
| Size | Price | Stock | Qty |
|---|---|---|---|
| 25mg | |||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g | |||
| Other Sizes |
Purity: ≥98%
Desloratadine (formerly SCH34117, NSC 675447; SCH-34117, NSC-675447; Clarinex; Neoclarityn; Descarboethoxyloratadine; Aerius) is the active metabolite of loratadine which is an anti-histamine drug of the tricyclic class used to treat allergies. Desloratadine is a potent antagonist of the human histamine H1 receptor with an IC50 of 51 nM.
| Targets |
Histamine H1 receptor ( IC50 = 51 nM )
Histamine H1 receptor (H1R) (human H1R, Ki=0.1 nM; rat H1R, Ki=0.2 nM; guinea pig H1R, Ki=0.3 nM) [1,3] |
|---|---|
| ln Vitro |
In vitro activity: Desloratadine is a competitive antagonist of contractions induced by carbachol in isolated rabbit iris smooth muscle with pA2 of 6.67.[1] In order to displace tritiated mepyramine, desloratadine binds to the human H1 receptor with a Ki value of 0.87 nM. Desloratadine (100 nM to 10 μM) inhibits human basophils' production of the cytokines IL-4 and IL-13, both IgE-mediated and non-IgE-mediated. Human peripheral blood basophils' IgE- and non-IgE-mediated histamine release is inhibited by desloratadine (300 nM to 100 μM). Desloratadine (0.1 μM to 10 μM) has also been demonstrated to block the adhesion of eosinophils derived from patients with allergic asthma or rhinitis, as well as the chemotaxis of eosinophils induced by platelet-activating factor and TNF-α.[2] Desloratadine (1 μM–10 μM) inhibits histamine and LTC4 release from human basophils in a dose-dependent manner. Desloratadine (0.1 μM–10 μM) inhibits the release of IL-13 from human basophils that have been activated with IL-3 and PMA in a dose-dependent manner. In cultured basophils, desloratadine (10 μM) pretreatment significantly reduces the induced cytokine message. In cultured basophils, desloratadine (10 μM) pretreatment reduces the IL-4 message accumulated with anti-IgE activation by about 80%. Moreover, desloratadine (10 μM) suppresses the histamine and IL-4 protein released into the cultured basophils' supernatants.[3] [3H]Desloratadine binds to the human histamine H1 receptor that is expressed in CHO cells with Kd of 1.1 nM. In competition-binding studies, desloratadine is 52, 57, 194, and 153 times more potent than cetirizine, ebastine, fexofenadine, and loratadine, respectively.[4]
Compound 48/80 (1 μg/mL)-activated human peripheral blood mast cells were treated with Desloratadine (Sch34117) (0.001 μM-10 μM). It dose-dependently inhibited histamine, TNF-α, and IL-4 release, with 85% inhibition of histamine at 1 μM and IC50=0.08 μM [2] - LPS (1 μg/mL)-induced human monocyte-derived macrophages were treated with Desloratadine (Sch34117) (0.1 μM-20 μM). At 5 μM, it reduced IL-6/IL-8 secretion by 62%/58% and suppressed NF-κB p65 phosphorylation by 55% (Western blot) [4] - Histamine (10 μM)-stimulated human bronchial epithelial cells were treated with Desloratadine (Sch34117) (0.01 μM-10 μM). It inhibited H1R-mediated intracellular Ca²+ elevation, EC50=0.15 μM, and reduced eotaxin secretion by 48% at 2 μM [5] - Radioligand binding assay with human H1R-expressing HEK293 cell membranes showed Desloratadine (Sch34117) competitively displaced [3H]-pyrilamine, with high affinity (Ki=0.1 nM) [1] |
| ln Vivo |
Desloratadine (1 mg/mL, 3 mg/mL, and 10 mg/mL) causes a long-lasting, dose-dependent mydriasis in guinea pigs in vivo.[1] Desloratadine has an ED50 of 0.15 mg/kg in mice, which prevents histamine-induced paw edema. For guinea pigs, desloratadine (ED50 = 0.9 μg) prevents the increase in microvascular permeability that occurs when the upper airway is challenged with histamine.[2] In conscious mice, desloratadine (5 mg/kg) inhibits oxotremorine-induced tremor by rupturing the blood-brain barrier. A shift in the dose-response curve to the right in the pithed rat indicates that desloratadine (1.0 mg/kg) significantly inhibits decreases in dP/dt induced by oxotremorine (0.00125 mg/kg, 0.0025 mg/kg, and 0.02 mg/kg). A shift in the dose-response curve to the right in the pithed rat indicates that desloratadine (1.0 mg/kg) significantly inhibits decreases in dP/dt induced by oxotremorine (0.00125 mg/kg, 0.0025 mg/kg, and 0.02 mg/kg). [5]
Clinical trial in allergic rhinitis patients: Oral administration of Desloratadine (Sch34117) (5 mg once daily) for 2 weeks reduced total nasal symptom score (TNSS) by 70% and ocular symptom score by 65% compared to placebo. Symptom relief persisted for 24 hours [2] - Clinical trial in chronic idiopathic urticaria patients: Oral Desloratadine (Sch34117) (5 mg once daily) for 6 weeks reduced urticarial lesion count by 78% and pruritus score by 72%. No sedative effects were reported [3] - Rat passive cutaneous anaphylaxis (PCA) model: Intradermal injection of anti-ovalbumin IgE-sensitized rats were given Desloratadine (Sch34117) (1 mg/kg, 3 mg/kg) via oral gavage 1 hour before antigen challenge. The 3 mg/kg dose inhibited skin wheal area by 75% and Evans blue extravasation by 70% [1] - Mouse LPS-induced systemic inflammation model: Intraperitoneal injection of Desloratadine (Sch34117) (5 mg/kg, 10 mg/kg) 30 minutes before LPS (5 mg/kg) administration reduced serum TNF-α/IL-6 levels by 55%/60% (10 mg/kg dose) and alleviated lung inflammation [4] |
| Enzyme Assay |
H1R binding assay: Prepare membrane fractions from HEK293 cells expressing human/rat/guinea pig H1R or human brain tissue. Incubate membranes with [3H]-pyrilamine (0.5 nM) and various concentrations of Desloratadine (Sch34117) (0.0001 nM-100 nM) at 25°C for 60 minutes. Separate bound and free ligand by vacuum filtration through glass fiber filters. Measure radioactivity with a liquid scintillation counter and calculate Ki values using the Cheng-Prusoff equation [1,3]
|
| Cell Assay |
Mast cell degranulation assay: Isolate human peripheral blood mast cells via density gradient centrifugation. Resuspend cells in buffer and pre-treat with Desloratadine (Sch34117) (0.001 μM-10 μM) for 30 minutes. Stimulate with compound 48/80 (1 μg/mL) for 60 minutes at 37°C. Centrifuge to collect supernatant, measure histamine via fluorometric assay and TNF-α/IL-4 via ELISA [2]
- Macrophage inflammation assay: Differentiate human monocytes into macrophages in culture medium for 7 days. Seed macrophages in 24-well plates and pre-treat with Desloratadine (Sch34117) (0.1 μM-20 μM) for 1 hour, then stimulate with LPS (1 μg/mL) for 24 hours. Collect supernatant to quantify IL-6/IL-8 via ELISA; extract total protein to detect NF-κB p65 phosphorylation via Western blot [4] - Bronchial epithelial cell assay: Seed human bronchial epithelial cells in 24-well plates and incubate for 24 hours. Pre-treat with Desloratadine (Sch34117) (0.01 μM-10 μM) for 1 hour, then stimulate with histamine (10 μM) for 24 hours. Load cells with Ca²+ fluorescent probe to monitor intracellular Ca²+ concentration; collect supernatant to quantify eotaxin via ELISA [5] |
| Animal Protocol |
Dissolved in 20% polyethylene glycol-300; 10 mg/kg; s.c. injection
Male guinea-pigs Mast cell degranulation assay: Isolate human peripheral blood mast cells via density gradient centrifugation. Resuspend cells in buffer and pre-treat with Desloratadine (Sch34117) (0.001 μM-10 μM) for 30 minutes. Stimulate with compound 48/80 (1 μg/mL) for 60 minutes at 37°C. Centrifuge to collect supernatant, measure histamine via fluorometric assay and TNF-α/IL-4 via ELISA [2] - Macrophage inflammation assay: Differentiate human monocytes into macrophages in culture medium for 7 days. Seed macrophages in 24-well plates and pre-treat with Desloratadine (Sch34117) (0.1 μM-20 μM) for 1 hour, then stimulate with LPS (1 μg/mL) for 24 hours. Collect supernatant to quantify IL-6/IL-8 via ELISA; extract total protein to detect NF-κB p65 phosphorylation via Western blot [4] - Bronchial epithelial cell assay: Seed human bronchial epithelial cells in 24-well plates and incubate for 24 hours. Pre-treat with Desloratadine (Sch34117) (0.01 μM-10 μM) for 1 hour, then stimulate with histamine (10 μM) for 24 hours. Load cells with Ca²+ fluorescent probe to monitor intracellular Ca²+ concentration; collect supernatant to quantify eotaxin via ELISA [5] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
In healthy volunteers, oral administration of 5 mg desloratadine tablets once daily for 10 consecutive days showed an average time to peak concentration (Tmax) of approximately 3 hours, an average steady-state peak plasma concentration (Cmax) of 4 ng/ml, and an average steady-state area under the curve (AUC) of 56.9 ng/hr/ml. Similar distribution characteristics were observed using 10 ml of oral solution containing 5 mg desloratadine. Food was found not to affect the absorption of desloratadine. Approximately 87% of the 14C-desloratadine dose was recovered equally in urine and feces as metabolites. Metabolism/Metabolites Desloratadine is metabolized to the active metabolite 3-hydroxydesloratadine, which is subsequently glucuronidated. Desloratadine is a known human metabolite of rupatadine and loratadine. Elimination pathway: Desloratadine (the major metabolite of loratadine) is extensively metabolized to the active metabolite 3-hydroxydesloratadine, which is subsequently glucuroninated. Approximately 87% of the 14C-desloratadine dose is recovered equally in urine and feces. Half-life: 50 hours Biological half-life The mean plasma elimination half-life of desloratadine is approximately 27 hours. Absorption: Oral bioavailability in humans is 80-85%; peak plasma concentration (Cmax) is reached 3-4 hours after oral administration (5 mg dose: Cmax = 210 ng/mL) [2,5] -Distribution: Volume of distribution (Vd) in humans is 19-25 L/kg; brain/plasma concentration ratio <0.01, indicating negligible blood-brain barrier penetration [2] -Metabolism: Metabolized in the liver by cytochrome P450 (CYP) 3A4 and 2C8 to inactive metabolites; no significant first-pass metabolism [5] -Excretion: 40% of the dose is excreted in urine (30% as metabolites, 10% as the original drug), and 55% is excreted in feces. The elimination half-life (t1/2) in humans is 27-30 hours [2,5] - Plasma protein binding rate: The plasma protein binding rate of desloratadine (Sch34117) in human plasma is 83-87% [5] |
| Toxicity/Toxicokinetics |
Toxicity Summary
Like other H1 receptor blockers, desloratadine competitively binds to H1 receptors in the gastrointestinal tract, uterus, large blood vessels, and bronchial smooth muscle with free histamine. This blocks the effects of endogenous histamine, thus temporarily relieving histamine-induced adverse symptoms (e.g., nasal congestion, tearing). Pregnancy and Lactation Effects ◉ Lactational Use Overview Because the concentration of desloratadine in breast milk is expected to be low and it does not have sedative or anticholinergic effects, maternal use of desloratadine is unlikely to affect breastfed infants or milk production. Concomitant use of desloratadine with sympathomimetic drugs (e.g., pseudoephedrine) may have a negative impact on lactation. ◉ Effects on Breastfed Infants As of the revision date, no relevant published information was found. ◉ Effects on Lactation and Breast Milk Higher doses of injected antihistamines can lower basal serum prolactin levels in non-lactating women and early postpartum women. However, pre-administration of antihistamines by postpartum mothers does not affect suckling-induced prolactin secretion. Whether lower doses of oral antihistamines have the same effect on serum prolactin, and whether this effect on prolactin has any impact on breastfeeding success, is currently unstudied. Prolactin levels in established lactating mothers may not affect their ability to breastfeed. Protein Binding Desloratadine binds to plasma proteins at a rate of approximately 82% to 87%, while its active metabolite, 3-hydroxydesloratadine, binds at a rate of approximately 85% to 89%. Acute toxicity: LD50 in rats and mice > 5000 mg/kg (oral); no deaths or serious clinical symptoms were reported [1] - Chronic toxicity: No significant hepatotoxicity, hematologic abnormalities or organ weight changes were observed in rats after oral administration of desloratadine (Sch34117) (100 mg/kg/day) for 6 consecutive months [1] - Clinical side effects: Mild headache (2-3% of patients), fatigue (1-2%) and dry mouth (1%) have been reported. No sedative, anticholinergic or cardiotoxic side effects were observed at therapeutic doses [2,3] - Drug interactions: No significant interactions were observed with CYP450 isoenzyme substrates/inhibitors, warfarin, digoxin or oral contraceptives. No enhanced central nervous system depressant effect [5] |
| References | |
| Additional Infomation |
Desloratadine is a derivative of loratadine, in which the ethoxycarbonyl group on the piperidine ring is replaced by a hydrogen atom. As the main metabolite of loratadine, desloratadine is an antihistamine used to relieve allergy symptoms, including rhinitis and chronic urticaria. It does not readily enter the central nervous system and therefore does not cause drowsiness. Desloratadine has multiple functions, including as an H1 receptor antagonist, an antihistamine, a cholinergic antagonist, and a drug metabolite. Desloratadine is a second-generation tricyclic antihistamine with selective peripheral H1 receptor antagonistic activity. It is the active decarboxyethoxylated metabolite of loratadine (a second-generation histamine). Desloratadine has a long duration of action and does not readily enter the central nervous system, therefore it does not cause drowsiness. Desloratadine is a histamine-1 receptor antagonist. Its mechanism of action is as a histamine H1 receptor antagonist.
There are reports on the use of desloratadine in bovine (Bos taurus) with relevant data. Desloratadine is a long-acting piperidine derivative with selective H1 antihistamine activity and no sedative effect. Desloratadine reduces the typical effects of histamine on H1 receptors in bronchial smooth muscle, capillaries, and gastrointestinal smooth muscle, including vasodilation, bronchoconstriction, increased vascular permeability, pain, pruritus, and gastrointestinal smooth muscle spasmodic contractions. Desloratadine is used to relieve allergy symptoms. Desloratadine is a second-generation tricyclic antihistamine with selective peripheral H1 receptor antagonism. It is the active decarboxyethoxylated metabolite of loratadine (a second-generation histamine). Desloratadine has a long duration of action and does not readily enter the central nervous system, therefore it does not cause drowsiness. See also: desloratadine; pseudoephedrine sulfate (component). Indications For the relief of symptoms of seasonal allergic rhinitis and perennial (non-seasonal) allergic rhinitis. Desloratadine is also used to treat itching and urticaria (hives) symptoms associated with chronic idiopathic urticaria. FDA Label Neoclarityn is indicated for the relief of the following symptoms: allergic rhinitis; urticaria. Aerius is indicated for the relief of the following symptoms: allergic rhinitis; urticaria. Azomyr is indicated for the relief of the following symptoms: allergic rhinitis (see section 5.1); urticaria (see section 5.1). Treatment of allergic rhinitis and urticaria. Desloratadine ratiopharm is indicated for adults for the relief of the following symptoms: allergic rhinitis; chronic idiopathic urticaria as initially diagnosed by a physician. Desloratadine Teva is indicated for the relief of the following symptoms: allergic rhinitis; urticaria. Dasselta is indicated for the relief of the following symptoms: allergic rhinitis; urticaria. Aerius is indicated for the relief of the following symptoms: - Allergic rhinitis (see Section 5.1) - Urticaria (see Section 5.1) Opulis is indicated for the relief of the following symptoms: - Allergic rhinitis (see Section 5.1) - Urticaria (see Section 5.1) Mechanism of Action Like other H1 receptor antagonists, desloratadine competitively binds to H1 receptors in the smooth muscle of the gastrointestinal tract, uterus, large blood vessels, and bronchi with free histamine. This blocks the action of endogenous histamine, thereby temporarily relieving histamine-induced adverse reactions (e.g., nasal congestion, tearing). Pharmacodynamics Desloratadine is a long-acting second-generation H1 receptor antagonist with selective peripheral H1 receptor antagonism. Histamine is a chemical substance that can cause many symptoms of allergic reactions, such as tissue swelling. Histamine is released from cells that store histamine (mast cells) and attaches to other cells that have histamine receptors. Histamine binds to its receptors, activating cells and releasing other chemicals, resulting in what we commonly call an allergic reaction. Desloratadine blocks a histamine receptor (H1 receptor), thus preventing histamine from activating cells. Unlike most other antihistamines, desloratadine does not enter the brain via the bloodstream and therefore does not cause drowsiness. Desloratadine (Sch34117) is a second-generation non-sedating histamine H1 receptor antagonist with potent anti-allergic and anti-inflammatory activity [1,2,3,4,5] Its core mechanisms include competitive H1R antagonism (high affinity for H1R), inhibition of mast cell degranulation, inhibition of pro-inflammatory cytokine (TNF-α, IL-6, IL-8) secretion, and blocking of the NF-κB signaling pathway [1,2,4,5] Indications include allergic rhinitis (seasonal and perennial), chronic idiopathic urticaria, and allergic conjunctivitis, which can relieve symptoms such as sneezing, runny nose, and itching. Urticarial skin lesions and ocular symptoms [2,3] Extremely low blood-brain barrier penetration, therefore it has no sedative effect, which is a key advantage over first-generation antihistamines [2,5] Long elimination half-life (27-30 hours), supporting once-daily oral administration (5 mg per dose) in adults and adolescents [2,5] Consistent efficacy against various allergic diseases, and well-tolerated with long-term use; no dose adjustment is required in elderly patients or patients with mild to moderate renal/hepatic impairment [3,5] |
| Molecular Formula |
C19H19CLN2
|
|
|---|---|---|
| Molecular Weight |
310.82
|
|
| Exact Mass |
310.123
|
|
| Elemental Analysis |
C, 73.42; H, 6.16; Cl, 11.41; N, 9.01
|
|
| CAS # |
100643-71-8
|
|
| Related CAS # |
Desloratadine-d4; 381727-29-3; Desloratadine-d9; 1795024-82-6; Desloratadine-3,3,5,5-d4; 2713301-38-1; Desloratadine-d5; 1020719-34-9
|
|
| PubChem CID |
124087
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.2±0.1 g/cm3
|
|
| Boiling Point |
467.9±45.0 °C at 760 mmHg
|
|
| Melting Point |
150-151°C
|
|
| Flash Point |
236.8±28.7 °C
|
|
| Vapour Pressure |
0.0±1.2 mmHg at 25°C
|
|
| Index of Refraction |
1.626
|
|
| LogP |
6.77
|
|
| Hydrogen Bond Donor Count |
1
|
|
| Hydrogen Bond Acceptor Count |
2
|
|
| Rotatable Bond Count |
0
|
|
| Heavy Atom Count |
22
|
|
| Complexity |
425
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
ClC1C([H])=C([H])C2=C(C=1[H])C([H])([H])C([H])([H])C1C([H])=C([H])C([H])=NC=1/C/2=C1\C([H])([H])C([H])([H])N([H])C([H])([H])C\1([H])[H]
|
|
| InChi Key |
JAUOIFJMECXRGI-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C19H19ClN2/c20-16-5-6-17-15(12-16)4-3-14-2-1-9-22-19(14)18(17)13-7-10-21-11-8-13/h1-2,5-6,9,12,21H,3-4,7-8,10-11H2
|
|
| Chemical Name |
13-chloro-2-piperidin-4-ylidene-4-azatricyclo[9.4.0.03,8]pentadeca-1(11),3(8),4,6,12,14-hexaene
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.04 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.04 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (8.04 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 30% Propylene glycol , 5% Tween 80 , 65% D5W: 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2173 mL | 16.0865 mL | 32.1730 mL | |
| 5 mM | 0.6435 mL | 3.2173 mL | 6.4346 mL | |
| 10 mM | 0.3217 mL | 1.6086 mL | 3.2173 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Efficacy and Safety of Desloratadine vs. Fexofenadine 180 mg. vs. Placebo for Treating Seasonal Allergic Rhinitis (SAR)(Study P04053)(COMPLETED)
CTID: NCT00783211
Phase: Phase 4 Status: Completed
Date: 2024-08-15
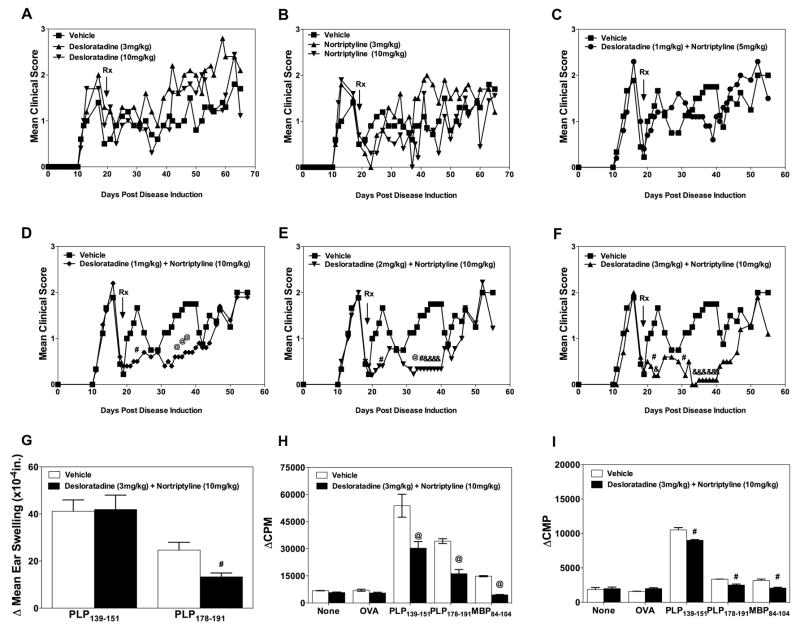
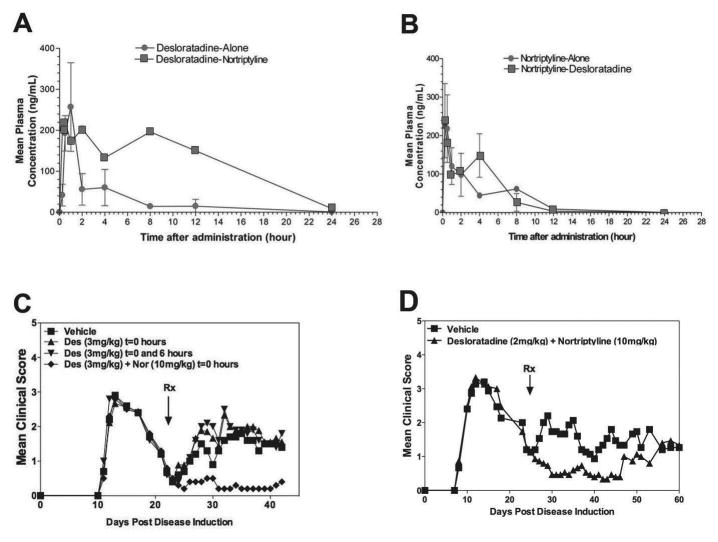
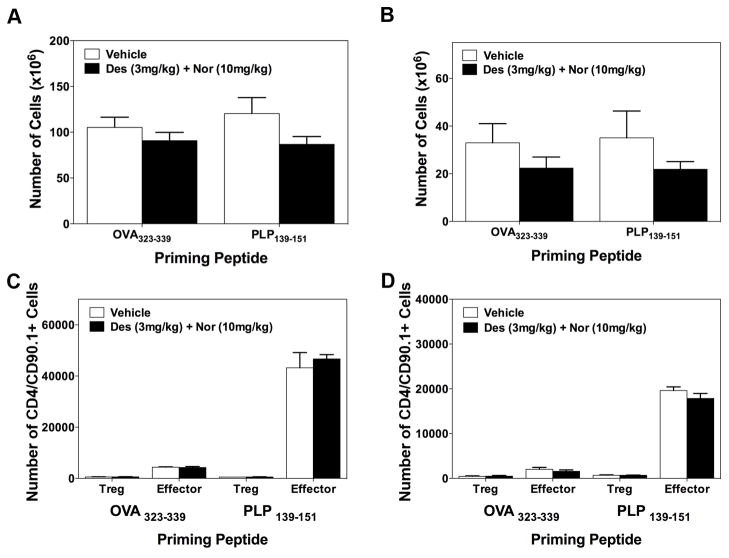
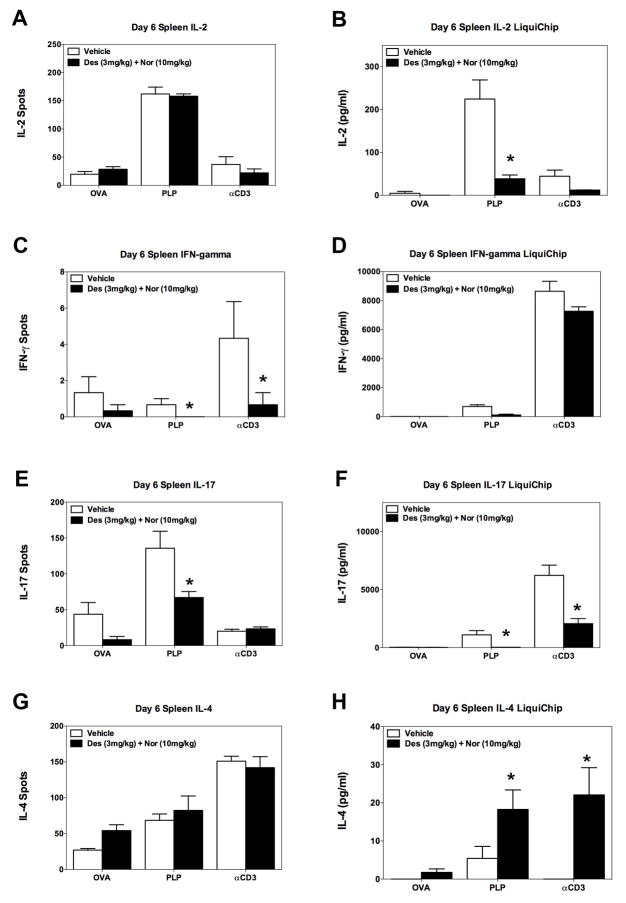
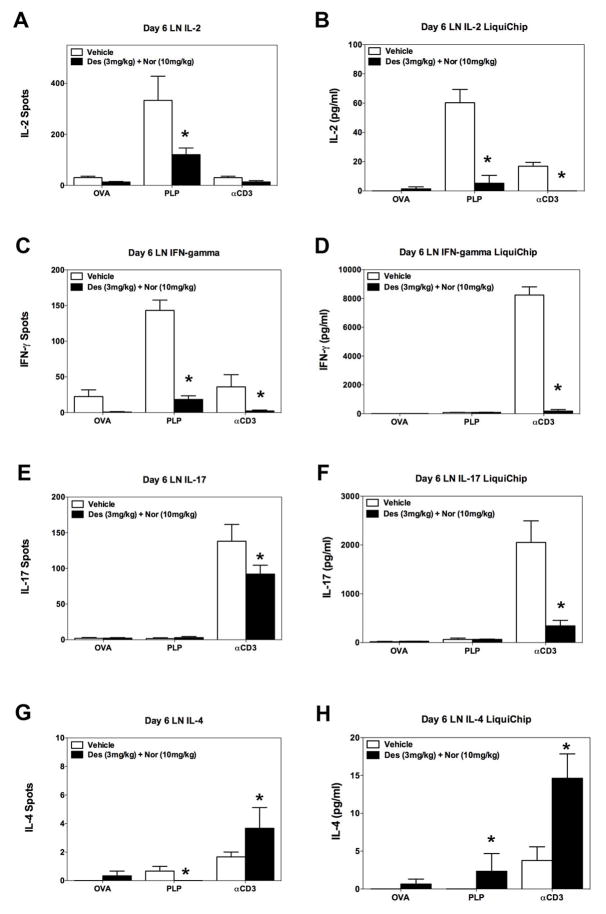
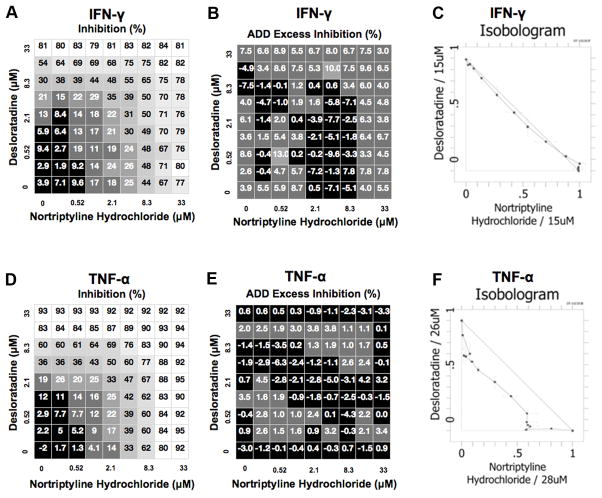 Identification of CRx-153, i.e., the combination of desloratadine and nortriptyline activity.Cell Immunol. 2011; 270(2): 237–250. Identification of CRx-153, i.e., the combination of desloratadine and nortriptyline activity.Cell Immunol. 2011; 270(2): 237–250. |
|---|
 |
 |
 |
|---|
 |
 |