288 specifically inhibits the BMP-induced Smad signaling pathway without affecting the TGF-β signaling pathway and induces dorsal lateralization in zebrafish embryos. Comparison of the crystal structures of ALK2 with K02288 and LDN-193189 revealed additional contacts in the K02288 complex, thereby enhancing shape complementarity and identifying exposed phenolic hydroxyl groups that can be used for further optimization of pharmacokinetics. The discovery of this new series of compounds provides an independent pharmacological tool for studying the BMP signaling pathway and offers multiple opportunities for preclinical development. [1] Activin receptor-like kinase 1 (ALK1, encoded by the gene ACVRL1) is a type I BMP/TGF-β receptor that mediates signal transduction in endothelial cells by phosphorylating SMAD1/5/8. During angiogenesis, budding endothelial cells differentiate into apical cells and stalk cells. ALK1 works synergistically with the Notch signaling pathway in stalk cells to induce the expression of Notch target genes HEY1 and HEY2, thereby inhibiting apical cell formation and angiogenesis budding. A soluble ALK1-Fc fusion protein has entered clinical trials as a therapeutic strategy for chelating the high-affinity extracellular ligand BMP9. This study resolved the crystal structure of the ALK1 intracellular kinase domain and investigated the effects of the small molecule kinase inhibitor K02288 on angiogenesis. K02288 inhibited BMP9-induced SMAD1/5/8 phosphorylation in human umbilical vein endothelial cells, thereby reducing SMAD and Notch-dependent transcriptional responses. In endothelial cell budding assays, K02288 treatment induced an over-budding phenotype similar to Notch inhibition. Furthermore, in chicken embryo chorioallantoic membrane angiogenesis assays, K02288 caused impaired angiogenesis. This activity may benefit small molecule inhibitors currently in preclinical development for the treatment of specific BMP-derived functional disorders, including diffuse endophytic pontine gliomas and progressive ossifying fibrosis, as well as broader applications in other areas of tumor biology. [2] K02288 is a novel small molecule inhibitor of the BMP signaling pathway that works by binding to the ATP-binding pockets of ALK2, ALK3, and ALK6, thereby blocking kinase activation and downstream Smad signaling. [1] K02288 is highly selective for ALK1, ALK2, ALK3, and ALK6, but less selective for other members of the ALK family, making it a valuable tool for studying BMP/ALK-mediated physiological and pathological processes, such as iron metabolism disorders and bone diseases. [1] K02288 blocks pathological angiogenesis. By inhibiting ALK1 signaling and disrupting the synergistic effect between ALK1 and the Notch pathway, its potential therapeutic value in tumors and angiogenesis-related diseases is highlighted. [2]
|
| Molecular Formula |
C20H20N2O4
|
|
|---|---|---|
| Molecular Weight |
352.3838
|
|
| Exact Mass |
352.142
|
|
| Elemental Analysis |
C, 68.17; H, 5.72; N, 7.95; O, 18.16
|
|
| CAS # |
1431985-92-0
|
|
| Related CAS # |
|
|
| PubChem CID |
46173038
|
|
| Appearance |
Off-white to yellow solid powder
|
|
| Density |
1.2±0.1 g/cm3
|
|
| Boiling Point |
522.2±50.0 °C at 760 mmHg
|
|
| Flash Point |
269.6±30.1 °C
|
|
| Vapour Pressure |
0.0±1.4 mmHg at 25°C
|
|
| Index of Refraction |
1.614
|
|
| LogP |
3.13
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
6
|
|
| Rotatable Bond Count |
5
|
|
| Heavy Atom Count |
26
|
|
| Complexity |
424
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
O(C([H])([H])[H])C1C(=C(C([H])=C(C=1[H])C1=C(N([H])[H])N=C([H])C(C2C([H])=C([H])C([H])=C(C=2[H])O[H])=C1[H])OC([H])([H])[H])OC([H])([H])[H]
|
|
| InChi Key |
CJLMANFTWLNAKC-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C20H20N2O4/c1-24-17-9-13(10-18(25-2)19(17)26-3)16-8-14(11-22-20(16)21)12-5-4-6-15(23)7-12/h4-11,23H,1-3H3,(H2,21,22)
|
|
| Chemical Name |
3-(6-amino-5-(3,4,5-trimethoxyphenyl)pyridin-3-yl)phenol
|
|
| Synonyms |
K02288; K 02288; 3-[6-Amino-5-(3,4,5-Trimethoxyphenyl)pyridin-3-Yl]phenol; 3-(6-amino-5-(3,4,5-trimethoxyphenyl)pyridin-3-yl)phenol; A3F; CHEMBL1230714; K-02288
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.09 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.09 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (7.09 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8378 mL | 14.1892 mL | 28.3785 mL | |
| 5 mM | 0.5676 mL | 2.8378 mL | 5.6757 mL | |
| 10 mM | 0.2838 mL | 1.4189 mL | 2.8378 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
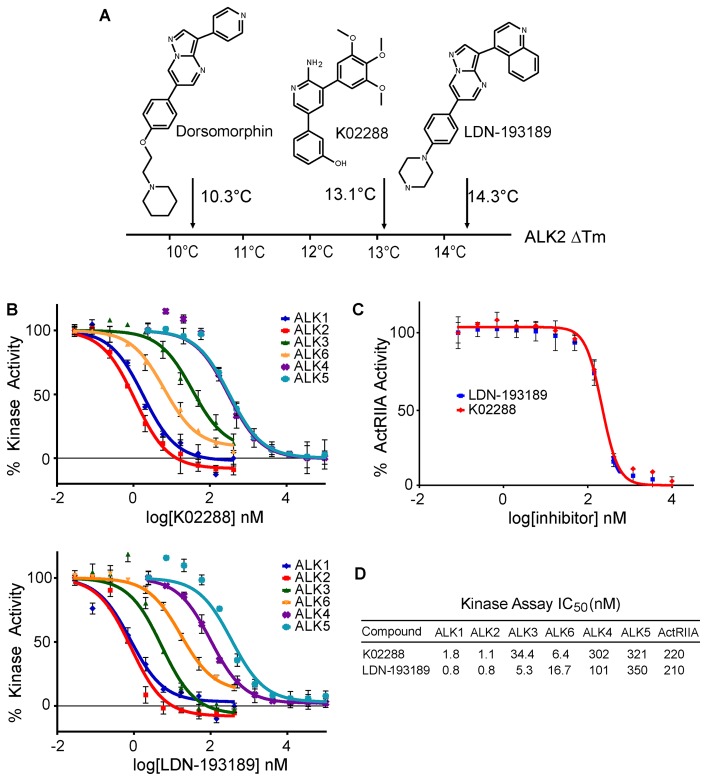 Identification of a novel 2-aminopyridine inhibitor of ALK2. |
|---|
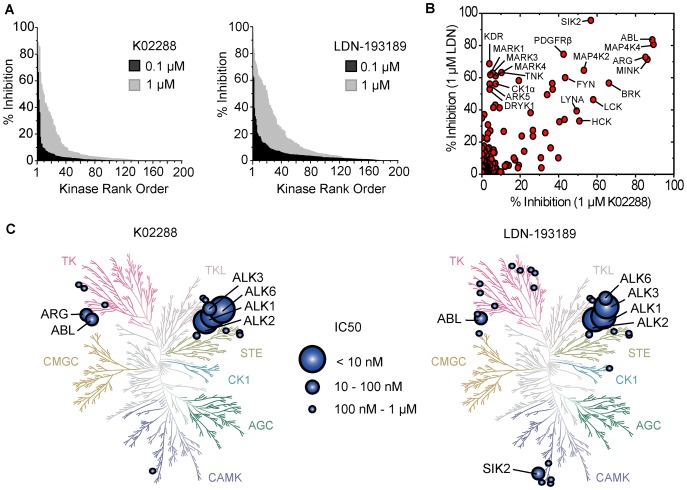 Kinome-wide selectivity of K02288 and LDN-193189.PLoS One.2013 Apr 30;8(4):e62721. |
PLoS One.2013 Apr 30;8(4):e62721. |
|
|---|
 |