
| Size | Price | Stock | Qty |
|---|---|---|---|
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
Glasdegib (formerly known as PF-04449913; PF4449913; PF04449913; PF-4449913; Daurismo) is a novel, potent, and orally bioavailable small-molecule inhibitor of Smoothened (Smo) in the Hedgehog (Hh) signaling pathway with potential antitumor activity. It is a drug that was authorized in 2018 to treat acute myeloid leukemia that has just been discovered. Smoothened (Smo) is inhibited by Glasdegib at a 5 nM IC50. Cellular development, differentiation, and repair are all significantly impacted by the Hh signaling pathway. Many forms of cancer have been shown to exhibit constitutive activation of the Hh pathway signaling. In a Ptch1+/-p53+/- allograft model, Glasdegib dramatically inhibits the growth of medulloblastoma at doses that also reduce the expression of the murine Shh target gene. When PF-04449913 is used in stromal co-culture experiments, FACS analysis shows a significant decrease in BC LSC relative to normal progenitors.
| Targets |
Smo ( IC50 = 4 nM )
SMO (IC50 = 4 nM for binding to human SMO amino acids 181-787; IC50 = 6.8 nM for inhibition of Shh-stimulated luciferase expression in mouse embryonic fibroblasts). [1] |
|---|---|
| ln Vitro |
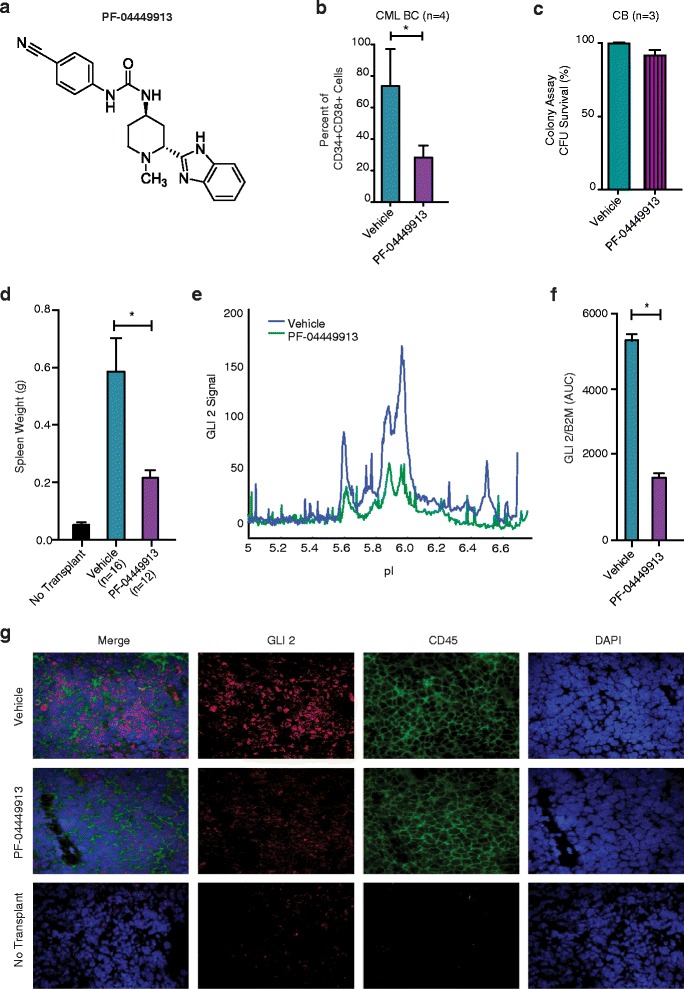
In vitro activity: Glasdegib (PF-04449913) at doses that decrease murine Shh target gene expression significantly reduces the growth of medulloblastoma in a Ptch1+/-p53+/- allograft model and inhibits sonic hedgehog (Shh) stimulated luciferase expression in mouse embryonic fibroblasts with an IC50 of 6.8 nM. When compared to normal progenitors, FACS analysis shows a significant reduction in BC LSC by Glasdegib (PF-04449913) in stromal co-culture experiments. Significantly, the spleen weight of human BC LSC engrafted RAG2-/-p53-/-mice treated daily with Glasdegib (PF-04449913) was significantly lower than that of vehicle-treated controls (p=0.006). This reduction in leukemic burden corresponded with decreased GLI2 protein expression, as determined by both nanoproteomic analysis of FACS purified human progenitors and GLI2 confocal fluorescence microscopic analysis of splenic sections[1]. In SL/M2 stromal co-cultures with human BC CML progenitors, PF-04449913 (1 μM) for 7 days significantly reduced leukemic progenitor survival compared to vehicle (p=0.047, Student's t-test) as shown by FACS analysis (Figure 2b). [1] In similar co-cultures with normal cord blood CD34+ cells, PF-04449913 (1 μM) for 7 days did not reduce colony forming unit (CFU) survival compared to vehicle (Figure 2c), indicating sparing of normal progenitors. [1] In OP9 stromal co-cultures with human cord blood CD34+ cells, PF-04449913 treatment did not affect survival or differentiation of normal hematopoietic stem/progenitor cells (Additional file 1: Figure S2a). [1] |
| ln Vivo |
Glasdegib (PF-04449913)-treated human BC LSC engrafted RAG2-/-p53-/- mice showed a significant reduction in spleen weight (p=0.006) when compared to vehicle-treated controls. Contrary to LSC, normal human HSC cell fate decisions are Hh pathway independent. This is demonstrated by the fact that when CD34+cord blood engrafted NSG mice are treated with Glasdegib (PF-04449913), the frequency of human CD45+cells, progenitors, and both myeloid and lymphoid cell fate commitment remained comparable to vehicle treated controls. These findings underscore the significant context- and cell-specific niche-dependent effects of selective SMO inhibition, which cause GLI2 downregulation[1].
In neonatal RAG2-/-yc-/- mice engrafted with human BC CML LSC, PF-04449913 (100 mg/kg daily by oral gavage for 14 days) significantly reduced spleen weight compared to vehicle (p=0.006) (Figure 2d). [1] Nanoproteomic analysis of FACS-purified human CD34+CD38+ cells from spleens of treated mice showed decreased GLI2 protein expression after PF-04449913 treatment (p=0.001) (Figure 2e-f). [1] Confocal fluorescence microscopy of spleen sections confirmed reduced GLI2 expression in human CD45+ cells after PF-04449913 treatment (Figure 2g). [1] In NSG mice engrafted with normal human cord blood CD34+ cells, PF-04449913 (100 mg/kg daily for 14 days) did not alter engraftment frequency of human CD45+ cells, progenitors, or myeloid/lymphoid differentiation (Additional file 1: Figure S2b-d). [1] Cell cycle FACS analysis of bone marrow from BC CML engrafted mice after PF-04449913 treatment showed a significant reduction in the G0 (dormant) fraction and increase in G1 fraction compared to vehicle (p<0.05) (Figure 4c-d). [1] RNA-seq on FACS-purified human progenitors from treated mice revealed that PF-04449913 significantly downregulated the "Regulation of Cell Cycle" pathway (family-wise p=0.02) (Figure 4e-g). [1] Combination of PF-04449913 (100 mg/kg) plus dasatinib (50 mg/kg) for 14 days significantly reduced myeloid sarcoma count (p<0.01, ANOVA), bone marrow LSC burden (p<0.05), and BCR-ABL1 expression (p<0.05) compared to single agents (Figure 5b-d). [1] Serial transplantation: LSC from mice treated with PF-04449913 plus dasatinib showed significantly reduced capacity to form myeloid sarcomas in secondary recipients (p<0.01, ANOVA) (Figure 5f). [1] Hedgehog pathway gene expression analysis: Combination therapy significantly increased expression of negative Hh regulators (NUMB, PRKACB, FKBP8, CSNK1A1, CSNK1D) (p=0.05, Limma method) (Figure 5e). [1] |
| Enzyme Assay |
Glasdegib, a novel potent small-molecule inhibitor with an IC50 of 5 nM, is an oral bioavailable inhibitor of Smoothened (Smo) in the Hedgehog (Hh) signaling pathway. It was previously known as PF-04449913. In cells, the Hh signaling pathway is essential for growth, differentiation, and repair. In many different kinds of cancer, constitutive activation of the Hh pathway signaling has been reported. In an allograft model with Ptch1+/-p53+/-, Glasdegib dramatically inhibits the growth of medulloblastoma at doses that also reduce the expression of the murine Shh target gene. When compared to normal progenitors, FACS analysis shows a significant reduction in BC LSC by PF-04449913 in stromal co-culture experiments.
PF-04449913 was tested for competition binding to human SMO protein (amino acids 181-787). The IC50 was determined to be 4 nM (Additional file 1: Figure S1a). [1] PF-04449913 was evaluated for inhibition of Sonic hedgehog (Shh)-stimulated luciferase expression in mouse embryonic fibroblasts. The compound inhibited the luciferase reporter activity with an IC50 of 6.8 nM (Additional file 1: Figure S1b, c). [1] |
| Cell Assay |
BMS-354825 (50 nM), Glasdegib (PF-04449913) (1 μM), or a combination of these treatments are used to treat CD34+cells in BC CML or normal cells. The M2-10B4 (M2) and SL/SL (SL) mouse bone marrow stromal cell lines are plated 1:1 mixture at a final concentration of 100,000 cells/mL one day before being co-cultured with 10,000–20,000 CD34+BC CML or normal progenitors. At 14 days, progenitors are scored and FACS sorted into hematopoietic progenitor assays following a week of culture. Normal human hematopoietic stem and progenitor cells are evaluated for survival using the following methods: irradiated (20 Gray) OP9 (M2 clone) stromal cells are co-cultured with 50,000 human CD34+cord blood cells, vehicle, or Glasdegib (PF-04449913) in AlphaMEM with 20% Hyclone FBS, 1% pen strep glutamine, and supplemented with 50 ng/mL SCF, 10 ng/mL thrombopoietin, and 10 ng/mL Flt3. The results are recorded using weekly FACS analysis[1].
Normal or BC CML CD34+ cells were plated on confluent mitomycin-C treated SL/M2 cells (mouse bone marrow stromal cell lines M2-10B4 and SL/SL in 1:1 mixture) with vehicle or PF-04449913 (1 μM) for 7 days. After culture, progenitors were FACS sorted into hematopoietic progenitor assays and colonies scored at 14 days. [1] To assess survival of normal human hematopoietic stem and progenitor cells, irradiated OP9 stromal cells were co-cultured with 50,000 human CD34+ cord blood cells, vehicle or PF-04449913 in AlphaMEM with 20% FBS, 50 ng/ml SCF, 10 ng/ml thrombopoietin, and 10 ng/ml Flt3, and quantified by weekly FACS analysis. [1] For cell cycle analysis, single cell suspensions of bone marrow cells from LSC xenografted mice treated with vehicle or PF-04449913 were immunostained with anti-human CD45, LIVE/DEAD stain, then with FITC-anti-Ki-67 and 7-AAD, followed by FACS analysis. [1] |
| Animal Protocol |
Mice: Equal amounts of BC LSC or normal progenitors are intrahepatically transplanted into RAG2-/-c-/- mice. After the identification of human CD45+cell peripheral blood engraftment, mice are given oral gavage every day for 14 days with either Glasdegib (100 mg/kg), BMS-354825 (50 mg/kg), or a combination of these drugs. The amount of human engraftment in hematopoietic niches is then measured using FACS. Eight weeks after sublethally irradiating seven to ten week old NOD Cg-PrkdcSCID IL2R1Wjl/SzJ mice, 100,000 CD34+human cord blood cells are transplanted retro-orbitally, and the mice are treated with vehicle or Glasdegib (100 mg/kg) for 14 days, followed by FACS engraftment analysis, in order to evaluate the effects on normal HSC function.
Neonatal immunocompromised RAG2-/-yc-/- mice were transplanted intrahepatically with equal numbers of normal progenitors or BC LSC. Upon detection of human CD45+ cell peripheral blood engraftment, mice were treated daily by oral gavage with vehicle (50% 1,2 Propanediol, 50% HBSS or methylcellulose), PF-04449913 (100 mg/kg), dasatinib (50 mg/kg), or the combination for 14 days followed by FACS to quantify human engraftment. [1] For normal HSC function assessment, 7-10 week old NOD.Cg-PrkdcSCID IL2R1Wj/SzJ (NSG) mice were sublethally irradiated, transplanted retro-orbitally with 100,000 CD34+ human cord blood cells and treated 8 weeks later with vehicle or PF-04449913 (100 mg/kg) for 14 days followed by FACS engraftment analysis. [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
The pharmacokinetic characteristics of Glasgow Gibberel are dose-proportional, exhibiting a broad dose-proportional distribution of peak plasma concentrations. In this study, at a 50 mg dose, the median time to reach the maximum concentration of 321 ng/ml was 4 hours, with an AUC of 9587 ng·h/ml. The oral bioavailability of Glasgow Gibberel is reported to be 55%. In a multiple-dose study of 50 mg, the Cmax, tmax, and AUC were 542 ng/ml, 4 hours, and 9310 ng·h/ml, respectively. In the same study, the steady-state mean concentration was 388 ng/ml. The absorption rate of Glasgow Gibberel can be altered by concomitant intake of high-fat, high-calorie foods. Following a single oral dose of 100 mg of radiolabeled Glasgow Gibberel, 49% was excreted in the urine, of which 17% was excreted unchanged; 42% was excreted in the feces, of which 20% was excreted unchanged. The volume of distribution (VZ) of 50 mg glasdegibr is reported to be 225 L. The geometric mean apparent volume of distribution (Vz/F) in patients with hematologic malignancies is 188 L (20%). The clearance of 50 mg glasdegibr is reported to be 5.22 L/h. In patients with hematologic malignancies, the geometric mean apparent clearance (%CV) after a once-daily dose of 100 mg glasdegibr is 6.45 L/h (25%). Metabolism/MetabolitesAfter oral administration, glasdegibr is primarily metabolized by CYP3A4, with smaller contributions from CYP2C8 and UGT1A9. Only 69% of the administered dose of glasdegibr remains unmetabolized in plasma. Biological Half-LifeThe half-life of glasdegibr is reported to be 17.4 hours. |
| Toxicity/Toxicokinetics |
Hepatotoxicity
Elevated serum ALT levels are common during treatment with gradigib, occurring in 31% of patients, with 11% of these patients having ALT levels exceeding 5 times the upper limit of normal. Clinical use of gradigib is limited, and no cases of acute liver injury with symptomatic or jaundice have been identified. Due to limited clinical experience with Hedgehog inhibitors, the likelihood of liver injury is unclear. Probability score: E (Unproven, but suspected cause of clinically significant liver injury). Pregnancy and Lactation Effects ◉ Overview of Use During Lactation There is currently no information regarding the use of gradigib during lactation. The drug binds to plasma proteins at a rate of 91%, therefore its concentration in breast milk is likely to be low. The half-life of gradigib is 17.4 hours, and it may be cleared from breast milk within 4 to 7 days after the last dose. However, the manufacturer recommends discontinuing breastfeeding during treatment with gradigib and for at least 30 days after the last dose. ◉ Effects on breastfed infants No published information found as of the revision date. ◉ Effects on lactation and breast milk No published information found as of the revision date. Protein binding Gladgib reportedly has a protein binding rate of 91%, which is related to its high lipophilicity. No significant toxicity was observed in normal human hematopoietic stem and progenitor cells treated with PF-04449913 in vitro and in vivo, as indicated by unchanged differentiation capacity, CFU survival, and engraftment (Figure 2c; Additional file 1: Figure S2). [1] |
| References | |
| Additional Infomation |
Pharmacodynamics
In preclinical studies, glasdegib significantly reduced the leukemia stem cell burden in xenograft models and decreased the number of cells expressing leukemia stem cell markers. In clinical trials, glasdegib significantly downregulated the expression of glioma-associated transcription factor GL11 in the skin, with a downregulation exceeding 80%. In the same study, 8% of acute myeloid leukemia patients achieved morphological complete remission, and 31% had stable disease. Recent clinical trials have confirmed that glasdegib can improve overall survival to 8.3 months, almost twice that of patients treated with low-dose cytarabine. Furthermore, there are reports of dose-dependent QTc interval prolongation in patients treated with glasdegib. PF-04449913 is a selective smoothened (SMO) antagonist that reduces GLI expression. It has been shown to inhibit Hedgehog signaling and reduce leukemia stem cell maintenance in chronic myeloid leukemia (CML), particularly in blast crisis. The compound induces cell cycle activation in dormant LSC, sensitizing them to tyrosine kinase inhibitors such as dasatinib. Based on this study, Phase 1b/2 clinical trials combining SMO inhibitor and TKI for advanced myeloid malignancies are ongoing. [1] |
| Molecular Formula |
C21H22N6O
|
|
|---|---|---|
| Molecular Weight |
374.44
|
|
| Exact Mass |
374.185
|
|
| Elemental Analysis |
C, 67.36; H, 5.92; N, 22.44; O, 4.27
|
|
| CAS # |
1095173-27-5
|
|
| Related CAS # |
1095173-27-5; 2030410-25-2; 1095173-64-0 (HCl); 1352568-48-9 (2HCl)
|
|
| PubChem CID |
25166913
|
|
| Appearance |
White solid powder
|
|
| Density |
1.3±0.1 g/cm3
|
|
| Boiling Point |
633.4±55.0 °C at 760 mmHg
|
|
| Flash Point |
336.9±31.5 °C
|
|
| Vapour Pressure |
0.0±1.9 mmHg at 25°C
|
|
| Index of Refraction |
1.686
|
|
| LogP |
2.77
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
3
|
|
| Heavy Atom Count |
28
|
|
| Complexity |
595
|
|
| Defined Atom Stereocenter Count |
2
|
|
| SMILES |
O=C(NC1=CC=C(C#N)C=C1)N[C@H]2C[C@H](C3=NC4=CC=CC=C4N3)N(C)CC2
|
|
| InChi Key |
SFNSLLSYNZWZQG-VQIMIIECSA-N
|
|
| InChi Code |
InChI=1S/C21H22N6O/c1-27-11-10-16(24-21(28)23-15-8-6-14(13-22)7-9-15)12-19(27)20-25-17-4-2-3-5-18(17)26-20/h2-9,16,19H,10-12H2,1H3,(H,25,26)(H2,23,24,28)/t16-,19-/m1/s1
|
|
| Chemical Name |
1-[(2R,4R)-2-(1H-benzimidazol-2-yl)-1-methylpiperidin-4-yl]-3-(4-cyanophenyl)urea
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (5.55 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.55 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (5.55 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6707 mL | 13.3533 mL | 26.7065 mL | |
| 5 mM | 0.5341 mL | 2.6707 mL | 5.3413 mL | |
| 10 mM | 0.2671 mL | 1.3353 mL | 2.6707 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Glasdegib (PF-04449913) With Temozolomide Newly Diagnosed Glioblastoma
CTID: NCT03466450
Phase: Phase 1/Phase 2 Status: Completed
Date: 2024
A phase Ib/IIa pilot trial of the oral Hedgehog Signalling Inhibitor, Glasdegib, in Patients with sclerotic chronic graft-versus-host disease refractory to second-line treatment
CTID: null
Phase: Phase 1, Phase 2 Status: Completed
Date: 2017-09-28
 Selective shh inhibition reduces lsc burden in stromal co-cultures.J Transl Med.2015 Mar 21;13:98. |
|---|
Shh inhibition induces cycling of dormant leukemic progenitors.J Transl Med.2015 Mar 21;13:98. |
PF-04449913 induced cell cycle activation enhances TKI sensitivity.J Transl Med.2015 Mar 21;13:98. |
GLI2 induces cell cycle arrest in leukemic progenitors.J Transl Med.2015 Mar 21;13:98. |
|---|
SHH pathway deregulation in chronic myeloid leukemia progression.J Transl Med.2015 Mar 21;13:98. |