
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
Gefapixant (also known as AF-219 and MK-7264) is a novel, potent, and orally bioactive P2X3 receptor (P2X3R) antagonist with antitussive effects, and may be used for chronic cough. It inhibits P2X3R with IC50s of ~30 nM versus recombinant hP2X3 homotrimers and 100-250 nM at hP2X2/3 heterotrimeric receptors. Treating pain by inhibiting ATP activation of P2X3-containing receptors heralds an exciting new approach to pain management, and Afferent's program marks the vanguard in a new class of drugs poised to explore this approach to meet the significant unmet needs in pain management. P2X3 receptor subunits are expressed predominately and selectively in so-called C- and Aδ-fiber primary afferent neurons in most tissues and organ systems, including skin, joints, and hollow organs, suggesting a high degree of specificity to the pain sensing system in the human body. P2X3 antagonists block the activation of these fibers by ATP and stand to offer an alternative approach to the management of pain and discomfort.
| Targets |
IC50: ~30 nM (recombinant hP2X3 homotrimers), 100-250 nM (hP2X2/3 heterotrimeric receptors)[1]. |
|---|---|
| ln Vitro |
For recombinant homotrimeric hP2X1, hP2X2, hP2X4, rP2X5, and hP2X7 channels, the IC50 values are greater than 10,000 nM, indicating that gefapixant has no inhibitory impact on any receptor that contains non-P2X3 subunits [1].
Potent and selective antagonist of P2X3 and P2X2/3 receptors: Gefapixant (AF219; MK-7264) competitively inhibited ATP-induced calcium influx in HEK293 cells expressing human P2X3 or P2X2/3 receptors, with IC50 values of 6.2 nM and 5.1 nM, respectively [1, 2] - High subtype selectivity: No significant inhibition of ATP-mediated responses in cells expressing P2X1, P2X4-P2X7 receptors at concentrations up to 10 μM, >160-fold selectivity for P2X3/P2X2/3 over other P2X subtypes [1, 2] - Inhibited sensory neuron activation: 10 nM Gefapixant (AF219; MK-7264) reduced ATP-induced action potential firing by ~75% in primary cultures of rat vagal sensory neurons [1] - No cytotoxicity to HEK293 cells or primary sensory neurons at concentrations up to 50 μM (cell viability > 90%) [1] |
| ln Vivo |
Within 14 days of intra-articular monoiodoacetate injection, gefapixant (7 days bid, oral) totally reversed overt hyperalgesia and decreased weight-bearing laterality at two higher doses in a rat model of knee osteoarthritis [2].
Antitussive activity in guinea pig citric acid-induced cough model: Oral administration of Gefapixant (AF219; MK-7264) (3, 10, 30 mg/kg) dose-dependently reduced cough frequency by ~35%, ~60%, and ~75%, respectively, compared to vehicle control [1, 2] - Suppressed cough reflex in rat capsaicin-induced cough model: 10 mg/kg oral dose reduced cough episodes by ~65% and cough duration by ~55% [2] - Attenuated afferent sensitization in rat bladder hyperalgesia model: 30 mg/kg oral Gefapixant (AF219; MK-7264) reduced bladder hyperresponsiveness to distension by ~50%, inhibiting P2X3-mediated sensory signaling [1] |
| Enzyme Assay |
The aryloxy-pyrimidinediamine, Gefapixant/AF-219 (Ford et al., 2013; Smith et al., 2013) is an orally active small molecule (Mol Wt. ∼350 Daltons) antagonist at human P2X3-containing receptors. The inhibitory potency (IC50) of AF-219 has been reported as ∼30 nM versus recombinant hP2X3 homotrimers and 100–250 nM at hP2X2/3 heterotrimeric receptors, potencies very similar to those reported for recombinant rat receptors, and it displays no inhibitory impact on any non-P2X3 subunit containing receptors (IC50 values ≫ 10,000 nM at recombinant homotrimeric hP2X1, hP2X2, hP2X4, rP2X5 and hP2X7 channels). Reports from other related chemical members of this P2X3 selective pyrimidinediamine class have shown that the mechanism of inhibition is non-competitive (allosteric) and have been mixed regarding species-independency of P2X3 receptor potency estimates: AF-353 (Gever et al., 2010) shows remarkable potency congruency between human and rat recombinant P2X3 homotrimers (IC50 values of 8.7 and 8.9 nM, respectively) whereas the more potent analog AF-792 (also referred to as RO-51; developed initially as a potential prodrug for AF-353) was shown to be less potent at human versus rat P2X3 receptors in one report (Serrano et al., 2012) and yet species-independent in another (Jahangir et al., 2009). It is important to note that some selectivity for P2X3 versus P2X2/3 channels has been a common claim across several chemical classes of inhibitors (see Gum et al., 2012: e.g., AF-219 analogs, nucleotides such as TNP-ATP, benzenetricarboxylic acids such as A-317491), although in most studies values reported are not affinity determinations but IC50 estimates. Under such circumstances true selectivity cannot be categorically inferred, especially for the competitive antagonists (such as TNP-ATP and A-317491) as the IC50 is a parameter that will change with agonist concentration used and depends on agonist potency at the different trimers.[1]
P2X3 radioligand binding assay: Recombinant human P2X3 receptors immobilized on microtiter plates were incubated with [3H]-labeled ATP analog and serial dilutions of Gefapixant (AF219; MK-7264) (0.01-1000 nM) in binding buffer. After incubation at 25°C for 90 minutes, unbound ligand was removed by washing. Bound radioactivity was measured, and Ki values were calculated via competition binding analysis [1, 2] - P2X3 functional assay (calcium influx): HEK293 cells expressing human P2X3 receptors were loaded with a fluorescent calcium indicator and pre-treated with Gefapixant (AF219; MK-7264) (0.01-1000 nM) for 30 minutes. ATP (10 μM) was added to induce calcium influx, and fluorescence intensity was measured. IC50 values were determined based on inhibition of calcium signaling [1, 2] |
| Cell Assay |
Primary sensory neuron activation assay: Rat vagal sensory neurons were isolated and cultured for 7 days. Neurons were pre-treated with Gefapixant (AF219; MK-7264) (0.1-100 nM) for 1 hour, then stimulated with ATP (10 μM). Action potential firing was recorded using patch-clamp electrophysiology, and inhibition rates were quantified [1]
- P2X subtype selectivity assay: HEK293 cells transfected with human P2X1, P2X4-P2X7 receptors were loaded with calcium indicator. Cells were pre-treated with Gefapixant (AF219; MK-7264) (1 μM) for 30 minutes, then stimulated with subtype-specific ATP concentrations. Calcium influx was measured to assess selectivity [1, 2] |
| Animal Protocol |
Guinea pig citric acid-induced cough model: Male Dunkin-Hartley guinea pigs (300-350 g) were randomly divided into vehicle and treatment groups. Gefapixant (AF219; MK-7264) was suspended in 0.5% carboxymethylcellulose sodium and administered orally at 3, 10, or 30 mg/kg. Thirty minutes later, guinea pigs were exposed to citric acid aerosol (10% w/v) for 10 minutes, and coughs were counted visually and acoustically [1, 2]
- Rat bladder hyperalgesia model: Female Sprague-Dawley rats (200-250 g) were induced with bladder inflammation via intravesical administration of cyclophosphamide. Gefapixant (AF219; MK-7264) was administered orally at 10 or 30 mg/kg 1 hour before bladder distension. Bladder pressure and voiding frequency were recorded to assess hyperresponsiveness [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
The absolute bioavailability of gefaspirant has not been assessed, but is estimated to be ≥78%. The recommended dose is 45 mg twice daily. Steady state is reached within 2 days, with steady-state mean plasma AUC and Cmax of 4,144 ng∙hr/mL and 531 ng/mL, respectively. The time to peak plasma concentration (Tmax) after oral administration is 1 to 4 hours. Concomitant administration of gefaspirant with high-fat, high-calorie foods has no effect on its AUC or Cmax. Gefaspirant is primarily excreted via the kidneys. In a healthy male subject, approximately 76.4% of the administered radiolabeled drug was recovered in the urine, and 22.6% in the feces. Of the drug recovered in the feces, 64% was unchanged drug, and of the drug recovered in the urine, 20% was unchanged drug. Based on population pharmacokinetic analysis, the estimated steady-state apparent volume of distribution after oral administration of 45 mg gefapixant twice daily is 133.8 L (Vc 101 L, Vp 32.8 L). Integrated population pharmacokinetic analysis of Phase I, II, and III data showed a geometric mean apparent clearance (Cl/F) of 10.8 L/h. In clinical pharmacology studies, the observed clearance was 14.8 L/h, with renal clearance of approximately 8.7 L/h. Metabolisms/Metabolites Gefapixant is relatively poorly metabolized. Following oral administration, only 14% of the administered dose is recovered as metabolites in urine and feces. The majority drug component in plasma is the unchanged parent drug (87%), with circulating metabolites each accounting for less than 10%. The major biotransformation pathways observed in the ADME study of gefapixant included hydroxylation, O-demethylation, dehydrogenation, oxidation, and direct glucuronidation. Secondary biotransformation pathways include glucuronidation of the O-demethylated metabolite, and the formation of O-demethylated and hydrogenated metabolites. The three most abundant circulating metabolites are M1 (O-demethylated glucuronide of gefapitant), M5 (direct glucuronidation of the parent drug), and M13 (hydroxylated metabolite), accounting for 1.0%, 6.3%, and 5.8% of the total drug-related components in plasma, respectively. Biological Half-Life The terminal half-life of gefapitant is 6–10 hours. |
| Toxicity/Toxicokinetics |
Protein Binding
Gefapistane exhibits a relatively low protein binding rate (55%) in vitro, therefore drug interactions due to protein displacement are not expected. Acute Toxicity: LD50 > 200 mg/kg (oral in rats and guinea pigs); no death or acute organ damage was observed at doses up to 200 mg/kg [1, 2] Subchronic Toxicity: No significant changes in body weight, liver and kidney function (ALT, AST, creatinine), or hematological parameters were observed in rats after oral administration of 30 mg/kg daily for 28 days [1] Clinical Tolerability: Mild to moderate adverse events reported in human studies included taste disturbance (25%), dry mouth (18%), and headache (10%), which were dose-related and reversible [1, 2] Plasma Protein Binding: Approximately 92% (humans); approximately 89% (rats) [1] |
| References |
[1]. Anthony P. Ford, et al. The therapeutic promise of ATP antagonism at P2X3 receptors in respiratory and urological disorders. Front Cell Neurosci. 2013; 7: 267.
[2]. Ford AP, In pursuit of P2X3 antagonists: novel therapeutics for chronic pain and afferent sensitization. Purinergic Signal. 2012 Feb;8(Suppl 1):3-26. [3]. Martin Nguyen A, et al. Validation of a visual analog scale for assessing cough severity in patients with chronic cough. Ther Adv Respir Dis. 2021 Jan-Dec;15:17534666211049743 |
| Additional Infomation |
It is estimated that 5% to 10% of adults worldwide suffer from chronic cough, defined as a cough lasting more than eight weeks. Some patients experience persistent symptoms despite thorough examination and treatment; if the cough is unresponsive to conventional treatments, it is called refractory chronic cough (RCC); if the definite cause of the cough cannot be determined, it is called unexplained chronic cough (UCC). Existing treatments for chronic cough have significant side effects, especially opioids such as codeine or dextromethorphan. Gefapixant is a novel P2X3 receptor antagonist that reduces the cough reflex in patients with chronic cough. It was approved in Japan and Switzerland in 2022 for the treatment of adults with RCC and UCC, and in the European Union in September 2023 for the same indication. It is the first therapy approved in the EU for the treatment of renal cell carcinoma (RCC) or ulcerative colon cancer (UCC).
Drug Indications Gefapixant is indicated for the treatment of refractory or unexplained chronic cough in adults. Lyfnua is indicated for the treatment of refractory or unexplained chronic cough in adults. Mechanism of Action Gefapixant is a selective P2X3 receptor antagonist with some activity against P2X2/3 receptor subtypes. P2X3 receptors are ATP-gated ion channels located on the sensory C fibers of the vagus nerve in the airway. Under inflammatory conditions, ATP is released from airway mucosal cells and subsequently binds to P2X3 receptors on C fibers. Activation of vagal C fibers generates a cough impulse and initiates the cough reflex. Gefapixant inhibits the binding of ATP to P2X3 receptors, thereby reducing the excessive activation of C fibers by extracellular ATP and inhibiting the subsequent cough reflex. Pharmacodynamics Gefapixant exerts its therapeutic effect by inhibiting the cough reflex induced by vagal sensory C fibers. In clinical studies, patients showed a significant reduction in 24-hour cough frequency compared to the placebo group—this reduction was observed as early as week 4 and persisted until the end of the primary efficacy assessment period. Since renal excretion is the primary clearance route for gefapitant, patients with severe renal impairment (eGFR < 30 mL/min/1.73m²) may require dose adjustment to maintain adequate systemic exposure. Gefapitant (AF219; MK-7264) is a first-in-class selective small molecule P2X3 and P2X2/3 receptor antagonist [1, 2] - Core mechanism of action: Competes with ATP to bind to P2X3/P2X2/3 receptors on sensory neurons, blocking ion channel activation and inhibiting the transmission of nociceptive, cough, and bladder sensory signals [1, 2] - Potential therapeutic applications: Chronic cough (primary indication), overactive bladder, and chronic pain associated with afferent sensory sensitization [1, 2] - It has good oral absorption properties. Bioavailability and subtype selectivity, minimizing off-target effects on other P2X receptors [1, 2] - represent a novel therapy for diseases caused by ATP-mediated sensory neuron activation, addressing an unmet need in respiratory and urological medicine [1] |
| Molecular Formula |
C14H19N5O4S
|
|---|---|
| Molecular Weight |
353.39676117897
|
| Exact Mass |
353.115
|
| Elemental Analysis |
C, 47.58; H, 5.42; N, 19.82; O, 18.11; S, 9.07
|
| CAS # |
1015787-98-0
|
| Related CAS # |
Gefapixant citrate;2310299-91-1
|
| PubChem CID |
24764487
|
| Appearance |
Typically exists as White to off-white solid at room temperature
|
| Density |
1.4±0.1 g/cm3
|
| Boiling Point |
606.3±65.0 °C at 760 mmHg
|
| Flash Point |
320.5±34.3 °C
|
| Vapour Pressure |
0.0±1.7 mmHg at 25°C
|
| Index of Refraction |
1.615
|
| LogP |
0.73
|
| Hydrogen Bond Donor Count |
3
|
| Hydrogen Bond Acceptor Count |
9
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
24
|
| Complexity |
512
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
S(C1=CC(=C(C=C1OC)C(C)C)OC1=CN=C(N)N=C1N)(N)(=O)=O
|
| InChi Key |
HLWURFKMDLAKOD-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C14H19N5O4S/c1-7(2)8-4-10(22-3)12(24(17,20)21)5-9(8)23-11-6-18-14(16)19-13(11)15/h4-7H,1-3H3,(H2,17,20,21)(H4,15,16,18,19)
|
| Chemical Name |
5-(2,4-diaminopyrimidin-5-yl)oxy-2-methoxy-4-propan-2-ylbenzenesulfonamide
|
| Synonyms |
AF-219; Gefapixant [USAN]; Benzenesulfonamide, 5-[(2,4-diamino-5-pyrimidinyl)oxy]-2-methoxy-4-(1-methylethyl)-; RO4926219; RG-1646; Ro-4926219;
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO : ~5 mg/mL (~14.15 mM)
|
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.07 mM) (saturation unknown) in 10% DMSO + 90% PBS (add these co-solvents sequentially from left to right, and one by one), clear solution.
Solubility in Formulation 2: ≥ 0.5 mg/mL (1.41 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 0.5 mg/mL (1.41 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: ≥ 0.5 mg/mL (1.41 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8297 mL | 14.1483 mL | 28.2965 mL | |
| 5 mM | 0.5659 mL | 2.8297 mL | 5.6593 mL | |
| 10 mM | 0.2830 mL | 1.4148 mL | 2.8297 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
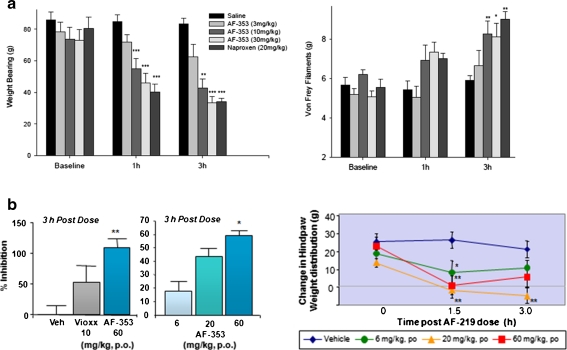 Nocifensive data showing the effect of a P2X3 antagonist in preclinical models of joint hyperalgesia.Purinergic Signal.2012 Feb;8(Suppl 1):3-26. |
|---|
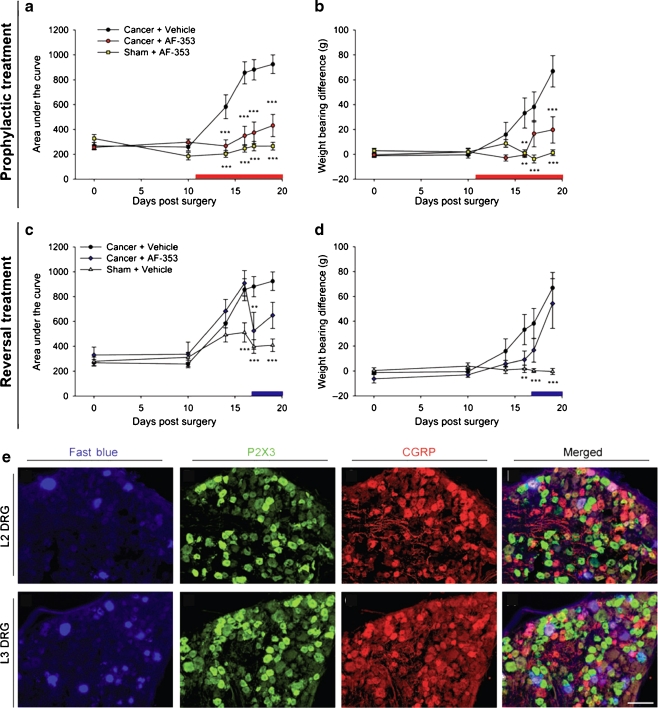 Systemic P2X3 receptor antagonism with AF-353 attenuates bone cancer pain behaviour in rats.Purinergic Signal.2012 Feb;8(Suppl 1):3-26. |
Effects of P2X3 antagonists in preclinical models of urinary bladder reflexes.Purinergic Signal.2012 Feb;8(Suppl 1):3-26. |