
| Size | Price | Stock | Qty |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g | |||
| 5g | |||
| Other Sizes |
Purity: ≥98%
Esmolol HCl (Brevibloc; ASL-8052; ASL 8052), the hydrochloride salt of esmolol, is a potent and cardioselective beta-blocker used to control rapid heartbeats or abnormal heart rhythms. Esmolol is an enantiomeric pair with one asymmetric center; the (+)-enantiomer is inactive and the (-)-enantiomer is active. This is similar to other P-blockers that have an oxypropranolamine nucleus. Esmolol exhibits a swift onset and offset of action, with its primary site of action being on the conduction systems of the sinus node and atrioventricular (AV) node.
| Targets |
Adrenergic receptor
β1-adrenergic receptor (Ki = 11 nM) [4] - β2-adrenergic receptor (Ki = 3400 nM, 309-fold lower affinity than β1 subtype) [4] |
|---|---|
| ln Vitro |
In vitro activity: Esmolol is an enantiomeric pair with one asymmetric center; the (+)-enantiomer is inactive and the (-)-enantiomer is active. This is similar to other P-blockers that have an oxypropranolamine nucleus. Esmolol exhibits a swift onset and offset of action, with its primary site of action being on the conduction systems of the sinus node and atrioventricular (AV) node. [1]
Incubation of human recombinant β1-adrenergic receptor-expressing cells with Esmolol HCl (0.1-1000 nM) dose-dependently inhibited isoproterenol-induced cAMP accumulation, with IC50 of 36 nM; inhibition of β2-mediated cAMP accumulation required 100-fold higher concentration (IC50 = 3600 nM) [4] - Esmolol HCl (10 μM) reduced lipopolysaccharide (LPS)-induced TNF-α and IL-6 release from human peripheral blood monocytes by 28% and 24% respectively, exerting mild anti-inflammatory effects [2] - In isolated guinea pig ventricular myocytes, Esmolol HCl (50 μM) prolonged action potential duration by 15% and reduced peak calcium current (ICa,L) by 22%, without affecting resting membrane potential [4] |
| ln Vivo |
Esmolol (20 mg/kg) infusion enhances myocardial oxygen utilization and maintains myocardial function in septic rats. In rabbits, Esmolol (5 mg/kg, intravenously) causes a dose-dependent reduction in heart rate (HR), with a maximum percent reduction of 13%. In rabbits, Esmolol (5 mg/kg, intravenously) lowers mean arterial pressure (MAP) in a dose-dependent manner, with a maximum percent reduction of 38.2%. [3] In dogs, Esmolol (300 mg/kg) suppresses AV nodal conduction, increases the effective refractory period of the right ventricle and the preload of the left ventricle, and significantly lowers heart rate, rate-pressure product, left ventricular contraction, cardiac output, and relative refractory period of the right ventricle.
Intravenous infusion of Esmolol HCl (50 μg/kg/min) to patients with severe sepsis reduced heart rate by 18 bpm and mean arterial pressure by 10 mmHg within 30 minutes, with no significant effect on cardiac index [2] - During general anesthesia, Esmolol HCl (loading dose 1 mg/kg, maintenance 200 μg/kg/min) reduced tracheal intubation-induced tachycardia (heart rate increase from 85 ± 10 to 98 ± 12 bpm vs. 115 ± 15 bpm in control group) [3] - In rats with myocardial ischemia-reperfusion injury, intravenous Esmolol HCl (10 mg/kg) administered 10 minutes before reperfusion reduced infarct size by 25% and decreased myocardial apoptotic index by 30% [4] - Oral administration of Esmolol HCl (20 mg/kg/day) to spontaneously hypertensive rats (SHR) for 7 days reduced resting heart rate by 22 bpm, with minimal effect on systolic blood pressure [4] |
| Enzyme Assay |
β1/β2-adrenergic receptor binding assay: Membrane fractions from HEK293 cells expressing human β1 or β2 receptors were prepared. Esmolol HCl (0.01-10000 nM) was incubated with membranes and [³H]dihydroalprenolol (non-selective β ligand) at 37°C for 45 minutes. Unbound ligand was removed by vacuum filtration, and bound radioactivity was quantified by liquid scintillation counting. Ki values were calculated using competitive binding curves [4]
- cAMP inhibition assay: β1 receptor-expressing cells were seeded in 24-well plates and cultured for 24 hours. Cells were pretreated with Esmolol HCl (0.01-10000 nM) for 30 minutes, then stimulated with isoproterenol (1 μM) for 15 minutes. Cells were lysed, and cAMP levels were measured by competitive ELISA. IC50 values were derived from dose-response inhibition curves [4] |
| Cell Assay |
Guinea pig ventricular myocyte electrophysiology assay: Ventricular myocytes were isolated by enzymatic dispersion and superfused with Tyrode's solution. Esmolol HCl (10-100 μM) was added to the superfusate, and action potentials and calcium currents were recorded using whole-cell patch-clamp technique [4]
- Monocyte anti-inflammatory assay: Human peripheral blood monocytes were isolated and plated in 96-well plates. After 24 hours of culture, cells were pretreated with Esmolol HCl (1-50 μM) for 1 hour, then exposed to LPS (1 μg/mL) for 6 hours. TNF-α and IL-6 concentrations in culture supernatants were quantified by ELISA [2] |
| Animal Protocol |
5 mg/kg, i.v.
Rabbits Severe sepsis patient model: Adult patients (n=40) with severe sepsis were randomized to receive Esmolol HCl intravenous infusion (50 μg/kg/min) or placebo for 24 hours. Heart rate, mean arterial pressure, cardiac index, and serum cytokine levels (TNF-α, IL-6) were monitored hourly [2] - Anesthesia-induced tachycardia model: Patients (n=30) undergoing elective surgery were given Esmolol HCl (1 mg/kg loading dose over 1 minute, followed by 200 μg/kg/min maintenance infusion) 2 minutes before tracheal intubation. Heart rate and blood pressure were recorded at baseline, intubation, and 5 minutes post-intubation [3] - Myocardial ischemia-reperfusion model: Male Sprague-Dawley rats (10 weeks old) were subjected to 30 minutes of coronary artery occlusion followed by 2 hours of reperfusion. Esmolol HCl (10 mg/kg) was administered intravenously 10 minutes before reperfusion. Infarct size was measured by TTC staining, and myocardial apoptosis by TUNEL assay [4] - SHR hypertension model: Male spontaneously hypertensive rats (12 weeks old) received Esmolol HCl (20 mg/kg/day) via oral gavage for 7 days. Resting heart rate and systolic blood pressure were measured daily using a tail-cuff system [4] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
It is currently unclear whether esmolol and/or ASL 8123 can cross the placental barrier, but it has been confirmed that the drug can. In animal studies, the concentration of esmolol in the fetal artery after infusion was approximately 10% of the maternal concentration. It is also unclear whether esmolol and/or ASL 8123 is excreted into breast milk. Following intravenous administration, esmolol is rapidly and widely distributed. In healthy adults, the apparent volume of distribution of esmolol and its deesterified metabolite (ASL 8123) after intravenous administration is approximately 3.4 and 0.41 L/kg, respectively. In healthy adults, the volume of distribution of esmolol in the central compartment and at steady state after intravenous administration is approximately 0.87 and 1.2 L/kg, respectively. The apparent volume of distribution appears to be reduced in patients undergoing coronary artery bypass grafting, while it is increased in patients with renal insufficiency undergoing peritoneal dialysis and in patients with cirrhosis. Following intravenous injection in rats, esmolol is distributed in the liver and kidneys, but only in small amounts in the cerebrospinal fluid, spleen, or testes. In vitro studies show that esmolol binds to approximately 55% of plasma proteins, primarily albumin and α1-acid glycoprotein. Protein binding to α1-acid glycoprotein appears to be concentration-independent within the esmolol concentration range of 3–110 μg/ml. In vitro studies show that ASL 8123 binds to approximately 10% of plasma proteins. For more complete data on the absorption, distribution, and excretion of esmolol hydrochloride (out of 10 items), please visit the HSDB record page. Metabolism/Metabolites Esmolol is rapidly and extensively metabolized primarily in the erythrocyte cytosol by esterases (likely aryl esterases). The drug may also be metabolized in hyperperfused tissues containing esterases, such as the liver and kidneys. The methyl ester portion hydrolyzes to produce the deesterified (free acid) metabolite 4-(2-hydroxy-3-((1-methylethyl)amino)propoxy)phenylpropionic acid (ASL 8123) and methanol. Esmolol appears to be poorly hydrolyzed by serum cholinesterase (pseudocholinesterase), acetylcholinesterase, or carbonic anhydrase. It is estimated that approximately 83% of esmolol doses are metabolized to ASL 8123. ASL 8123 has a low affinity for β-adrenergic receptors and exhibits only very low β-receptor blocking activity in animals (approximately 1000 to 1500 times less potent than esmolol), with no significant blocking effect in humans. Unlike esmolol, ASL 8123 is primarily excreted by the kidneys, and the elimination half-life of its metabolites can be prolonged by up to 10 times in patients with renal insufficiency; however, due to the extremely low β-receptor blocking activity of ASL 8123, this accumulation is considered clinically insignificant. The amount of methanol produced during drug hydrolysis appears to be clinically insignificant. Intravenous infusion of esmolol hydrochloride at doses of 300 μg/kg/min over 16 hours and 150 μg/kg/min over 24 hours resulted in blood methanol concentrations of 2.8–5.9 μg/ml and 2.9–13.2 μg/ml, respectively, both below 2% of concentrations typically associated with methanol poisoning. Biological Half-Life: Following intravenous infusion in adults, the average half-life of esmolol is approximately 2 minutes in the initial distribution phase and approximately 9 minutes in the terminal elimination phase (range: 5–23 minutes), but significant inter-individual differences exist in the plasma elimination half-life. Free Acid Metabolites: Approximately 3.7 hours (can increase up to 10-fold in renal failure). Due to its low oral bioavailability (<5%), esmolol hydrochloride can only be administered via intravenous infusion. [1] - In healthy volunteers, after intravenous infusion of esmolol hydrochloride (1 mg/kg), the peak plasma concentration (Cmax) at the end of the infusion reached 15 μg/mL, and the elimination half-life (t1/2) was 9.2 minutes. [1] - The drug is rapidly metabolized by erythrocyte esterases to the inactive metabolite ASL-8123, with a systemic clearance of 285 mL/kg/min and a volume of distribution (Vd) of 3.4 L/kg. [1] - Approximately 70% of the metabolized dose is excreted in the urine within 24 hours, and less than 5% of the drug is excreted unchanged. [1] |
| Toxicity/Toxicokinetics |
Interactions
When esmolol is taken concomitantly with catecholamine-depleting drugs (such as reserpine), the effects of the two drugs may have an additive effect. In healthy adults, concomitant administration of esmolol and morphine can increase steady-state esmolol concentrations by approximately 50%, but the pharmacokinetics of morphine are not affected. Esmolol may prolong the action of succinylcholine, but does not affect the onset time of neuromuscular blockade. In some patients, concomitant administration of esmolol can prolong succinylcholine-induced neuromuscular blockade by approximately 60%. Furthermore, in other patients receiving combination therapy, the duration of neuromuscular blockade was not prolonged. During concomitant warfarin treatment, blood esmolol concentrations may be slightly elevated… For more complete data on interactions of esmolol hydrochloride (11 types), please visit the HSDB record page. Non-human toxicity values Mice intravenous LD50: 93 mg/kg Rat intravenous LD50: 71 mg/kg Rabbit intravenous LD50: 40 mg/kg Dog intravenous LD50: 32 mg/kg Mice intravenous LD50: 452 mg/kg /Esmolol deesterified metabolite (ASL 8123)/ In clinical use, esmolol hydrochloride (infusion rate 50-300 μg/kg/min) was associated with mild to moderate adverse reactions, including hypotension (15%), bradycardia (12%), and nausea (4%); no serious hepatotoxicity or nephrotoxicity has been reported [2,3] -Esmolol hydrochloride has a plasma protein binding rate of 55% in human plasma [1] -The acute intravenous LD50 of esmolol hydrochloride in mice is 110 mg/kg, and 130 mg/kg in rats[4] - No significant drug interactions were observed when used in combination with anesthetics (isoflurane, propofol), vasopressors or antiarrhythmics[3] |
| References | |
| Additional Infomation |
Esmolol hydrochloride is the hydrochloride salt form of esmolol, a short-acting, rapid-acting β-adrenergic receptor antagonist, belonging to class II antiarrhythmic drugs, and lacking intrinsic sympathomimetic activity. Esmolol hydrochloride competitively blocks β1-adrenergic receptors in the myocardium, reducing myocardial contractility and heart rate, thereby decreasing cardiac output and myocardial oxygen consumption. It also reduces central sympathetic output and inhibits renin secretion. At high doses, esmolol hydrochloride can also block β2 receptors located in bronchial and vascular smooth muscle, leading to smooth muscle relaxation.
See also: Esmolol (containing the active ingredient). Mechanism of Action Esmolol selectively inhibits the response to adrenergic stimulation by competitively blocking cardiac β1-adrenergic receptors, while having little effect on β2-adrenergic receptors in bronchial and vascular smooth muscle. At high doses (e.g., greater than 300 μg/kg/min), esmolol's selectivity for β1-adrenergic receptors typically decreases, as the drug competitively inhibits both β1 and β2 adrenergic receptors. Esmolol produces negative chronotropic and negative inotropic effects by inhibiting myocardial β1-adrenergic receptors. Esmolol reduces resting and exercise-induced heart rate, reflex orthostatic tachycardia, myocardial contractility, left ventricular pressure rise rate (dp/dt), right ventricular contractility, and cardiac index through its myocardial β1-adrenergic blocking effect. The decrease in myocardial contractility, arterial blood pressure, and heart rate induced by esmolol can lead to a reduction in myocardial oxygen consumption, which may explain its effectiveness in myocardial ischemia. Therapeutic Use Adrenergic Beta-Blockers Esmolol is primarily used intravenously for rapid, temporary control of ventricular rate in patients with supraventricular tachycardia (SVT) (e.g., atrial flutter and/or atrial fibrillation, sinus tachycardia). Esmolol may be superior to long-acting drugs. Because beta-adrenergic blockers have a rapid onset and short duration of action, they can be used for short-term control of ventricular rate in patients with supraventricular tachycardia (SVT)... Esmolol is indicated for rapid, short-term control of ventricular rate in patients with atrial fibrillation or atrial flutter during the perioperative, postoperative, or other emergency periods. It is also indicated for uncompensated sinus tachycardia where the physician deems intervention necessary. (Esmolol is used to control heart rate in patients with myocardial ischemia. /Not included in US product label/) It is not recommended for use in chronic cases where a switch to another medication is anticipated. /Included in US Product Label/ For more complete data on the therapeutic uses of esmolol hydrochloride (6 types), please visit the HSDB record page. Drug Warnings Because esmolol carries a risk of inducing hypotension, blood pressure should be closely monitored during treatment with this drug, especially in patients with pre-treatment low blood pressure (e.g., systolic blood pressure below 105 mmHg). Esmolol can cause hypotension at any dose level, but it is usually dose-related; the manufacturer recommends doses exceeding 200 μg/kg/min for the treatment of supraventricular tachycardia.Hypotension usually reverses within 30 minutes after discontinuation of the drug or reduction of the intravenous infusion rate. …Esmolol is contraindicated in patients with second- or third-degree atrioventricular block, sinus bradycardia, cardiogenic shock, or significant heart failure. Esmolol may mask the signs and symptoms of hypoglycemia (e.g., tachycardia, palpitations, changes in blood pressure, tremors, anxiety, but not sweating or dizziness) and may enhance insulin-induced hypoglycemia; therefore, caution should be exercised when using this drug in patients with diabetes or hypoglycemia. Because the deesterified metabolite of esmolol (ASL 8123) is primarily excreted by the kidneys, caution should be exercised when using this drug in patients with renal impairment, especially severe renal impairment. For more complete data on drug warnings for esmolol hydrochloride (16 in total), please visit the HSDB record page. Esmolol hydrochloride is a short-acting, highly selective β1-adrenergic receptor antagonist with a rapid onset (1-2 minutes) and short duration of action, attributed to its rapid metabolism by erythrocyte esterases [1]. -Clinically approved indications include acute supraventricular tachycardia, intraoperative and postoperative tachycardia/hypertension, and intraoperative myocardial ischemia prevention [3,4]. - This drug reduces myocardial oxygen consumption by lowering heart rate, contractility, and blood pressure, and does not produce significant bronchoconstriction due to its low affinity for β2 receptors [4] - In severe sepsis, esmolol hydrochloride can regulate hyperadrenergic state and reduce the release of pro-inflammatory cytokines, thereby improving hemodynamic stability [2] |
| Molecular Formula |
C16H26CLNO4
|
|
|---|---|---|
| Molecular Weight |
331.83
|
|
| Exact Mass |
331.155
|
|
| Elemental Analysis |
C, 57.91; H, 7.90; Cl, 10.68; N, 4.22; O, 19.29
|
|
| CAS # |
81161-17-3
|
|
| Related CAS # |
Esmolol-d7 hydrochloride; 1346598-13-7
|
|
| PubChem CID |
104769
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.026
|
|
| Boiling Point |
430.2ºC at 760 mmHg
|
|
| Melting Point |
48-50ºC
|
|
| Flash Point |
214ºC
|
|
| LogP |
2.722
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
5
|
|
| Rotatable Bond Count |
10
|
|
| Heavy Atom Count |
22
|
|
| Complexity |
288
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
Cl.O=C(CCC1C=CC(OCC(CNC(C)C)O)=CC=1)OC
|
|
| InChi Key |
GEKNCWBANDDJJL-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C16H25NO4.ClH/c1-12(2)17-10-14(18)11-21-15-7-4-13(5-8-15)6-9-16(19)20-3;/h4-5,7-8,12,14,17-18H,6,9-11H2,1-3H3;1H
|
|
| Chemical Name |
methyl 3-[4-[2-hydroxy-3-(propan-2-ylamino)propoxy]phenyl]propanoate;hydrochloride
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.75 mg/mL (8.29 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.75 mg/mL (8.29 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.75 mg/mL (8.29 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 100 mg/mL (301.36 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0136 mL | 15.0680 mL | 30.1359 mL | |
| 5 mM | 0.6027 mL | 3.0136 mL | 6.0272 mL | |
| 10 mM | 0.3014 mL | 1.5068 mL | 3.0136 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Effect of Controlled Hypotension on Cerebral Oxygen Saturation
CTID: NCT02967029
Phase: Phase 4 Status: Completed
Date: 2021-09-01
--
A prospective, randomized, double blinded, crossover, two-treatment, two-sequence, short term pharmacokinetic, pharmacodynamic and tolerability, single centre study to compare AOP200704 vs. Esmolol in healthy subjects.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2011-02-04
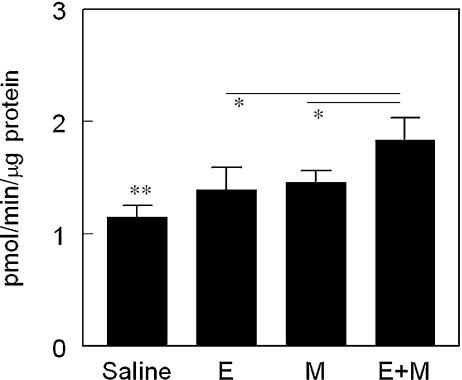
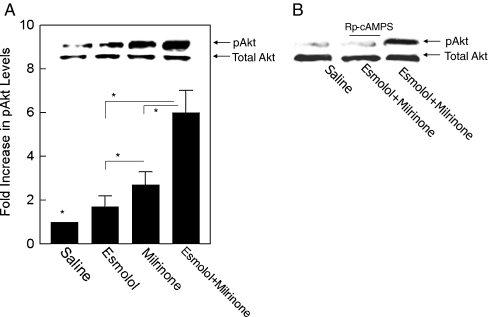
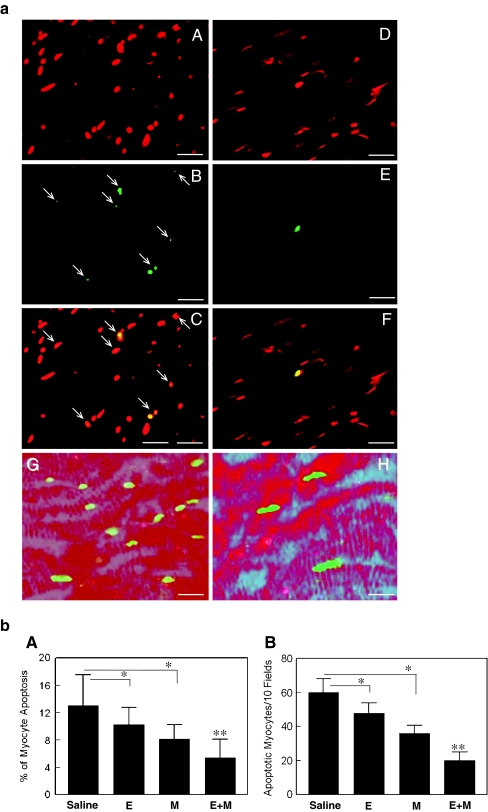
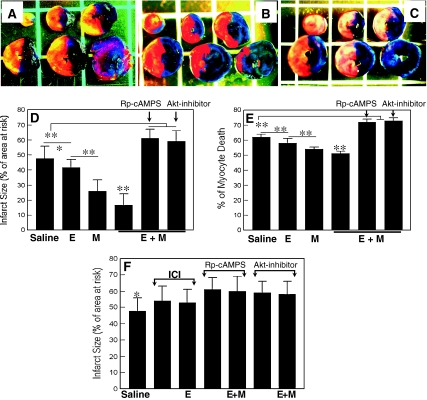 Panels A, B and C show representative cases of IS in the animals treated with saline (a), esmolol + milrinone (E + M) (b) and E + M in the presence of Rp-cAMPS (c). Infarcted tissue is indicated with yellow color.Cardiovasc Drugs Ther. 2011 Jun; 25(3): 223–232. Panels A, B and C show representative cases of IS in the animals treated with saline (a), esmolol + milrinone (E + M) (b) and E + M in the presence of Rp-cAMPS (c). Infarcted tissue is indicated with yellow color.Cardiovasc Drugs Ther. 2011 Jun; 25(3): 223–232. |
|---|
  |
 |