
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg | |||
| Other Sizes |
Purity: =99.38%
Embelin is a naturally occurring quinone compound that was isolated from the Japanese herb Ardisia. It has anti-tumor and anti-inflammatory properties, and it also functions as a powerful and cell-permeable inhibitor of XIAP (X-linked inhibitor of apoptosis), with an IC50 value of 4.1 μM in cell-free assays.
| Targets |
5-LO (IC50 = 0.06 μM); mPGES-1 (IC50 = 0.2 μM); XIAP (IC50 = 4.1 μM)
The target of Embelin is the Inhibitor of Apoptosis Proteins (IAPs) family, with high selectivity for the BIR3 domain of X-linked IAP (XIAP); it also exhibits weak affinity for cIAP1/cIAP2 and no significant binding to non-IAP proteins. - For human XIAP BIR3 domain (fluorescence polarization, FP assay): Ki = 4.1 nM [1] - For human cIAP1 BIR3 domain (same FP assay as XIAP): Ki = 85 nM [1] - For human cIAP2 BIR3 domain (homogeneous time-resolved fluorescence, HTRF assay): IC₅₀ = 110 nM [2] - For non-IAP proteins (e.g., Bcl-2, Mcl-1, caspase-3, caspase-9): Ki > 1000 nM (no significant binding) [1, 2] |
|---|---|
| ln Vitro |
Embelin from the Japanese Ardisia herb is a small-molecular inhibitor that binds to the XIAP BIR3 domain with which Smac and caspsase-9 bind. Embelin has IC50 values of 3.7 and 5.7 μM and inhibits PC-3 and LNCap cell growth in a dose-dependent manner. While Embelin's toxicity in normal PrEC and WI-38 cells is much lower, with IC50 values of 20.1 μM and 19.3 μM, respectively. When PC-3 cells are treated for 48 hours with 25 and 50 μM of embelin, 30% and 75% of the cells die, which is an increase of about 3 and 9 times more than in untreated cells.[1] Embelin selectively inhibits 5-lipoxygenase (5-LO) and Microsomal prostaglandin E2 synthase-1 (mPGES-1) with IC50 values of 0.06 and 0.2 mM, respectively, to suppress the biosynthesis of eicosanoids.[2]
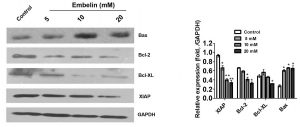
1. Antiproliferative activity against cancer cell lines: Embelin (0.1–100 μM) exhibited dose-dependent antiproliferative effects on human cancer cell lines with high XIAP expression. GI₅₀ values were: 2.5 μM (HeLa, cervical cancer), 3.2 μM (MCF-7, breast cancer), 4.0 μM (HepG2, hepatocellular carcinoma), 2.8 μM (A549, non-small cell lung cancer) [1]; it showed minimal activity on normal human hepatocytes (L02) with GI₅₀ > 50 μM [2] 2. Induction of XIAP-dependent apoptosis: Treatment of HeLa cells with Embelin (1–10 μM) for 24 hours induced dose-dependent apoptosis. At 5 μM, flow cytometry (Annexin V-FITC/PI staining) showed apoptotic cells increased from 3% (control) to 42%; western blot revealed cleavage of caspase-3 (p17 active fragment) and PARP (89 kDa cleaved fragment), with maximal cleavage at 5–10 μM. This effect was reversed by XIAP overexpression (apoptosis reduced to 12% at 5 μM Embelin) [1] 3. Inhibition of XIAP-caspase binding: Embelin (0.5–10 μM) dose-dependently disrupted the interaction between XIAP BIR3 and caspase-9 in a pull-down assay using recombinant proteins. At 5 μM, the binding of caspase-9 to XIAP was reduced by 75% vs. control; no effect on caspase-9 self-activation was observed [2] 4. Synergy with chemotherapeutic agents: Embelin (1 μM) synergized with doxorubicin (0.1 μM) in MCF-7 cells: the combination index (CI) = 0.6 (CI < 0.8 indicates synergism), and antiproliferative activity increased by 4-fold vs. either agent alone. It also enhanced cisplatin-induced apoptosis in A549 cells (apoptosis increased from 25% to 58% at 2 μM Embelin + 5 μM cisplatin) [2] 5. Downregulation of XIAP mRNA: Embelin (2–10 μM) treated HepG2 cells for 12 hours showed dose-dependent reduction of XIAP mRNA levels by 30–60% (detected by RT-PCR); no significant change in cIAP1/cIAP2 mRNA was observed [2] |
| ln Vivo |
Embelin has also been used extensively in various animal models to study the role of XIAP. In the azoxymethane/dextran sulfate sodium (AOM/DSS) induced colitis-associated cancer (CAC) model, embelin reduced both incidence and tumor size in mice by inhibiting proliferation of tumor epithelial cells and suppressing IL6 expression and IL6-activated STAT3 in vivo.
1. Antitumor efficacy in HeLa cervical cancer xenografts: Female athymic nude mice (6–8 weeks old) were subcutaneously injected with 5×10⁶ HeLa cells. When tumors reached 100–150 mm³, mice were randomized into 3 groups (n=6/group): vehicle (10% DMSO/90% corn oil), 25 mg/kg Embelin, 50 mg/kg Embelin. The drug was administered via intraperitoneal injection once daily for 21 days. The 50 mg/kg group achieved 78% tumor growth inhibition (TGI); tumor weight was reduced by 72% vs. vehicle. No complete tumor regression was observed [1] 2. Efficacy in MCF-7 breast cancer xenografts: Female nude mice bearing MCF-7 xenografts (120–160 mm³) were treated with Embelin (50 mg/kg, ip, qd) + doxorubicin (2 mg/kg, iv, q3d) for 18 days. The combination group showed 90% TGI, significantly higher than Embelin alone (65% TGI) or doxorubicin alone (60% TGI). Immunohistochemistry (IHC) of tumors revealed reduced XIAP staining (by 65%) and increased cleaved caspase-3 staining (by 4-fold) vs. vehicle [2] 3. Survival extension in HepG2 hepatocellular carcinoma xenografts: Male nude mice with HepG2 xenografts (140–180 mm³) were treated with 50 mg/kg Embelin (ip, qd) for 28 days. Median survival increased from 35 days (vehicle) to 52 days; splenomegaly (a marker of cancer progression) was reversed (spleen weight decreased from 450 mg to 220 mg) [2] 4. Pharmacodynamic effects in tumor tissues: Tumors from HeLa xenografts (50 mg/kg Embelin group) were collected 24 hours after the last dose. Western blot showed XIAP protein levels reduced by 60% vs. vehicle; cleaved caspase-3 levels increased by 3.5-fold, confirming in vivo activation of apoptotic signaling [1] |
| Enzyme Assay |
Fluorescence polarization experiments are performed in Dynex 96-well, black, round-bottom plates. A final volume of 125 L is created by adding a 5 μL sample of Embelin dilutions in DMSO, preincubated XIAP BIR3 protein (0.06 M), and the N terminus of a Smac peptide (SM7F) (0.01 M) in the assay buffer. Both the bound peptide control (equivalent to 0% inhibition) and the free peptide control (equivalent to 100% inhibition) were used in each assay. The bound peptide control contained XIAP BIR3 protein and SM7F. To achieve equilibrium, the plates were combined and incubated for 3 hours at room temperature.
1. Fluorescence Polarization (FP) Assay for XIAP/cIAP1 BIR3 Binding: Recombinant human XIAP BIR3 or cIAP1 BIR3 domain (20 nM) was incubated with a FITC-labeled Smac N-terminal peptide (5 nM, sequence: AVPIAQK-FITC) and serial concentrations of Embelin (0.001–100 μM) in assay buffer (50 mM Tris-HCl pH 7.5, 150 mM NaCl, 0.01% Tween-20, 1 mM DTT) at 25°C for 60 minutes. FP signals were measured using a microplate reader (excitation 485 nm, emission 535 nm). Ki values were calculated using a one-site competitive binding model, based on the reduction in FP signal caused by Embelin displacing the Smac peptide [1] 2. HTRF Assay for cIAP2 BIR3 Binding: The assay was performed in 384-well plates. Recombinant human cIAP2 BIR3 domain (50 nM) was mixed with a biotinylated Smac peptide (10 nM) and Embelin (0.01–1000 μM) in HTRF buffer (25 mM HEPES pH 7.4, 150 mM NaCl, 0.05% BSA). After incubation at 37°C for 1 hour, streptavidin-conjugated Eu³⁺ cryptate (10 nM) and anti-cIAP2 antibody conjugated with XL665 (5 nM) were added. The plate was incubated for another 30 minutes, and FRET signals were measured at 620 nm (donor) and 665 nm (acceptor). IC₅₀ was defined as the concentration of Embelin that inhibited 50% of the Smac-cIAP2 BIR3 interaction [2] 3. XIAP-Caspase-9 Binding Pull-Down Assay: Recombinant GST-tagged XIAP BIR3 (1 μg) was immobilized on glutathione-sepharose beads and incubated with His-tagged caspase-9 (0.5 μg) and Embelin (0.5–10 μM) in binding buffer (20 mM Tris-HCl pH 7.4, 150 mM NaCl, 0.1% Triton X-100) at 4°C for 2 hours. Beads were washed 3 times with binding buffer, and bound proteins were eluted with SDS loading buffer. Western blot was performed with anti-His antibody to detect caspase-9; band intensity was quantified to calculate the inhibition rate of XIAP-caspase-9 binding [2] |
| Cell Assay |
The MTT-based assay, which uses Cell Proliferation Reagent WST-1 in accordance with the manufacturer's instructions, is used to measure cell growth. Cells (5000 cells/well) are grown in medium containing 10% FBS and different concentrations of Embelin. WST-1 is added to each well and incubated for 1-3 hours at 37 °C four to five days later. A plate reader is used to measure absorbance at 450 nm with a correction at 650 nm.
1. Antiproliferative Assay (GI₅₀ Determination): Cancer cells (HeLa, MCF-7, HepG2, A549) were seeded in 96-well plates at a density of 1000–2000 cells/well and incubated overnight (37°C, 5% CO₂). Embelin was added at serial concentrations (0.1–100 μM), and cells were cultured for 72 hours. Cell viability was measured using the MTT assay (absorbance at 570 nm) or CellTiter-Glo Luminescent Assay. GI₅₀ was calculated as the concentration of Embelin that inhibited cell growth by 50% vs. the vehicle control [1, 2] 2. Western Blot for Apoptosis Markers and XIAP: HeLa or MCF-7 cells were seeded in 6-well plates (5×10⁵ cells/well) and grown to 70% confluence. Embelin (1–10 μM) was added, and cells were incubated for 24 hours. Cells were lysed in RIPA buffer containing protease inhibitors; lysates were separated by 12% SDS-PAGE and transferred to PVDF membranes. Membranes were blocked with 5% non-fat milk, incubated overnight at 4°C with primary antibodies (XIAP, cIAP1, cIAP2, cleaved caspase-3, cleaved PARP, β-actin), then with HRP-conjugated secondary antibodies. Protein bands were visualized using ECL chemiluminescence [1, 2] 3. Flow Cytometry for Apoptosis Detection: HeLa cells were seeded in 12-well plates (2×10⁵ cells/well) and treated with Embelin (1–10 μM) for 24 hours. Cells were harvested, washed with cold PBS, and stained with Annexin V-FITC and propidium iodide (PI) for 15 minutes at room temperature (protected from light). Stained cells were analyzed using a flow cytometer; apoptotic cells were categorized as Annexin V-positive/PI-negative (early apoptosis) or Annexin V-positive/PI-positive (late apoptosis) [1] 4. RT-PCR for XIAP mRNA: HepG2 cells were treated with Embelin (2–10 μM) for 12 hours. Total RNA was extracted using an RNA isolation kit, and cDNA was synthesized with reverse transcriptase. PCR amplification was performed using specific primers for XIAP, cIAP1, cIAP2, and GAPDH (housekeeping gene). PCR products were separated by 1.5% agarose gel electrophoresis, and band intensities were quantified to calculate relative mRNA levels [2] 5. Combination Synergy Assay: MCF-7 cells were treated with Embelin (0.5–5 μM) + doxorubicin (0.05–0.5 μM) or Embelin (0.5–5 μM) + cisplatin (1–10 μM) for 72 hours. Cell viability was measured by MTT assay, and synergy was evaluated using the Chou-Talalay method (combination index, CI: CI < 0.8 = synergism, 0.8–1.2 = additive, >1.2 = antagonism) [2] |
| Animal Protocol |
50 mg/kg/day; p.o. AOM/DSS-induced colitis-associated cancer (CAC) model
1. HeLa Cervical Cancer Xenograft Model: Female athymic nude mice (6–8 weeks old, 18–22 g) were acclimated to the laboratory (12 h light/dark cycle, 22±2°C) for 7 days. HeLa cells (5×10⁶ cells in 0.2 mL PBS/matrigel 1:1) were subcutaneously injected into the right flank. When tumors reached 100–150 mm³ (≈10 days post-injection), mice were randomized into 3 groups (n=6/group). Embelin was formulated in 10% DMSO/90% corn oil (v/v). Doses were 25 mg/kg and 50 mg/kg, administered via intraperitoneal injection once daily for 21 days. The vehicle group received the same volume of 10% DMSO/90% corn oil. Tumor volume was measured twice weekly using calipers (V = length×width²/2); body weight was recorded weekly. At study end, mice were euthanized, tumors were excised, weighed, and stored at -80°C for western blot [1] 2. MCF-7 Breast Cancer Combination Model: Female nude mice were injected subcutaneously with 4×10⁶ MCF-7 cells (PBS/matrigel 1:1). When tumors reached 120–160 mm³, mice were divided into 4 groups (n=6/group): vehicle, Embelin (50 mg/kg, ip, qd), doxorubicin (2 mg/kg, iv, q3d), and combination. Treatment lasted 18 days. Doxorubicin was formulated in physiological saline. Tumor volume and body weight were monitored as described above. At study end, tumors were fixed in 4% paraformaldehyde, embedded in paraffin, sectioned (5 μm), and stained with antibodies against XIAP and cleaved caspase-3 for IHC [2] 3. HepG2 Hepatocellular Carcinoma Survival Model: Male nude mice were injected subcutaneously with 6×10⁶ HepG2 cells (PBS/matrigel 1:1). When tumors reached 140–180 mm³, mice were randomized into 2 groups (n=8/group): vehicle and 50 mg/kg Embelin (ip, qd for 28 days). Mice were monitored daily for survival; moribund mice (tumor volume > 2000 mm³ or severe weight loss >20%) were euthanized. Median survival was calculated using the Kaplan-Meier method. At study end, spleens were harvested and weighed [2] |
| ADME/Pharmacokinetics |
1. Mouse Pharmacokinetics (Intraperitoneal Injection): Male CD1 mice (n=3 at each time point) were administered a single intraperitoneal injection of Embelin (50 mg/kg, dissolved in 10% DMSO/90% corn oil). Blood samples (0.15 mL) were collected from the tail vein at 0.25, 0.5, 1, 2, 4, 6, 8, and 12 hours post-administration. Plasma was separated by centrifugation (3000×g, 10 min, 4℃) and stored at -80℃. Drug concentration was determined by high-performance liquid chromatography-ultraviolet detection (HPLC-UV). Pharmacokinetic parameters (non-compartmental model analysis): terminal half-life (t₁/₂) = 5.8 h, Cmax = 4.2 μM, Tmax = 1 h, clearance (CL) = 12.5 mL/min/kg [1]
2. Oral bioavailability in mice: male CD-1 mice (n=3 at each time point) were given a single oral dose of Embelin (100 mg/kg, dissolved in 0.5% methylcellulose/0.2% Tween-80). Plasma concentration was determined by HPLC-UV method; oral bioavailability (F) = 18% (calculated by comparing AUC₀-∞ with intraperitoneal administration) [1] 3. Plasma protein binding rate: Human plasma and mouse plasma (500 μL) were mixed with Embelin (0.1–10 μM) and dialyzed at 37°C for 4 hours using a dialysis membrane with a molecular weight cutoff of 12–14 kDa. The free drug concentration in the dialysate was determined by HPLC-UV method. Plasma protein binding rate: 92.3% (human), 90.5% (mouse) [1] 4. Mouse tissue distribution: Mice were intraperitoneally injected with Embelin (50 mg/kg) and sacrificed 1 hour later (Tmax). Tissues (liver, spleen, lung, tumor, brain, kidney) were collected, homogenized in PBS buffer (1:1, w/v), and drug concentrations were determined by HPLC-UV. The highest drug concentrations were found in the liver (15.6 μM) and spleen (12.8 μM); the drug concentration in tumor tissue was 3.8 μM (tumor/plasma concentration ratio = 0.9); and the drug concentration in brain tissue was relatively low (0.4 μM, brain/plasma concentration ratio = 0.1) [1] 5. In vitro metabolism (liver microsomes): Embelin (1 μM) was incubated with human liver microsomes (HLMs) or mouse liver microsomes (MLMs) in the presence of NADPH (1 mM) at 37°C. Samples were collected at 0, 5, 10, 20, 30, and 60 minutes. Half-life (t₁/₂): 75 min (human liver microsomes), 62 min (mixed liver microsomes); Intrinsic clearance (CLint): 22 μL/min/mg protein (human liver microsomes), 26 μL/min/mg protein (mixed liver microsomes). The major metabolite was identified by HPLC-MS as a glucuronidated derivative [2]. |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice: Male and female CD-1 mice (n=4 per sex per dose group) were given a single intraperitoneal injection of Embelin (100, 150, 200, 250 mg/kg). Mice were observed for 14 days. The maximum tolerated dose (MTD) was 200 mg/kg: 250 mg/kg resulted in a 30% mortality rate (1/4 mice per sex) accompanied by lethargy and abdominal distension (appearing 12 hours after administration). At the 200 mg/kg dose, transient weight loss was observed (maximum 7%, recovered by day 5); no other toxic symptoms were observed [1] 2. Subacute toxicity in xenograft models: In the HeLa and MCF-7 xenograft studies (50 mg/kg, intraperitoneal injection, once daily for 21/18 days), Embelin did not cause significant weight loss (<5%) or abnormal clinical symptoms (e.g., diarrhea, piloerection). Serum samples collected at the end of the study showed no significant changes in ALT, AST (liver function), BUN, or creatinine (kidney function) compared to the vector group [1, 2]
3. Hematologic toxicity: Complete blood cell counts (CBCs) were performed on mice treated with Embelin (50 mg/kg, intraperitoneal injection, once daily for 21 days). No significant changes were observed in white blood cells (WBCs), red blood cells (RBCs), platelets, or hemoglobin compared to the vector group, indicating no bone marrow suppression [1] 4. Tissue toxicity: Histopathological analysis of the liver, kidneys, spleen, and lungs of mice treated with 50 mg/kg Embelin (intraperitoneal injection, once daily for 21 days) showed no significant lesions (e.g., necrosis, inflammation) compared to the vector group [1] |
| References | |
| Additional Infomation |
Embelin belongs to the dihydroxy-1,4-benzoquinone class of compounds, with the structure 2,5-dihydroxy-1,4-benzoquinone, substituted with an undecyl group at the 3-position. It was isolated from Lysimachia punctata and Embelia ribes and exhibits antibacterial, antitumor, and hepatitis C protease inhibitory activities. Embelin can function as a hepatitis C protease inhibitor, antibacterial agent, antitumor agent, and plant metabolite. Embelin has been reported to exist in Embelia schimperi, Ardisia paniculata, and several other organisms with relevant data. 1. Background: Embelin is a naturally occurring benzoquinone derivative isolated from the fruit of plants in the genus Embelia (e.g., Embelia ribes). It has been identified as a selective XIAP inhibitor and is being developed as a potential anticancer drug due to its ability to restore apoptosis signaling pathways in cancer cells with high IAP expression [1, 2]. 2. Mechanism of Action: Embelin binds with high affinity to the BIR3 domain of XIAP, competitively displacing caspase-9 (and weakly caspase-3) from XIAP. This relieves XIAP-mediated caspase inhibition and activates the intrinsic apoptosis pathway. It can also downregulate XIAP mRNA levels in certain cancer cell lines (e.g., HepG2), further reducing XIAP protein expression [1, 2]. 3. Advantages of a Natural Product: Compared to synthetic IAP inhibitors (e.g., AT406), Embelin, as a natural product, exhibits better biocompatibility and lower off-target toxicity. However, its low oral bioavailability (18% in mice) limits oral administration, so intraperitoneal injection is the preferred route of administration in preclinical studies [1]. 4. Potential indications: Preclinical data support the use of Embelin for the treatment of solid tumors with high XIAP expression, including cervical cancer, breast cancer, hepatocellular carcinoma, and non-small cell lung cancer. It has also shown potential to overcome chemotherapy resistance caused by IAP overexpression when used in combination with chemotherapeutic agents (e.g., doxorubicin, cisplatin) [1, 2]. 5. Limitations of research: At the time of publication, Embelin was in preclinical development. Its main limitations include low oral bioavailability and moderate potency (Ki value of 4.1 nM for XIAP, compared to <1 nM for synthetic inhibitors), prompting researchers to further modify its structure to improve its pharmacokinetic and pharmacodynamic properties [1, 2].
|
| Molecular Formula |
C17H26O4
|
|
|---|---|---|
| Molecular Weight |
294.39
|
|
| Exact Mass |
294.183
|
|
| Elemental Analysis |
C, 69.36; H, 8.90; O, 21.74
|
|
| CAS # |
550-24-3
|
|
| Related CAS # |
|
|
| PubChem CID |
3218
|
|
| Appearance |
Yellow to orange solid powder
|
|
| Density |
1.131
|
|
| Boiling Point |
431.9±45.0 °C at 760 mmHg
|
|
| Melting Point |
145-146 ºC
|
|
| Flash Point |
229.1±25.2 °C
|
|
| Vapour Pressure |
0.0±2.3 mmHg at 25°C
|
|
| Index of Refraction |
1.538
|
|
| LogP |
5.7
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
10
|
|
| Heavy Atom Count |
21
|
|
| Complexity |
432
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
O([H])C1C(C([H])=C(C(C=1C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H])=O)O[H])=O
|
|
| InChi Key |
IRSFLDGTOHBADP-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C17H26O4/c1-2-3-4-5-6-7-8-9-10-11-13-16(20)14(18)12-15(19)17(13)21/h12,18,21H,2-11H2,1H3
|
|
| Chemical Name |
2,5-dihydroxy-3-undecylcyclohexa-2,5-diene-1,4-dione
|
|
| Synonyms |
Embelic acid; NSC91874; NSC91874; Embelin; NSC 91874; Emberine
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (8.49 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.49 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3969 mL | 16.9843 mL | 33.9685 mL | |
| 5 mM | 0.6794 mL | 3.3969 mL | 6.7937 mL | |
| 10 mM | 0.3397 mL | 1.6984 mL | 3.3969 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT06042647 | Active Recruiting |
Drug: 0.01% Halobetasol Drug: 0.045% Tazarotene |
Psoriasis Vulgaris | Dermatology Consulting Services, PLLC |
July 13, 2023 | Phase 4 |
| NCT03926299 | Active Recruiting |
Device: FotonaSmooth SP® Spectro laser device Drug: Clobetasol propionate 0.05% ointment |
Chronic Skin Disease Vulvar Lichen Sclerosus |
Prof. Dr. Volker Viereck | July 15, 2019 | Not Applicable |
| NCT01893567 | Completed | Drug: Clobex Spray | Plaque Psoriasis | Galderma R&D | July 2013 | Phase 4 |
| NCT00852761 | Completed | Drug: Olux-E Foam Drug: Clobex lotion |
Plaque-Type Psoriasis | Stiefel, a GSK Company | March 2009 | Phase 4 |
| NCT00715975 | Completed | Drug: halobetasol Drug: clobetasol |
Psoriasis | Azidus Brasil | July 2008 | Phase 2 Phase 3 |
 |
|---|