
| Size | Price | Stock | Qty |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
Iloperidone (Fanapt, HP-873; HP 873; Zomaril), an atypical antipsychotic agent, is a potent antagonist of dopamine (D2)/serotonin (5-HT2) receptor which has been approved for the treatment of schizophrenia symptoms.
| Targets |
Rat D2 Receptor ( Ki = 54 nM ); Rat 5-HT2 Receptor ( Ki = 3.1 nM ); Rat D1 Receptor ( Ki = 546 nM );
Rat 5-HT1A Receptor ( Ki = 168 nM ); Rat 5-HT6 Receptor ( Ki = 42.7 nM ); Rat 5-HT7 Receptor ( Ki = 21.6 nM ); Human D1 Receptor ( Ki = 216 nM ); Human D3 Receptor ( Ki = 7.1 nM ); Human D4 Receptor ( Ki = 25 nM ); Human D5 Receptor ( Ki = 319 nM ); Human 5-HT2A Receptor ( Ki = 5.6 nM ); Human 5-HT2C Receptor ( Ki = 42.8 nM ) |
|
|---|---|---|
| ln Vitro |
|
|
| ln Vivo |
Iloperidone has a mean half-life of 13.5 to 14.0 hours, indicating a slow elimination rate. AUC, tmax, and Cmax were not significantly impacted by coadministration with food. These findings suggest that taKing iloperidone with food slows down the drug's absorption rate while maintaining the same level of overall bioavailability. The most frequently reported adverse events were somnolence, dizziness, and orthostatic hypotension[2].
In vivo studies: effects of Iloperidone in animal models [3] A series of behavioural pharmacology studies were conducted in different animals Citation. Iloperidone affinity for the dopamine and noradrenaline receptors was confirmed in vivo: the drug demonstrated to be able to prevent the prepulse inhibition-disruptive effect of apomorphine (a direct dopamine receptor agonist) and cirazoline (a α1 receptor agonist) Citation. The potential efficacy of iloperidone as an antipsychotic was demonstrated in several behavioural assays including antagonised apomorphine-induced climbing behaviour in mice, pole climb avoidance in rats and continuous avoidance responding behaviour in monkeys Citation. Potential efficacy of iloperidone against negative symptoms of schizophrenia was demonstrated by a series of experiments conducted in rodents. Iloperidone, like other atypical antipsychotics, was able to increase social behaviour in unfamiliar rats, a property not observed when testing classical neuroleptics such as haloperidol Citation. Positive results were obtained in the rat-elevated plus maze assay Citation, suggestive of an anxiolytic profile of iloperidone. More recently, the effect of iloperidone on working memory of rats was examined and compared with the effects of clozapine and haloperidol using a ‘delayed non-matching-to-position’ paradigm. The study demonstrated that iloperidone, differently from haloperidol and clozapine, was able to improve choice accuracy in rats (suggestive of a positive effect on working memory), although an impaired task performance was obtained Citation. In vivo experiments were carried out to demonstrate iloperidone's low tendency for causing EPS Citation. Iloperidone, compared with risperidone and haloperidol, resulted much less potent in causing catalepsy (ED50 = 30.7 mg/kg for iloperidone; ED 50 = 0.65 mg/kg for haloperidol and ED50 = 5.7 mg/kg for risperidone). Iloperidone was also much less potent in preventing apomorphine-induced stereotyped behaviour (ED50 = 34.8 mg/kg for iloperidone; ED50 = 0.6 mg/kg for haloperidol and ED50 = 3.2 mg/kg for risperidone). Both assays can be used to evaluate EPS liability; in fact, it should be noted that clozapine does not present cataleptic activity and does not inhibit apomorphine-induced stereotyped behaviour even at toxic doses. |
|
| Enzyme Assay |
Receptor binding assays [1]
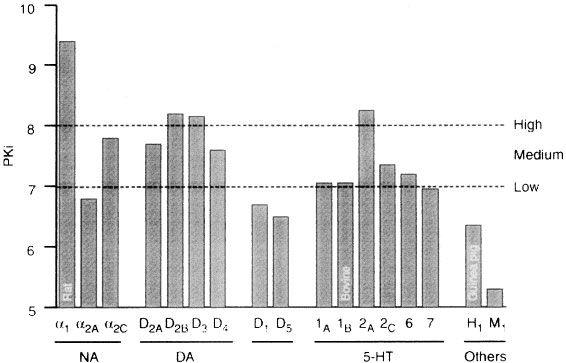
All assays were conducted at 37°C in a Tris buffer containing salts (50 mM Tris buffer, pH 7.7; 120 mM NaCl; 5 mM KCl; 2 mM CaCl2; 1 mM MgCl2), with the exception of the 5-HT2C receptor assay where a different buffer was used (50 mM Tris, 4 mM CaCl2 and 1% ascorbate, pH 7.4). Various binding parameters (ligand, ligand concentration, incubation times, ligand Kd values, displacing agent to define specific binding and tissue/cell line used) are summarized in Table 1. Except where indicated, all binding parameters were optimized at Hoechst Marion Roussel; ligand Kd values were determined using both saturation analysis (Scatchard) as well as kinetic analysis (association and dissociation rates). Membranes from rat tissues were freshly prepared; cell membranes (previously prepared and frozen) were rapidly thawed. Membranes were diluted to an appropriate concentration (between 50–500 μg protein/assay point depending on receptor expression level) in Tris buffer and homogenized. CYP2D Enzyme Activity in Brain and Liver Microsomes [4] The CYP2D activity was determined using the CYP2D specific reaction, i.e., 1′-hydroxylation of bufuralol in microsomes prepared from the brains or livers of control rats (Experiment I) and Iloperidone-treated animals (Experiment II), as described previously. The metabolism of bufuralol was investigated in terms of the linear dependence of product formation on time, substrate and protein concentration. In Experiment I (inhibition studies), the experiments were performed on the brain microsomes from the whole brain (2 mg of protein/mL) or liver microsomes (0.5 mg of protein/mL) obtained from control rats. The specific reaction, i.e., 1′-hydroxylation of bufuralol, proceeded at the substrate concentrations of 50, 100 and 200 µM for brain microsomes or 5, 10 and 20 µM for liver microsomes, in the absence or presence of in vitro added Iloperidone (1–250 µM for liver microsomes or 25–500 µM for brain microsomes), and was studied under the in vitro conditions described below. In the study with Iloperidone-treated animals (Experiment II), the bufuralol 1′-hydroxylation reaction proceeded in a system containing brain microsomes derived from selected brain structures of 1–3 rats (ca. 0.4 mg of protein/mL for the nucleus accumbens, 0.7 mg of protein/mL for the hippocampus and the substantia nigra, 1.2 mg of protein/mL for the hypothalamus, 1.5 mg of protein/mL for the striatum, the brain stem and the cerebellum and 2 mg of protein/mL for the frontal cortex and remainder of the brain) or liver microsomes (0.5 mg of protein/mL), potassium phosphate buffer (2 mM, pH = 7.4), NADP (1.6 mM), MgCl2 (4 mM), glucose 6-phosphate (5 mM) and glucose 6-phosphate-dehydrogenase (2.5 U in every sample), as described earlier.. Bufuralol was added to the incubation medium containing brain microsomes at a concentration of 125 µM or liver microsomes at a concentration of 10 µM to the final volume of 0.4 mL. The total level of microsomal protein was measured by the method of Lowry et al. using bovine serum albumin as a standard. In all experiments, the amount of 1′-hydroxybufuralol formed from bufuralol was measured by an HPLC method with fluorometric detection. Evaluation of CYP2D Protein in Brain and Liver Microsomes [4] The CYP2D protein levels in microsomes from the brains and livers of control and Iloperidone-treated animals were quantified by Western blotting, as previously described. Briefly, microsomal proteins (10 μg of brain and liver microsomes per each sample) were separated using an SDS polyacrylamide gel electrophoresis, and then the protein bands were transferred onto nitrocellulose membranes. The polyclonal rabbit anti-rat CYP2D4 antibody and polyclonal rabbit anti-human CYP2D6 antibody were used as the primary antibodies for CYP2D4 in brain microsomes and CYP2D enzymes in liver microsomes, respectively. Horseradish peroxidase-labeled goat anti-rabbit IgG was used as a secondary antibody. For the estimation of β-actin level, the primary mouse polyclonal anti-rat β-actin antibody and goat anti-mouse antibody were used. Rat cDNA-expressed CYP2D4 (2.5 µg) and human CYP2D6 (1 µg) were used as standards. The band intensity of the CYP2D protein was evaluated with the Luminescent Image Analyzer LAS-1000 and Image Gauge 3.11 programs. The collected data were normalized to protein loading based on the β-actin levels. Iloperidone (HP 873) is an atypical antipsychotic that works as a D2/5-HT2 receptor antagonist to treat symptoms of schizophrenia. Compared to the dopamine D4 receptor (Ki = 25 nM), Iloperidone exhibited a greater affinity for the dopamine D3 receptor (Ki = 7.1 nM). Iloperidone was found to have a higher affinity for the 5-HT2A receptor (Ki = 5.6 nM) than for the 5-HT2C receptor (Ki = 42.8 nM), and to have a high affinity for both the 5-HT6 and 5-HT7 receptors (Ki = 42.7 and 21.6 nM, respectively). |
|
| Animal Protocol |
Animal Treatment and Preparation of Brain and Liver Microsomes [4]
To differentiate between the direct effect of Iloperidone on the activity of CYP2D and changes evoked by their chronic in vivo treatment, two experimental approaches were applied. To study the direct effect on the CYP2D protein (inhibition), iloperidone was added in vitro to control brain or liver microsomes (Experiment I). To study the possible influence of iloperidone on CYP2D expression, the drug was administered to rats in vivo for two weeks (Experiment II). Rats (n = 12) were injected intraperitoneally once a day with a pharmacological dose of Iloperidone(1 mg/kg ip.) or vehicle (1% Tween 80 in sterile water) for a period of two weeks. The dose administered was consistent with previous pharmacological studies on rats, and the dose was active in neurochemical and behavioral paradigms. Rats were killed by decapitation 24 h after the last dose. Brains and livers were removed and the selected brain structures (in accordance with the Paxinos and Watson atlas), receiving dopaminergic and/or serotonergic innervation (the nucleus accumbens, frontal cortex, substantia nigra, striatum, hippocampus, hypothalamus, brain stem, cerebellum, and the remainder of the brain), were isolated and frozen in dry ice and stored at −80 °C until use. Microsomal fraction from the whole control brains, selected brain structures or livers was prepared by differential centrifugation, according to Hiroi et al. and Haduch et al. Brain microsomes were immediately used to determine CYP2D activity, while those of liver microsomes were stored at −80 °C until use. |
|
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
This product is well absorbed from the gastrointestinal tract, reaching peak plasma concentration (Cmax) within 2-4 hours. Steady-state plasma concentrations are reached 3-4 days after administration of iloperone. The relative bioavailability of tablets compared to oral solution is 96%. Drug accumulation occurs in a predictable manner. Renal excretion (<1% of iloperone is excreted unchanged). Apparent volume of distribution (Vd) = 1340-2800 L. Apparent clearance (clearance/bioavailability) = 47-102 L/h. The apparent clearance (clearance/bioavailability) of iloperone is 47 to 102 L/h, and the apparent volume of distribution is 1340 to 2800 L. At therapeutic concentrations, the free fraction of iloperone in plasma is approximately 3%, and the free fraction of each metabolite (P88 and P95) is approximately 8%. Most of the radiolabeled ilopiperidone was recovered in urine (mean recoveries of 58.2% and 45.1% in individuals with strong and weak CYP2D6 metabolism, respectively), and 19.9% in feces (mean recoveries of 45.1% in individuals with strong metabolism). 22.1% of the radiolabeled dose was recovered from those of metabolizers and those of poor metabolizers. Ilopiperidone tablets are well absorbed after administration, with peak plasma concentrations occurring within 2 to 4 hours; the relative bioavailability of the tablet formulation compared to the oral solution is 96%. Concomitant administration of ilopiperidone with a standard high-fat meal does not significantly affect the Cmax or AUC of ilopiperidone, P88, or P95, but it delays the Tmax of ilopiperidone by 1 hour, P88 by 2 hours, and P95 by 6 hours. Fanapt can be taken without being affected by meals. ...Metabolite P88 can penetrate the central nervous system and is thought to contribute to the drug's antipsychotic activity, while metabolite P95 does not readily penetrate the central nervous system... Metabolism/Metabolites Ilopride is metabolized in the liver by cytochrome enzymes that mediate O-dealkylation (CYP3A4), hydroxylation (CYP2D6), and decarboxylation/reduction processes. The resulting metabolites are P89, P95, and P88. P89 is a minor metabolite, while P95 and P88 are major metabolites. The affinity of ilopride metabolite P88 is generally equal to or lower than that of the parent compound. In contrast, metabolite P95 exhibits affinity only for the 5-HT2A receptor (Ki value 3.91) and NEα1A, NEα1B, NEα1D, and NEα2C receptors (Ki values 4.7, 2.7, 8.8, and 4.7 nM, respectively). Ilopiperidone is primarily metabolized via carbonyl reduction, cytochrome P-450 (CYP) isoenzyme 2D6-mediated hydroxylation, and CYP3A4-mediated O-demethylation. The drug's two major metabolites, P88 and P95, are further oxidized and/or conjugated with glucuronic acid. The P88 metabolite can penetrate the central nervous system and is thought to contribute to the drug's antipsychotic activity; while the P95 metabolite does not readily penetrate the central nervous system and primarily contributes to adverse drug reactions. Iloperone is primarily metabolized via three biotransformation pathways: carbonyl reduction, hydroxylation (mediated by CYP2D6), and O-demethylation (mediated by CYP3A4). Iloperone has two major metabolites: P95 and P88. At steady state, for rapid metabolizers (EM), the iloperone metabolite P95 accounts for 47.9% of the plasma AUC of iloperone and its metabolites; for slow metabolizers (PM), it accounts for 25%. The active metabolite P88 accounts for 19.5% and 34.0% of the total plasma exposure in rapid and slow metabolizers, respectively. Approximately 7% to 10% of Caucasians and 3% to 8% of Black/African Americans lack the ability to metabolize CYP2D6 substrates and are classified as poor metabolizers (PM), while the remaining population are classified as intermediate metabolizers, strong metabolizers, or ultrarapid metabolizers. Concomitant use of fanapt with known potent CYP2D6 inhibitors (such as fluoxetine) results in a 2.3-fold increase in iloperone plasma exposure; therefore, the dose of fanapt should be halved. Similarly, individuals with slow CYP2D6 metabolism (PM) have higher iloperone exposure compared to those with fast metabolism (EM); therefore, the dose of PM should be halved. The known human metabolites of ilopiperidone include 4-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-methoxy-α-methylbenzyl alcohol, 1-[4-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-methoxyphenyl]-2-hydroxyethyl ketone and 1-[4-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-hydroxyphenyl]ethyl ketone. Biological Half-Life The observed mean elimination half-lives were 18, 26 and 23 hours for iloperone, P88 and P95 in CYP2D6 fast metabolizers (EM), and 33, 37 and 31 hours for CYP2D6 slow metabolizers (PM). The mean elimination half-lives of iloperone, P88 and P95 were 18, 26 and 23 hours in CYP2D6 fast metabolizers, and 33, 37 and 31 hours in CYP2D6 slow metabolizers. Pharmacokinetics: Distribution and Metabolism[3] Oral iloperone is well absorbed. Human pharmacokinetic characteristics can be found in a phase I study conducted on healthy volunteers. The single dose of iloperone is 3 or 5 mg. The observed Cmax increased with increasing dose (2.2 ng/ml in the 3 mg dose group and 5.2 ng/ml in the 5 mg dose group), and the Tmax value was reached 2-3 hours after administration. Drug elimination is slow, with a half-life of 13.5-14 hours. Food administration had no significant effect on pharmacokinetics. When iloperone was taken after a meal, Tmax (increasing from 2.2 hours to 4.3 hours) and Cmax (increasing from 2.3 ng/ml to 2.0 ng/ml) increased slightly, but bioavailability remained unchanged. In Phase III clinical trials, iloperone was administered at doses ranging from 4 mg/day to 24 mg/day. A clinical trial in patients with Alzheimer's disease tested lower doses (0.5-6 mg/day) of iloperone. At concentrations ranging from 5 to 500 ng/ml, iloperone binds to proteins in the human body up to 93%; most of the administered dose (oral or intravenous) is excreted in feces; therefore, bile excretion is likely the primary clearance pathway. Recently, researchers established the clinical chemistry correlation of the minimum effective exposure level for iloperone; the results showed that a plasma iloperone concentration of 5 ng/ml can be considered the minimum therapeutic concentration. Iloperone is extensively metabolized into a variety of compounds in humans, rats, and dogs. Mutlib et al. used liquid chromatography-mass spectrometry (LC-MS) and nuclear magnetic resonance (NMR) to identify all metabolites produced in vitro and in vivo (in rats, dogs, and humans). The study found that in rats and humans, the main metabolic pathway of iloperone is the reduction of the acetophenone ring structure to generate reduced iloperone (compound I, Figure 2) [1]. This biotransformation may be primarily mediated by cytoplasmic enzymes; studies of chemical inhibitors have shown that CYP450 isoenzymes may also be involved.[1] It has been reported that in dogs, the metabolite reduced iloperone can be interconverted into iloperone.[1] To our knowledge, there is currently no data on the possible role of this reaction in humans. Recently, researchers discovered another compound (metabolite (II), Figure 2). This is another major circulating metabolite in the human body; it is generated from α-hydroxy ketone metabolites via oxidation and decarboxylation.[1] In the human body, the plasma concentrations of metabolites (I) and (II) are higher than those of the parent compound. It has been reported that when iloperone was administered at 16 mg/day, the Cmax values of compound (I) were 25 ng/ml and 40 ng/ml, respectively (the Cmax of iloperone is 20 ng/ml) [References]. Other metabolites include O-demethyl ilopiperonone (compound (III), Figure 2) and 2-hydroxyilopiperonone (compound (IV), Figure 2), which are generated via CYP3A4 and CYP2D6, respectively [References][74,76], although their concentrations in human biological fluids are much lower. However, studies have shown that in individuals with weaker CYP2D6 metabolic capacity, the elimination half-life of ilopiperonone is prolonged by 88%, and that of reduced ilopiperonone by 46% (References). Further metabolic steps include oxidation and conjugation with glucuronic acid (References). Studies have been conducted to evaluate the receptor-binding properties of the two most abundant ilopiperonone metabolites (compounds (I) and (II)) and their potential therapeutic/side effects. Studies have found that reduced ilopiperonone has a similar affinity for the D2 receptor as the parent compound, but a 2.5-fold lower affinity for the 5-HT2A receptor. Its pharmacokinetic profile is similar to that of the parent compound, suggesting that reduced ilopiperidone may have antipsychotic activity; however, although metabolite (II) has a high affinity for the 5-HT2A receptor, it cannot cross the blood-brain barrier and is likely associated with side effects rather than therapeutic effects[78]. A study evaluated the pharmacokinetics of ilopiperidone after a single dose of 3 mg or 2 mg, respectively, in patients with chronic severe renal impairment or mild to moderate hepatic impairment. The results showed that the pharmacokinetic profiles of ilopiperidone and its major metabolites were not significantly altered in patients with renal or hepatic impairment (with a slight increase in metabolite exposure in patients with hepatic impairment)[78]. |
|
| Toxicity/Toxicokinetics |
Toxicity Summary
Identification and Use: Iloperone is a white to off-white fine crystalline powder, formulated as oral tablets. Iloperone is considered an atypical or second-generation antipsychotic. It is used to treat schizophrenia in adults. Human Exposure and Toxicity: In premarketing trials involving more than 3,210 patients, eight cases of accidental or intentional overdose of iloperone were recorded, with no deaths. Typically, reported signs and symptoms are due to an overemphasis on known pharmacological effects (e.g., somnolence and sedation, tachycardia, and hypotension). The risk of death is increased in patients with Alzheimer's-related psychosis treated with iloperone; therefore, iloperone is not approved for use in this population. During pregnancy, iloperone should only be used if the potential benefit outweighs the potential risk to the fetus. Newborns exposed to antipsychotic drugs during late pregnancy (third trimester) are at risk of developing extrapyramidal symptoms and/or withdrawal symptoms after delivery. Reports have indicated that these newborns exhibit restlessness, increased muscle tone, decreased muscle tone, tremors, lethargy, respiratory distress, and feeding difficulties. The severity of these complications varies; some cases resolve spontaneously, while others require intensive care and prolonged hospitalization. It has been reported that taking antipsychotic drugs, including ilopiperidone, can trigger a potentially fatal symptom cluster, sometimes referred to as neuroleptic malignancy (NMS). Hyperglycemia has also been reported in patients taking atypical antipsychotic drugs, including ilopiperidone, sometimes severely, and may be accompanied by ketoacidosis, hyperosmolar coma, or death. Ilopiperidone has two main metabolites, P88 and P95. The P88 metabolite can penetrate the central nervous system and is thought to contribute to the drug's antipsychotic activity; while the P95 metabolite does not easily penetrate the central nervous system and primarily causes adverse drug reactions. Animal studies: Lifetime carcinogenicity studies have been conducted in mice and rats. Mice were administered ilopiperidone orally at doses of 2.5, 5.0, and 10 mg/kg/day, while rats were given oral doses up to 16 mg/kg/day. An increased incidence of malignant mammary tumors was observed only in female mice treated with the lowest dose (2.5 mg/kg/day). No treatment-related increase in tumor incidence was observed in rats. In male rats, oral doses up to 200 mg/kg/day were administered, and in female rats, the carcinogenicity of the ilopiperidone metabolite P95 was investigated at oral doses up to 250 mg/kg/day. Drug-related neoplastic lesions occurred in the pituitary gland (distal adenoma) and the pancreas (islet cell adenoma). Ilopiperidone also caused developmental toxicity, but no teratogenic effects were observed in rats and rabbits. During organogenesis, pregnant rats were administered ilopiperidone daily at doses up to 64 mg/kg. The highest dose resulted in increased early intrauterine mortality and decreased survival of full-term fetuses; this dose also caused maternal toxicity. In a similar study, pregnant rabbits were given up to 25 mg/kg of ilopiperidone daily during organogenesis. The highest dose resulted in increased early intrauterine mortality and reduced survival of full-term fetuses; this dose also caused maternal toxicity. In additional studies in rats, ilopiperidone caused adverse reproductive effects, including prolonged gestation and parturition, increased stillbirth rate, increased incidence of fetal visceral variations, decreased fetal and pup weight, and reduced postpartum pup survival, regardless of whether it was administered before conception or from day 17 of gestation until weaning. The drug had no effect on the neurobehavioral or reproductive development of surviving pups. In a study treating both male and female rats, ilopiperidone reduced fertility at doses of 12 and 36 mg/kg. Ilopiperidone was negative in the Ames test and in vivo in mouse bone marrow and liver micronucleus tests. Ilopiperidone can induce chromosomal aberrations in Chinese hamster ovary (CHO) cells in vitro, and its concentrations also cause some cytotoxicity. Hepatotoxicity In patients taking ilopridone long-term, 1% to 3% will experience abnormal liver function, but the incidence is similar in the placebo and control groups. Elevations in ALT are usually mild and transient, and usually resolve spontaneously even without dose adjustment or discontinuation. There are currently no published reports of clinically significant liver injury (with symptoms or jaundice) caused by ilopridone treatment. Probability score: E (unlikely to be the cause of clinically significant liver injury). Pregnancy and Lactation Effects ◉ Overview of Use During Lactation Due to a lack of information on the use of ilopridone during lactation, alternative medications are recommended, especially when breastfeeding newborns or premature infants. ◉ Effects on Breastfed Infants Patients taking second-generation antipsychotics while breastfeeding (n = 576) registered in the National Atypical Antipsychotics Pregnancy Registry were compared with a breastfeeding control group (n = 818) who did not take second-generation antipsychotics. Among patients taking second-generation antipsychotics, 60.4% were also taking more than one antipsychotic medication. A review of pediatric records showed no adverse effects in infants, regardless of whether they received monotherapy or combination therapy with second-generation antipsychotics. The number of women taking ilopiperidone was not reported. ◉ Effects on lactation and breast milk Ilopiperidone can cause a slight increase in serum prolactin levels. For mothers who have established lactation, the increase in prolactin levels may not affect their ability to breastfeed. Patients taking second-generation antipsychotics while breastfeeding (n = 576) registered with the National Atypical Antipsychotic Pregnancy Registry were compared with a control group of breastfeeding patients with a primary diagnosis of major depressive disorder and anxiety (n = 818), who typically received selective serotonin reuptake inhibitors (SSRIs) or selective serotonin and norepinephrine reuptake inhibitors (SNRIs) but not second-generation antipsychotics. Among women taking second-generation antipsychotics, 60.4% were taking more than one other antipsychotic medication, compared to 24.4% in the control group. 59.3% of women taking second-generation antipsychotics reported breastfeeding, compared to 88.2% in the control group. At 3 months postpartum, 23% of women taking second-generation antipsychotics exclusively breastfed, compared to 47% in the control group. The number of women taking ilopiperidone was not reported. Protein Binding: 95% of ilopiperidone is bound to proteins. Kidney or liver dysfunction or concomitant use with ketoconazole does not alter the binding rate. Drug Interactions: Due to the potential to interfere with thermoregulation, ilopiperidone should be used with caution in patients taking medications with anticholinergic activity. Because ilopiperidone has an additive effect on QT interval prolongation, it should be used with caution in combination with other drugs known to prolong the corrected QT interval (QTc), including class IA antiarrhythmic drugs (e.g., quinidine, procainamide), class III antiarrhythmic drugs (e.g., amiodarone, sotalol), certain antipsychotics (e.g., chlorpromazine, thioridazine, haloperidol, asenapine, olanzapine, paliperidone, pimozide, quetiapine, ziprasidone), certain anti-infectives (e.g., gatifloxacin; it should be avoided in combination with drugs such as moxifloxacin), and other drugs (e.g., levomethadone (discontinued in the US), methadone, pentamifil, buphenazine). Ilopride may produce additive central nervous system effects when used in combination with other central nervous system medications or alcohol. Caution should be exercised when using ilopride in combination with other central nervous system medications; alcohol should be avoided during ilopride treatment. Because ilopride has α1-adrenergic blocking activity and may cause orthostatic hypotension and syncope, the manufacturer recommends that patients taking antihypertensive drugs and other medications that may cause hypotension use ilopride with caution; orthostatic vital sign monitoring should be considered in such patients. For more complete data on drug interactions with ilopride (out of 13), please visit the HSDB record page. |
|
| References |
|
|
| Additional Infomation |
Therapeutic Uses
A non-typical antipsychotic for the treatment of negative symptoms. Fanapt tablets are indicated for the treatment of schizophrenia in adults. /US product label contains/ Drug Warnings /Black Box Warning/ Warning: Increased mortality in patients with dementia-related psychosis. Patients with dementia-related psychosis receiving antipsychotic medication have an increased risk of death. An analysis of 17 placebo-controlled trials (mean duration 10 weeks) showed that the risk of death was 1.6 to 1.7 times higher in the medication group than in the placebo group. These trials primarily involved patients taking non-typical antipsychotic medications. In a typical 10-week controlled trial, the mortality rate was approximately 4.5% in the medication group and approximately 2.6% in the placebo group. Although the causes of death varied, most deaths appeared to be related to cardiovascular disease (e.g., heart failure, sudden death) or infectious diseases (e.g., pneumonia). Observational studies suggest that, similar to non-typical antipsychotic medications, treatment with conventional antipsychotic medications may increase mortality. The extent to which the increased mortality observed in observational studies is attributable to the antipsychotic drugs themselves, rather than certain patient characteristics, is currently unclear. Fanapt is not approved for the treatment of dementia-related psychosis. Elderly patients with dementia-related psychosis treated with ilopiperidone have a higher risk of death than those treated with placebo. Furthermore, in placebo-controlled studies, an increased incidence of adverse cerebrovascular events (cerebrovascular accidents and transient ischemic attacks), and even deaths, have been observed in elderly patients with dementia-related psychosis treated with certain atypical antipsychotics (such as aripiprazole, olanzapine, and risperidone). The manufacturer states that the safety and efficacy of ilopiperidone in treating Alzheimer's disease-related psychosis have not been established, and the drug is not approved for the treatment of dementia-related psychosis. If clinicians decide to use ilopiperidone to treat such patients, the manufacturer recommends close monitoring. In placebo-controlled studies, an increased incidence of adverse cerebrovascular events (cerebrovascular accidents and transient ischemic attacks), including deaths, was observed in elderly patients with dementia-related psychosis treated with certain atypical antipsychotics (aripiperazole, olanzapine, risperidone). The manufacturer states that ilopiperidone is not approved for the treatment of dementia-related psychosis. A potentially fatal symptom cluster, sometimes referred to as neuroleptic malignancy (NMS), has been reported with antipsychotics (including fanalipone). Clinical manifestations include high fever, muscle rigidity, altered mental status (including symptoms of catatonic psychosis), and evidence of autonomic dysfunction (irregular pulse or blood pressure, tachycardia, excessive sweating, and arrhythmias). Other signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure. For more complete data on drug warnings for ilopiperidone (28 in total), please visit the HSDB record page. Pharmacodynamics Ilopride exhibits high affinity and maximum receptor occupancy for dopamine D2 receptors in the caudate nucleus and putamen of the brain in patients with schizophrenia. Improvements in cognitive function are attributed to ilopride's high affinity for α-adrenergic receptors. Ilopride also binds with high affinity to serotonin 5-HT2a receptors and dopamine 3 receptors. Ilopride binds with moderate affinity to dopamine D4 receptors, serotonin 5-HT6 and 5-HT7 receptors, and norepinephrine NEα1 receptors. Furthermore, ilopride shows weaker affinity for serotonin 5-HT1A receptors, dopamine D1 receptors, and histamine H1 receptors. Ilopiperidone (HP 873; 1-[4-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-methoxyphenyl]ethyl ketone) is a compound currently undergoing clinical trials for schizophrenia. Ilopiperidone exhibits affinity for dopamine D2 and 5-HT2A receptors and possesses a variety of in vivo activities, suggesting it may be an atypical antipsychotic. This study investigated the affinity of ilopiperidone for various human and murine dopamine and 5-HT receptor subtype homologs. We employed receptor binding assays using cell membranes stably expressing human dopamine D1, D2S, D2L, D3, D4, and D5 receptors, as well as 5-HT2A and 5-HT2C receptors, and rat 5-HT6 and 5-HT7 receptors. Iloperone has a higher affinity for dopamine D3 receptors (Ki = 7.1 nM) than for dopamine D4 receptors (Ki = 25 nM). Iloperone also has high affinity for 5-HT6 and 5-HT7 receptors (Ki = 42.7 nM and 21.6 nM, respectively) and a higher affinity for 5-HT2A receptors (Ki = 5.6 nM) than for 5-HT2C receptors (Ki = 42.8 nM). This article will explore the potential implications of this receptor binding property when compared with data from other antipsychotic drugs. [1] Iloperone is a new generation of atypical antipsychotic drug developed by Vanda Pharmaceuticals as a serotonin/dopamine (5-HT(2A)/D(2)) receptor antagonist for the treatment of schizophrenia, bipolar disorder and other mental illnesses. Ilopiperidone has a similar chemical structure to risperidone, both being benzisoxazole compounds, and like other atypical antipsychotics, it has multiple receptor binding properties. After oral administration, the drug is highly bound to plasma proteins and is extensively metabolized. Several clinical trials have been conducted to examine its efficacy, safety, and side effects. To introduce ilopiperidone as a drug for the treatment of schizophrenia, this article will briefly outline the disease and the most important antipsychotic drugs currently on the market or under development. This article introduces the pharmacokinetics and pharmacodynamics of ilopiperidone and evaluates its clinical safety and efficacy results. [3] CYP2D enzymes are involved in the synthesis of endogenous neuroactive substances (dopamine, serotonin) and the metabolism of neurosteroids. This study investigated the effects of ilopiperidone on the expression and activity of CYP2D enzymes in the rat brain and liver. In vitro experiments showed that ilopiperidone had a weak direct inhibitory effect on CYP2D activity in the liver and brain microsomes (Ki values of 11.5 μM and 462 μM, respectively). However, intraperitoneal injection of ilopiperidone (1 mg/kg) for two consecutive weeks significantly reduced hepatic CYP2D activity, and this reduction was positively correlated with decreases in CYP2D1, CYP2D2, and CYP2D4 protein and mRNA levels. Similar to the liver, ilopiperidone reduced CYP2D activity and protein levels in the frontal cortex and cerebellum, but increased CYP2D activity and protein levels in the nucleus accumbens, striatum, and substantia nigra. Long-term ilopiperidone administration did not alter brain CYP2D4 mRNA levels, with a significant increase observed only in the striatum. In conclusion, ilopiperidone may alter its pharmacological effects by affecting brain CYP2D activity, thereby influencing the synthesis rates of dopamine and serotonin or the metabolism of neurosteroids. Ilopride can alleviate extrapyramidal symptoms by increasing the expression/activity of CYP2D in the substantia nigra and striatum (i.e., the dopaminergic nigrostriatal pathway); at the same time, it can also reduce the activity of CYP2D and the metabolism of neurosteroids in the frontal cortex and cerebellum, thus having a beneficial effect on the treatment of schizophrenia. In the liver, long-term use of ilopride may have pharmacokinetic interactions with CYP2D substrates. [4] |
| Molecular Formula |
C24H27FN2O4
|
|
|---|---|---|
| Molecular Weight |
426.48
|
|
| Exact Mass |
426.195
|
|
| Elemental Analysis |
C, 67.59; H, 6.38; F, 4.45; N, 6.57; O, 15.01
|
|
| CAS # |
133454-47-4
|
|
| Related CAS # |
Iloperidone metabolite Hydroxy Iloperidone; 133454-55-4; Iloperidone hydrochloride; 1299470-39-5; Iloperidone-d3; 1071167-49-1
|
|
| PubChem CID |
71360
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.2±0.1 g/cm3
|
|
| Boiling Point |
593.7±50.0 °C at 760 mmHg
|
|
| Melting Point |
118-120°C
|
|
| Flash Point |
312.8±30.1 °C
|
|
| Vapour Pressure |
0.0±1.7 mmHg at 25°C
|
|
| Index of Refraction |
1.570
|
|
| LogP |
3.81
|
|
| Hydrogen Bond Donor Count |
0
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
8
|
|
| Heavy Atom Count |
31
|
|
| Complexity |
586
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
FC1C([H])=C([H])C2=C(C=1[H])ON=C2C1([H])C([H])([H])C([H])([H])N(C([H])([H])C([H])([H])C([H])([H])OC2C([H])=C([H])C(C(C([H])([H])[H])=O)=C([H])C=2OC([H])([H])[H])C([H])([H])C1([H])[H]
|
|
| InChi Key |
XMXHEBAFVSFQEX-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C24H27FN2O4/c1-16(28)18-4-7-21(23(14-18)29-2)30-13-3-10-27-11-8-17(9-12-27)24-20-6-5-19(25)15-22(20)31-26-24/h4-7,14-15,17H,3,8-13H2,1-2H3
|
|
| Chemical Name |
1-[4-[3-[4-(6-fluoro-1,2-benzoxazol-3-yl)piperidin-1-yl]propoxy]-3-methoxyphenyl]ethanone
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.86 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.86 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. View More
Solubility in Formulation 3: 2% DMSO +40% PEG 300 +dd H2O: 2mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3448 mL | 11.7239 mL | 23.4478 mL | |
| 5 mM | 0.4690 mL | 2.3448 mL | 4.6896 mL | |
| 10 mM | 0.2345 mL | 1.1724 mL | 2.3448 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04819776 | Active Recruiting |
Drug: Iloperidone Drug: Iloperidone Placebo |
Bipolar I Disorder | Vanda Pharmaceuticals | March 22, 2021 | Phase 3 |
| NCT05344365 | Recruiting | Drug: Iloperidone | Parkinson Disease Psychosis | Vanda Pharmaceuticals | June 2022 | Phase 2 |
| NCT05648591 | Recruiting | Drug: Iloperidone | Schizophrenia Bipolar I Disorder |
Vanda Pharmaceuticals | May 24, 2023 | Phase 4 |
| NCT02413918 | Completed | Drug: iloperidone | Bipolar Disorder | The University of Texas Health Science Center at San Antonio |
April 2012 | Phase 4 |
| NCT04712734 | Completed | Drug: Iloperidone | Schizophrenia | Vanda Pharmaceuticals | January 13, 2021 | Phase 1 |
 |
|---|