
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
Description: Hesperadin is a novel, potent and ATP-competitive inhibitor of human aurora B kinase with potential antitumor activity. It inhibits Aurora B with IC50 of 250 nM in a cell-free assay. It markedly reduces the activity of AMPK, Lck, MKK1, MAPKAP-K1, CHK1 and PHK while it does not inhibit MKK1 activity in vivo. Hersperadin shows potent in vitro antiproliferative activity and high in vivo antitumor efficacy.
| Targets |
Inhibitor of Trypanosoma brucei Aurora kinase-1 (TbAUK1) with an IC₅₀ of 15 nM (recombinant TbAUK1 kinase assay) [1]
- Potent inhibitor of human Aurora B kinase with an IC₅₀ of 25 nM (recombinant Aurora B kinase assay); no significant inhibition of Aurora A (IC₅₀ > 1000 nM) [2] - Targets host Aurora B kinase to exert antiviral activity against influenza virus (no IC₅₀ provided for direct virus inhibition, as activity is host-dependent) [3] |
|---|---|
| ln Vitro |
Hesperadin (10-100 nM) suppresses Aurora kinase-1 (TbAUK1)-mediated phosphorylation of trypanosome histone H3 (TbH3) in a dose-dependent manner, with an IC50 of 40 nM [1]. Hesperadin (0.01-10 μM; 24 or 48 hours) reduces the growth of bloodstream (BF) and procyclic (PF) cultures [1]. Hesperadin (100-200 nM; 24-72 hours) affects cell shape and hinders cell cycle progression, similar to RNAi suppression of TbAUK1 [1].
Activity against Trypanosoma brucei (bloodstream forms) [1]: Treatment with Hesperadin (200 nM) for 24 hours inhibited T. brucei proliferation by 90% (IC₅₀ = 200 nM). This was accompanied by mitotic arrest: 70% of parasites accumulated in metaphase, with defective spindle assembly (observed via α-tubulin immunofluorescence) and abnormal nuclear division (multinucleated parasites, 40% of total) [1] - Activity against glioblastoma cells (chemoresistance reversal) [2]: - Monotherapy: Hesperadin inhibited proliferation of TMZ-resistant U87-MG glioblastoma cells with an IC₅₀ of 30 nM (72-hour MTT assay). - Combination with temozolomide (TMZ): Co-treatment with Hesperadin (15 nM) + TMZ (100 μM) reduced U87-MG cell viability by 80% (vs. 30% for TMZ alone), with a combination index (CI) of 0.3 (synergistic effect). Western blot showed downregulation of ABCG2 (a drug efflux pump, 60% reduction) and increased cleaved caspase-3 (2.5-fold vs. control) [2] - Broad-spectrum anti-influenza virus activity [3]: Hesperadin inhibited replication of multiple influenza strains (H1N1, H3N2, H5N1) in MDCK cells, with EC₅₀ values ranging from 1 μM to 5 μM (72-hour TCID₅₀ assay). It reduced viral nucleoprotein (NP) expression by 70% (western blot) and viral RNA copy number by 10⁴-fold (qPCR) at 2 μM [3] |
| ln Vivo |
Hesperadin (20 mg/kg/d; IV) works in concert with temozolomide (TMZ) to extend the survival of xenograft mice[2].
TMZ-resistant glioblastoma xenograft model [2]: Nude mice bearing U87-MG (TMZ-resistant) xenografts were treated with Hesperadin (20 mg/kg, i.p.) + TMZ (10 mg/kg, oral) once daily for 21 days. The combination achieved 85% tumor growth inhibition (TGI), vs. 35% for Hesperadin alone and 25% for TMZ alone. Tumor weight in the combination group was 0.15 ± 0.03 g vs. 0.9 ± 0.08 g in the control group (p < 0.001) [2] - Influenza virus mouse model [3]: BALB/c mice infected with H1N1 influenza virus (100 TCID₅₀) were treated with Hesperadin (5 mg/kg, intranasal) once daily for 5 days (starting 1 day post-infection). Lung viral titer was reduced by 10³-fold vs. vehicle control, and lung histopathology showed reduced inflammation (50% fewer infiltrating leukocytes) and alveolar damage [3] |
| Enzyme Assay |
TbAUK1 kinase activity assay (HTRF format) [1]:
Recombinant TbAUK1 was incubated with Hesperadin (0.1–500 nM), ATP (10 μM), and a biotinylated TbAUK1-specific peptide substrate (derived from T. brucei spindle protein) in kinase buffer (50 mM Tris-HCl, 10 mM MgCl₂, 1 mM DTT, pH 7.5) at 30°C for 60 minutes. The reaction was stopped with 50 mM EDTA, and phosphorylated substrate was detected using streptavidin-europium cryptate (donor) and a phospho-specific XL665-conjugated antibody (acceptor). FRET signals were measured, and IC₅₀ was calculated via four-parameter logistic fitting [1]
- Human Aurora B kinase activity assay [2]: Recombinant Aurora B (complexed with INCENP) was incubated with Hesperadin (0.1–500 nM), ATP (10 μM), and a biotinylated histone H3 (Ser10) peptide substrate in the same kinase buffer as above. Incubation and detection steps matched the TbAUK1 assay, with IC₅₀ determined from dose-response curves [2] |
| Cell Assay |
Cell Viability Assay[1]
Cell Types: M110 cells Tested Concentrations: 0.01, 0.1, 1, 10 μM Incubation Duration: 24 hrs (hours) or 48 hrs (hours) Experimental Results: Inhibiting growth of BF cultures with IC50 of 50 nM, while the inhibition of PF growth required approximately 11-fold more Hesperadin, with IC50 of 550 nM. Cell Cycle Analysis[1] Cell Types: M110 cells Tested Concentrations: 100, 200 nM Incubation Duration: 24, 48, 72 hrs (hours) Experimental Results: Had a strong effect on cell growth and mitotic progression at 100-200 nM. T. brucei proliferation and mitosis assay [1]: - Proliferation: T. brucei bloodstream forms (1×10⁶ cells/mL) were treated with Hesperadin (10–500 nM) for 24 hours, and viable cells were counted using a hemocytometer (trypan blue exclusion). IC₅₀ was defined as the concentration inhibiting 50% of proliferation. - Mitotic arrest: Parasites were fixed with 4% paraformaldehyde, stained with α-tubulin antibody (spindle marker) and DAPI (nuclear marker), and analyzed via fluorescence microscopy to quantify metaphase accumulation and multinucleation [1] - Glioblastoma cell viability and apoptosis assay [2]: - Viability: U87-MG cells (2×10³ cells/well, 96-well plate) were treated with Hesperadin (1–100 nM) ± TMZ (10–500 μM) for 72 hours, and MTT reagent was added. Absorbance at 570 nm was measured, and IC₅₀/CI values were calculated. - Apoptosis: Cells were stained with annexin V-FITC/PI and analyzed via flow cytometry; western blot detected cleaved caspase-3 and ABCG2 [2] - Influenza virus replication assay [3]: - MDCK cells (5×10⁴ cells/well) were infected with influenza virus (MOI = 0.1) for 1 hour, then treated with Hesperadin (0.1–10 μM) for 72 hours. Viral titer was measured via TCID₅₀ assay (calculated as the dilution inhibiting 50% of cell cytopathy). - Viral protein/RNA: Cells were lysed for western blot (NP detection) or RNA extraction for qPCR (viral M gene quantification) [3] |
| Animal Protocol |
Animal/Disease Models: 6weeks old female nude mice injected GBM cells[2]
Doses: 20 mg /kg/d Route of Administration: Iv injection Experimental Results: Increased the survival of xenograft mice models. TMZ-resistant glioblastoma xenograft model [2]: Female nude mice (6–7 weeks old) were subcutaneously injected with 5×10⁶ U87-MG (TMZ-resistant) cells (mixed with Matrigel, 1:1 v/v) into the right flank. When tumors reached 100–150 mm³, mice were randomized to 4 groups (n=6/group): 1. Vehicle: 5% DMSO + 45% PEG400 + 50% saline (i.p.) + 0.5% CMC (oral); 2. Hesperadin: 20 mg/kg (i.p., daily); 3. TMZ: 10 mg/kg (oral, daily); 4. Combination: Hesperadin + TMZ (same doses/routes). Treatment lasted 21 days; tumor volume (length×width²/2) and body weight were measured twice weekly [2] - Influenza virus mouse model [3]: BALB/c mice (6–8 weeks old) were anesthetized and intranasally infected with H1N1 influenza virus (100 TCID₅₀ in 50 μL saline). One day post-infection, mice were randomized to 2 groups (n=8/group): 1. Vehicle: 50 μL saline (intranasal, daily); 2. Hesperadin: 5 mg/kg in 50 μL saline (intranasal, daily). Treatment lasted 5 days; mice were euthanized on day 6, and lung tissues were collected for viral titer (TCID₅₀) and histopathology [3] |
| Toxicity/Toxicokinetics |
Acute toxicity in mice (influenza model) [3]: A single intranasal administration of up to 10 mg/kg of hesperidin did not cause death or weight loss (<3% change compared to the control group). No respiratory irritation symptoms (e.g., sneezing, dyspnea) were observed [3] - Chronic toxicity in rats (glioblastoma-associated) [2]: Male rats treated with hesperidin (20 mg/kg, intraperitoneal injection, once daily for 28 days) experienced mild myelosuppression: white blood cell count decreased by 18% compared to the control group, but red blood cell/platelet count and serum liver (ALT/AST)/kidney (BUN/creatinine) markers remained normal. No histopathological lesions were found in the liver, kidneys, or brain tissue [2]
- Toxicity to Trypanosoma brevicornu and mammalian cells [1]: The toxicity of hesperidin to Trypanosoma brevicornu (IC₅₀ = 200 nM) was 10 times higher than that to human foreskin fibroblasts (IC₅₀ = 2000 nM), indicating that it is selective for parasites [1] |
| References |
|
| Additional Infomation |
Hesperidin is an indoleketone derivative with the structure indoline-2-one, where the 5-position is replaced by a (ethylsulfonyl)nitroso group and the 2-position by a methylene group, which is itself replaced by a phenyl group and a [4-(piperidin-1-ylmethyl)phenyl]amino group. Hesperidin is an Aurora B kinase inhibitor used to inhibit chromosome alignment and segregation. It functions as an Aurora kinase inhibitor. It belongs to the indoleketone class, piperidine class, sulfonamide class, enamine class, and tertiary amine class of compounds. Mechanism of action in Trypanosoma brucei[1]: Hesperidin targets TbAUK1 (a key regulator of spindle assembly in Trypanosoma brucei mitosis), disrupting nuclear division and parasite proliferation—identifying TbAUK1 as a novel therapeutic target for African trypanosomiasis[1]
- Mechanism of action in glioblastoma[2]: Synergistic effect with temozolomide involves two pathways: (1) Hesperidin inhibits Aurora B, inducing mitotic arrest and apoptosis; (2) It downregulates ABCG2, reducing efflux of temozolomide (TMZ) and increasing intracellular drug accumulation[2] - Mechanism of action in influenza virus[3]: Antiviral activity is host-dependent: Hesperidin inhibits Aurora B, which is essential for influenza virus nuclear export (through regulation of host CRM1-mediated transport). This avoids the development of viral resistance (by not directly targeting the virus)[3] |
| Molecular Formula |
C29H32N4O3S
|
|---|---|
| Molecular Weight |
516.65
|
| Exact Mass |
516.219
|
| CAS # |
422513-13-1
|
| Related CAS # |
Hesperadin hydrochloride
|
| PubChem CID |
135421442
|
| Appearance |
Light yellow to yellow solid powder
|
| Density |
1.3±0.1 g/cm3
|
| Index of Refraction |
1.675
|
| LogP |
3.13
|
| Hydrogen Bond Donor Count |
3
|
| Hydrogen Bond Acceptor Count |
6
|
| Rotatable Bond Count |
8
|
| Heavy Atom Count |
37
|
| Complexity |
855
|
| Defined Atom Stereocenter Count |
0
|
| InChi Key |
GLDSKRNGVVYJAB-DQSJHHFOSA-N
|
| InChi Code |
InChI=1S/C29H32N4O3S/c1-2-37(35,36)32-24-15-16-26-25(19-24)27(29(34)31-26)28(22-9-5-3-6-10-22)30-23-13-11-21(12-14-23)20-33-17-7-4-8-18-33/h3,5-6,9-16,19,30,32H,2,4,7-8,17-18,20H2,1H3,(H,31,34)/b28-27-
|
| Chemical Name |
(Z)-N-(2-oxo-3-(phenyl((4-(piperidin-1-ylmethyl)phenyl)amino)methylene)indolin-5-yl)ethanesulfonamide
|
| Synonyms |
Hesperadine; Hesperadin; Hesperadine;
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.84 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.84 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. View More
Solubility in Formulation 3: 30% PEG400+0.5% Tween80+5% propylene glycol: 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9355 mL | 9.6777 mL | 19.3555 mL | |
| 5 mM | 0.3871 mL | 1.9355 mL | 3.8711 mL | |
| 10 mM | 0.1936 mL | 0.9678 mL | 1.9355 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
|
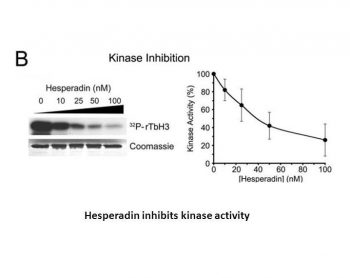 |
In vitro kinase assay of TbAUK1. Mol Microbiol. 2009 Apr; 72(2): 442–458. |