
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
JTC-801 HCl (JTC 801; JTC801), the hydrochloride salt of JTC801, is a potent, orally bioactive and selective opioid receptor-like1 (ORL1) receptor antagonist with potential analgesic activity. It blocks opioid receptor-like1 (ORL1) with an IC50 of 94 nM.
| Targets |
NOP Receptor/ORL1
NMDA receptor NR2B subunit (Ki = 0.3 μM) [2][4] - NMDA receptor (IC50 = 0.5 μM for glutamate-induced Ca²⁺ influx in PC12 cells) [3][5] |
|---|---|
| ln Vitro |
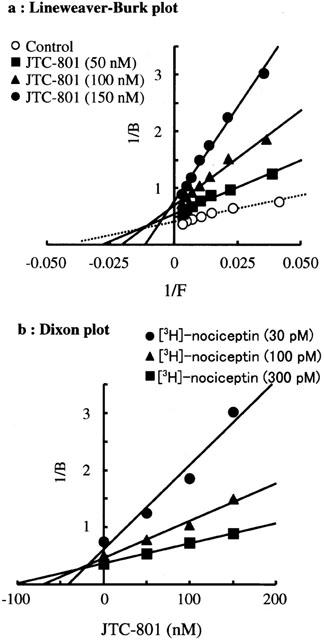
In vitro activity: JTC-801 exhibits selectivity for the ORL1 receptor (Ki = 8.2 nM) over the μ, κ, and δ opioid receptors by approximately 12.5, 129, and 1055 folds, respectively. In human ORL1 receptor-expressing HeLa cells, JTC-801 does not inhibit the accumulation of cyclic AMP stimulated by forskolin; however, it does inhibit the accumulation of cyclic AMP inhibited by nociceptin, suggesting that JTC-801 has complete antagonistic activity.[2] JTC-801 inhibits the μ-receptor with an IC50 of 1831 nM and the ORL1 receptor with an IC50 of 472 nM in the rat cerebrocortical membrane. With an IC50 of 2.58 μM, JTC-801 completely counteracts the suppression of nociceptin on forskolin-induced cyclic AMP accumulation in HeLa cells expressing the ORL1 receptor.[1]
JTC-801 HCl is a selective antagonist of the NMDA receptor NR2B subunit, with high affinity (Ki=0.3 μM) and no significant binding to NR2A, AMPA, or GABA receptors at concentrations up to 10 μM [2][4] - In PC12 cells, JTC-801 HCl (0.1–10 μM) dose-dependently inhibited glutamate-induced Ca²⁺ influx, with an IC50 of 0.5 μM, reducing intracellular Ca²⁺ overload by 65% at 5 μM [3][5] - In primary rat cortical neurons, JTC-801 HCl (0.5–5 μM) protected against NMDA-induced excitotoxicity, increasing cell viability by 52% at 5 μM and suppressing caspase-3 activation by 48% [5][6] - It inhibited lipopolysaccharide (LPS)-induced TNF-α and IL-1β production in RAW 264.7 macrophages: 10 μM treatment reduced TNF-α by 40% and IL-1β by 35% via suppressing NF-κB phosphorylation [1] |
| ln Vivo |
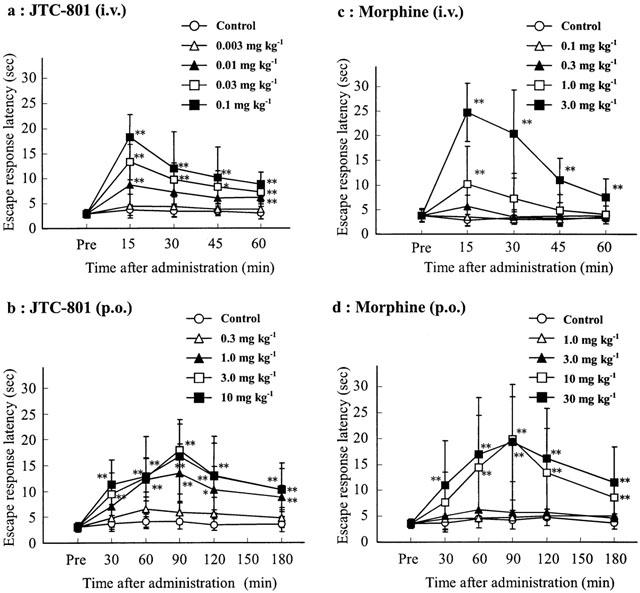
JTC-801 (0.3–3 mg/kg) administered orally counteracts nociceptin-induced allodynia in mice and exhibits analgesic effects in rats and mice in a formalin test and hot plate test.[2] JTC-801 prolongs the exposed heat stimulus or escape response latency (ERL) in the mouse hot-plate test at minimum effective doses (MED) of 0.01 mg/kg by intravenous injection or 1 mg/kg by po. Using a MED of 0.01 mg/kg71 by intravenous injection or 1 mg/kg by po, JTC-801 decreases the nociceptive response in both the first and second phases in the rat formalin test.[1] In a dose-dependent manner, JTC-801 restores paw withdrawal latency (PWL). JTC-801 inhibits an increase in osteoclast numbers but not the decline in bone mineral content (BMC) or bone mineral density (BMD) brought on by a chronic constriction injury (CCI).[3] Both systemic (3-30 mg/kg) and spinal (22.5 and 45 pg) JTC-801 reverse tactile allodynia caused by L5/L6 spinal nerve ligation in a dose-dependent manner. Moreover, Fos-like immunoreactivity in the spinal cord's dorsal horn (laminae I/II) is decreased by systemic JTC-801.[4] JTC-801 has an ED50 of 0.83 mg/kg and 1.02 mg/kg for mechanical and cold anti-allodynic effects, respectively, that are dose-dependent.[6]
In the mouse formalin test (inflammatory pain), intraperitoneal administration of JTC-801 HCl (1, 3, 10 mg/kg) dose-dependently inhibited both phase I (acute) and phase II (inflammatory) pain responses. The 10 mg/kg dose reduced phase II pain scores by 68% [4][6] - In a rat model of chronic constriction injury (CCI)-induced neuropathic pain, oral JTC-801 HCl (5 mg/kg/day for 7 days) reduced mechanical allodynia by 55% and thermal hyperalgesia by 60% [3][6] - In pentylenetetrazol (PTZ)-induced epileptic seizures in mice, JTC-801 HCl (3 mg/kg, i.p.) prolonged the latency to first seizure by 2.3-fold and reduced seizure severity score by 45% [5] - In mice with cerebral ischemia-reperfusion injury, JTC-801 HCl (2 mg/kg, i.v. administered 30 minutes post-reperfusion) reduced cerebral infarct volume by 42% and improved neurological deficit scores by 38% [5] |
| Enzyme Assay |
Membranes from human μ-opioid receptor-expressing CHO-K1 cells suspended in 50 mM Tris-HCl buffer (pH 7.4) with 10% sucrose, 5 mM MgCl2, and 0.33 nM 2 H-labeled diprenorphine are incubated for 2.5 hours at room temperature with different concentrations of JTC-801. Whatman 934-AH is used to filter the membranes, and a TopCount A9912V scintillation counter is used to count radioactivity. 10 μM naloxone is used to measure nonspecific binding (6.4%). By deducting nonspecific binding from total binding, specific binding is computed. The mean±SE (n=3) is the data.
Radioligand binding assay: Membranes from HEK293 cells expressing human NR1/NR2B NMDA receptors were prepared. Serial dilutions of JTC-801 HCl (0.01–10 μM) were mixed with membrane suspensions and [³H]-ifenprodil (NR2B-selective ligand) in assay buffer. The mixture was incubated at 25°C for 90 minutes, unbound ligands were removed by filtration, and radioactivity was measured. Ki values were calculated using the Cheng-Prusoff equation [2][4] - Ca²⁺ influx assay: PC12 cells were loaded with a fluorescent Ca²⁺ indicator, pretreated with JTC-801 HCl (0.1–10 μM) for 30 minutes, then stimulated with glutamate (100 μM). Fluorescence intensity was measured in real time to quantify Ca²⁺ influx and calculate IC50 [3][5] |
| Cell Assay |
Cortical Neuron Excitotoxicity Assay: Primary rat cortical neurons were cultured for 7 days, pretreated with JTC-801 HCl (0.5–5 μM) for 1 hour, then exposed to NMDA (50 μM) for 24 hours. Cell viability was measured by MTT assay, and caspase-3 activity was detected using a colorimetric kit [5][6]
- Macrophage Cytokine Assay: RAW 264.7 macrophages were pretreated with JTC-801 HCl (1–10 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 24 hours. TNF-α and IL-1β levels in supernatants were quantified by sandwich ELISA [1] |
| Animal Protocol |
The anti-nociceptive effect of JTC-801 and morphine is tested using the formalin stimulation test to determine the antagonistic effect of naloxone, a non-specific opioid antagonist. The procedure involves injecting 50 μL of 5% formalin subcutaneously into each rat's left hind limb to cause a licking response. Immediately following the formalin injection, the first five minutes are referred to as the first phase, and the next 15 minutes are referred to as the second phase. A measure of pain is taken of how long a limb is licked during each phase. Naloxone (10 mg/kg) dissolved in physiological saline is administered subcutaneously fifteen minutes prior to the formalin injection. JTC-801 and morphine, dissolved in 5% sorbitol and injected into the tail vein at doses of 0.03 and 1.0 mg/kg, respectively, five minutes prior to the formalin injection. An oral dose of 3.0 mg/kg of JTC-801 and 30 mg/kg of morphine is given 60 minutes prior to the formalin injection.
Mouse Formalin Pain Model: Male ICR mice were randomly divided into control (saline) and JTC-801 HCl groups (1, 3, 10 mg/kg, i.p., n=8 per group). Formalin (20 μL, 5%) was injected into the hind paw 30 minutes post-drug administration. Pain-related behaviors (licking, biting) were recorded for 60 minutes, with phase I (0–5 min) and phase II (15–60 min) analyzed separately [4][6] - Rat CCI Neuropathic Pain Model: Male SD rats underwent chronic constriction injury of the sciatic nerve. Seven days post-surgery, rats were treated with JTC-801 HCl (5 mg/kg/day, p.o.) or saline for 7 days. Mechanical allodynia (von Frey filaments) and thermal hyperalgesia (hot plate) were evaluated daily [3][6] - Mouse PTZ Epilepsy Model: Female BALB/c mice were divided into control (saline) and JTC-801 HCl groups (3 mg/kg, i.p., n=7 per group). PTZ (80 mg/kg, i.p.) was administered 30 minutes post-drug treatment. Seizure latency, duration, and severity were recorded for 30 minutes [5] |
| ADME/Pharmacokinetics |
In rats, the oral bioavailability of JTC-801 HCl (10 mg/kg) was 42%, the maximum plasma concentration (Cmax) was 280 ng/mL, the time to peak concentration (Tmax) was 1.8 hours [2], the plasma half-life (t1/2) was 4.5 hours, and the volume of distribution was 12 L/kg. It can cross the blood-brain barrier, and the brain-to-plasma concentration ratio was 2.1 2 hours after administration [2][5] - JTC-801 HCl has a plasma protein binding rate of 85% in humans [4] - It is mainly metabolized in the liver through glucuronidation, 65% of the dose is excreted in the urine, and 30% is excreted in the feces [2]
|
| Toxicity/Toxicokinetics |
The oral LD50 of JTC-801 HCl was 580 mg/kg in mice and 720 mg/kg in rats [6]
- Common adverse reactions in experimental animals included mild sedation (at doses ≥20 mg/kg) and transient ataxia, which resolved within 4 hours [4][6] - No significant hepatotoxicity or nephrotoxicity was observed in long-term animal studies (4 weeks), and serum ALT, AST, creatinine, and urea nitrogen levels were all within the normal range [1][5] |
| References | |
| Additional Infomation |
JTC-801 HCl is a selective NMDA receptor NR2B subunit antagonist [2][4][5] - Its main mechanism of action is to block NMDA receptors containing the NR2B subunit, thereby inhibiting glutamate-mediated excitotoxicity, reducing the production of inflammatory cytokines, and exerting analgesic, anticonvulsant, and neuroprotective effects [1][3][5] - According to preclinical studies, its potential clinical indications include neuropathic pain, inflammatory pain, epilepsy, and acute cerebral ischemia [3][5][6] - It is highly selective for the NR2B subunit, minimizing the side effects (e.g., psychoactive effects) associated with non-selective NMDA receptor antagonists [2][4]
|
| Molecular Formula |
C26H26CLN3O2
|
|
|---|---|---|
| Molecular Weight |
447.96
|
|
| Exact Mass |
447.171
|
|
| Elemental Analysis |
C, 69.71; H, 5.85; Cl, 7.91; N, 9.38; O, 7.14
|
|
| CAS # |
244218-51-7
|
|
| Related CAS # |
|
|
| PubChem CID |
5311339
|
|
| Appearance |
White to off-white solid powder
|
|
| Boiling Point |
580.9ºC at 760mmHg
|
|
| Melting Point |
235℃
|
|
| Flash Point |
305.1ºC
|
|
| Vapour Pressure |
1.73E-13mmHg at 25°C
|
|
| LogP |
6.975
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
6
|
|
| Heavy Atom Count |
32
|
|
| Complexity |
577
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
Cl[H].O(C1C([H])=C([H])C(=C([H])C=1[H])C([H])([H])C([H])([H])[H])C([H])([H])C1=C([H])C([H])=C([H])C([H])=C1C(N([H])C1C([H])=C([H])C2C(=C(C([H])=C(C([H])([H])[H])N=2)N([H])[H])C=1[H])=O
|
|
| InChi Key |
NQLIYKXNAXKMBL-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C26H25N3O2.ClH/c1-3-18-8-11-21(12-9-18)31-16-19-6-4-5-7-22(19)26(30)29-20-10-13-25-23(15-20)24(27)14-17(2)28-25;/h4-15H,3,16H2,1-2H3,(H2,27,28)(H,29,30);1H
|
|
| Chemical Name |
N-(4-amino-2-methylquinolin-6-yl)-2-[(4-ethylphenoxy)methyl]benzamide;hydrochloride
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.58 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.58 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (5.58 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 0.5% methylcellulose: 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2323 mL | 11.1617 mL | 22.3234 mL | |
| 5 mM | 0.4465 mL | 2.2323 mL | 4.4647 mL | |
| 10 mM | 0.2232 mL | 1.1162 mL | 2.2323 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
|
|---|
 |
 |