
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
S1RA HCl (also known as E-52862 HCl) is a potent and selective sigma-1 receptor(σ1R, Ki=17 nM) antagonist, showed good selectivity against σ2R (Ki > 1000 nM). E-52862 showed high activity in the mouse capsaicin model of neurogenic pain, emerged as the most interesting candidate. In addition, compound 28 exerted dose-dependent antinociceptive effects in several neuropathic pain models. This, together with its good physicochemical, safety, and ADME properties, led compound 28 to be selected as clinical candidate.
| Targets |
Sigma-1 Receptor (Ki = 1.3 nM) [1]
- Sigma-2 Receptor (Ki > 10 μM, low affinity) [1] |
|---|---|
| ln Vitro |
For guinea pig σ1R and human σ1R, the Ki values of S1RA hydrochloride (0.1–10,000 nM) are 17 and 23.5 nM, respectively [1]. With a Ki value of 9.3 μM, S1RA hydrochloride (0.1-10000 nM) exhibits affinity for human and guinea pig σ2R [1]. S1RA hydrochloride has an IC50 value of 4.7 μM and a Ki value of 328 nM for human 5-HT2B, indicating that it is a potent receptor [1].
S1RA HCl (E-52862) is a selective sigma-1 receptor antagonist: it bound to sigma-1 receptor with high affinity (Ki = 1.3 nM) and showed negligible binding to sigma-2 receptor (Ki > 10 μM) or other receptors (e.g., opioid, cannabinoid receptors, Ki > 10 μM) [1] - It inhibited sigma-1 receptor-mediated cellular responses: in NG108-15 cells (neuroblastoma-glioma hybrid cells), it dose-dependently blocked SKF-10047 (sigma-1 agonist)-induced Ca²⁺ influx with an IC50 of 3.7 nM; 10 nM reduced Ca²⁺ peak amplitude by ~75% [1] - It suppressed neurotransmitter release in rat spinal cord slices: 1-10 μM S1RA HCl (E-52862) dose-dependently inhibited formalin-induced glutamate release (inhibition rate ~35% at 1 μM, ~68% at 10 μM) and substance P release (inhibition rate ~32% at 1 μM, ~62% at 10 μM) [2] - No significant cytotoxicity to NG108-15 cells, rat spinal neurons, or astrocytes at concentrations up to 100 μM (cell viability > 90% by MTT assay) [1][2] |
| ln Vivo |
In a model, the effects of capsaicin-induced mechanical hypersensitivity, formalin-induced nociception, and sciatic nerve damage induction are simulated by S1RA hydrochloride (16–80 mg/kg intraperitoneally or 32-128 mg/kg intracavitally; once).
In chronic constriction injury (CCI)-induced neuropathic pain model in rats, S1RA HCl (E-52862) exerted dose-dependent analgesic effects. Oral administration (3 mg/kg, 10 mg/kg, 30 mg/kg) increased paw withdrawal latency (PWL) to thermal stimuli by ~25% (3 mg/kg), ~48% (10 mg/kg), and ~65% (30 mg/kg) compared to vehicle group; intraperitoneal injection (1 mg/kg, 3 mg/kg) showed similar efficacy (PWL increased by ~30% and ~55%) [1] - It inhibited activity-induced spinal sensitization: in rats with repeated electrical stimulation of the sciatic nerve, oral S1RA HCl (E-52862) (10 mg/kg) reduced spinal dorsal horn neuron hyperexcitability by ~58% and blocked the increase in phosphorylated ERK (p-ERK) expression in spinal cord tissues [1] - In formalin-induced inflammatory pain model in rats, intraperitoneal S1RA HCl (E-52862) (3 mg/kg, 10 mg/kg) inhibited pain behavior in both phases: phase 1 (0-10 min, acute pain) inhibition rate ~32% (3 mg/kg) and ~50% (10 mg/kg); phase 2 (15-60 min, inflammatory pain) inhibition rate ~45% (3 mg/kg) and ~70% (10 mg/kg). It also reduced formalin-induced glutamate and substance P levels in spinal cord by ~48% and ~42% at 10 mg/kg [2] |
| Enzyme Assay |
Sigma-1 receptor binding assay: Membrane fractions from rat brain (enriched in sigma-1 receptors) were incubated with [³H]-pentazocine (sigma-1 selective ligand) and S1RA HCl (E-52862) (0.001-100 μM) at 25°C for 60 minutes. Unbound ligand was removed by filtration, and membrane-bound radioactivity was measured by liquid scintillation counting to calculate Ki value [1]
- Sigma-2 receptor binding assay: Membrane fractions from rat liver (enriched in sigma-2 receptors) were incubated with [³H]-DTG (sigma-2 selective ligand) and S1RA HCl (E-52862) (0.1-100 μM) at 25°C for 60 minutes. Filtration and radioactivity measurement were performed to evaluate binding affinity for sigma-2 receptor [1] |
| Cell Assay |
Ca²⁺ influx assay in NG108-15 cells: Cells were seeded in 96-well plates and loaded with fluorescent Ca²⁺ probe for 30 minutes. After pretreatment with S1RA HCl (E-52862) (0.01-100 nM) for 20 minutes, SKF-10047 (1 μM) was added to induce Ca²⁺ influx. Fluorescence intensity was measured in real-time to quantify Ca²⁺ concentration changes [1]
- Spinal cord neurotransmitter release assay: Rat spinal cord slices were prepared and placed in an organ bath containing oxygenated Krebs-Ringer buffer. After pretreatment with S1RA HCl (E-52862) (1-10 μM) for 30 minutes, formalin (1%) was added to stimulate neurotransmitter release. Culture supernatants were collected, and glutamate/substance P levels were detected by ELISA [2] |
| Animal Protocol |
Animal/Disease Models: Male CD1 mice [1]
Doses: 32, 64, 128 and 160 mg/kg Route of Administration: intraperitoneal (ip) injection; mechanical and thermal hypersensitivity [1]. 32, 64, 128 and 160 mg/kg, one Experimental Results:diminished persistence time on the rotarod in the rotarod test. Animal/Disease Models: Formalin-induced nociceptive male CD1 mice [1] Doses: 20, 40 and 80 mg/kg Route of Administration: intraperitoneal (ip) injection; 20, 40 and 80 mg/kg, one Experimental Results:Formalin Lin-induced nociceptive behavior in mice has a dose-dependent analgesic effect. Animal/Disease Models: Male CD1 mice with capsaicin-induced mechanical hypersensitivity [1] Doses: 16, 32, and 64 mg/kg (ip); 32, 64, and 128 mg/kg (oral) Route of Administration: i.p. Injectable or oral; 16, 32, and 64 mg/kg (ip); 32, 64, and 128 mg/kg (oral), once Experimental Results: Dose-dependent reversal of capsaicin-induced mechanical hypersensitivity in mice. CCI-induced neuropathic pain model: Male Sprague-Dawley rats underwent chronic constriction injury of the sciatic nerve. Seven days post-surgery, S1RA HCl (E-52862) was dissolved in 0.5% carboxymethylcellulose sodium (oral) or normal saline (intraperitoneal) and administered at doses of 1-30 mg/kg. Paw withdrawal latency (PWL) to thermal stimuli was measured at 1, 3, 6, 12 hours post-administration. Spinal cord tissues were collected for p-ERK expression analysis by Western blot [1] - Formalin-induced pain model: Male Wistar rats were randomly divided into vehicle and S1RA HCl (E-52862) treatment groups (3 mg/kg, 10 mg/kg). The drug was dissolved in normal saline and administered via intraperitoneal injection 30 minutes before formalin (5%, 50 μL) hind paw injection. Pain behavior (licking, biting) was scored for 60 minutes. Spinal cord tissues were collected to measure glutamate and substance P levels by ELISA [2] |
| ADME/Pharmacokinetics |
Oral bioavailability: approximately 70% in rats (oral dose 10 mg/kg) [1] - Plasma elimination half-life (t1/2): approximately 4.2 hours in rats [1] - Peak plasma concentration (Cmax): 1.8 μg/mL in rats (oral dose 10 mg/kg), 1.5 hours after administration [1] - Volume of distribution (Vd): 2.8 L/kg in rats [1] - Plasma protein binding: approximately 90% in rat plasma [1] - The drug is well distributed in the central nervous system (CNS), with a brain/plasma concentration ratio of approximately 0.8 2 hours after administration [1]
|
| Toxicity/Toxicokinetics |
In vitro experiments showed that S1RA HCl (E-52862) at concentrations up to 100 μM had no significant cytotoxicity to nerve cells [1][2]
- In vivo experiments showed that the acute intraperitoneal LD50 in mice was > 1000 mg/kg; the oral LD50 was > 2000 mg/kg [1] - Repeated oral administration in rats (30 mg/kg/day for 28 consecutive days) did not cause significant changes in body weight, organ indices (brain, liver, kidney) or serum ALT/AST/creatinine levels [1] - At therapeutic doses (1-30 mg/kg), no adverse effects on motor activity, anxiety-like behavior or cognitive function were observed in rats [1] |
| References |
|
| Additional Infomation |
S1RA HCl (E-52862) is a synthetic selective σ1 receptor antagonist[1][2]
- Its core mechanism is to competitively bind to σ1 receptors in the central nervous system (CNS), inhibiting σ1-mediated Ca²⁺ influx and neurotransmitter (glutamate, substance P) release, thereby blocking pain signal transmission and spinal cord sensitization[1][2] - In neuropathic and inflammatory pain models, it exhibits potent analgesic effects without significant sedation or motor side effects[1][2] - It can effectively penetrate the blood-brain barrier, supporting its use in the treatment of CNS-related pain[1] - In animal models, it can be administered orally or intraperitoneally, exhibiting good pharmacokinetic characteristics (high bioavailability, CNS distribution, moderate half-life) and low toxicity[1] |
| Molecular Formula |
C20H24CLN3O2
|
|---|---|
| Molecular Weight |
373.8765
|
| Exact Mass |
373.156
|
| CAS # |
1265917-14-3
|
| Related CAS # |
S1RA;878141-96-9
|
| PubChem CID |
50914801
|
| Appearance |
White to off-white solid powder
|
| LogP |
3.784
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
4
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
26
|
| Complexity |
416
|
| Defined Atom Stereocenter Count |
0
|
| InChi Key |
SHRYQZBTQDMGLZ-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C20H23N3O2.ClH/c1-16-14-20(25-13-10-22-8-11-24-12-9-22)21-23(16)19-7-6-17-4-2-3-5-18(17)15-19;/h2-7,14-15H,8-13H2,1H3;1H
|
| Chemical Name |
4-[2-(5-methyl-1-naphthalen-2-ylpyrazol-3-yl)oxyethyl]morpholine;hydrochloride
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~267.47 mM)
H2O : ~16.67 mg/mL (~44.59 mM) |
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.69 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.69 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (6.69 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 110 mg/mL (294.21 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6747 mL | 13.3733 mL | 26.7465 mL | |
| 5 mM | 0.5349 mL | 2.6747 mL | 5.3493 mL | |
| 10 mM | 0.2675 mL | 1.3373 mL | 2.6747 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
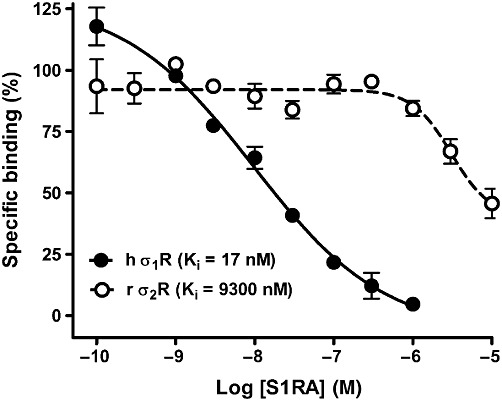 Binding of S1RA to σ1receptors (σ1R) and σ2receptors (σ2R).Br J Pharmacol.2012 Aug;166(8):2289-306. |
|---|
Effect of S1RA on electrophysiological recordings of spinal cord responses upon application of electrical C-fibre intensity stimuli. |
Dose–response effect of S1RA on the rotarod, capsaicin-induced mechanical hypersensitivity and formalin (phase I and II) tests.Br J Pharmacol.2012 Aug;166(8):2289-30 |