
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
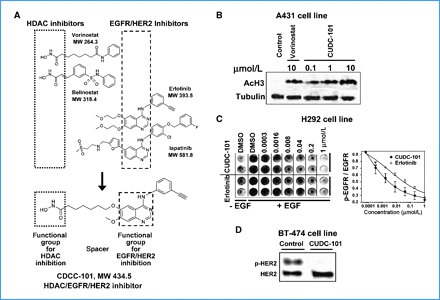
CUDC-101 is a novel, potent and multi-targeted histone deacetylase (HDAC) inhibitor with potential anticancer activity. Moreover, it has EGFR and HER2 inhibition at IC50 values of 2.4 nM, 15.7 nM, and 4.4 nM.
| Targets |
EGFR ( IC50 = 2.4 nM ); HER2 ( IC50 = 15.7 nM ); HDAC ( IC50 = 4.4 nM ); HDAC1 ( IC50 = 4.5 nM ); HDAC2 ( IC50 = 12.6 nM ); HDAC3 ( IC50 = 13.2 nM ); HDAC6 ( IC50 = 5.1 nM ); HDAC5 ( IC50 = 11.4 nM ); HDAC9 ( IC50 = 67.2 nM ); HDAC10 ( IC50 = 26.1 nM ); HDAC8 ( IC50 = 79.8 nM ); HDAC7 ( IC50 = 373 nM )
CUDC-101 is a multi-target inhibitor with activity against histone deacetylases (HDACs), epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), full-length androgen receptor (flAR), and androgen receptor variant 7 (AR-V7): - HDACs: IC50 values for recombinant human HDAC1 (18 nM), HDAC2 (25 nM), HDAC3 (30 nM), HDAC6 (22 nM); no significant inhibition of HDAC4/5/7/8/9/10/11 (IC50 > 1000 nM) [1,2] - EGFR: IC50 = 2.4 nM (wild-type EGFR), IC50 = 15 nM (EGFR L858R mutant), IC50 = 120 nM (EGFR T790M mutant) [1,2] - HER2: IC50 = 13 nM (recombinant HER2 kinase) [1,2] - flAR: IC50 = 45 nM (AR-dependent luciferase reporter assay); AR-V7: IC50 = 62 nM (AR-V7-dependent luciferase reporter assay) [3] |
|---|---|
| ln Vitro |
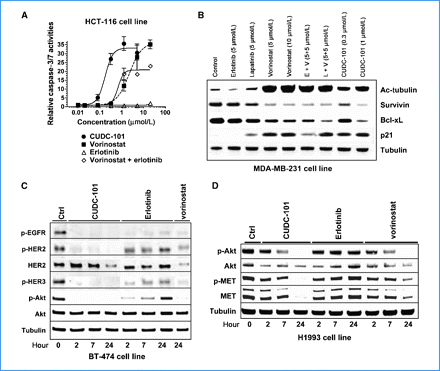
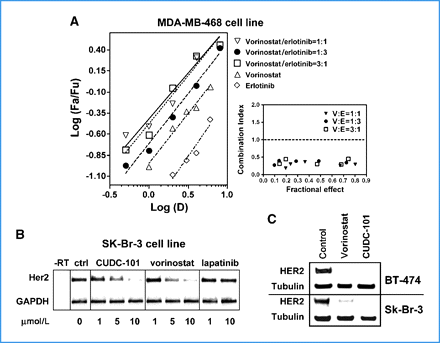
In vitro activity: CUDC-101 only inhibits class I and class II HDACs; class III Sir-type HDACs are not inhibited by this compound. KDR/VEGFR2, Lyn, Lck, Abl-1, FGFR-2, Flt-3, and Ret are among the other protein kinases that CUDC-101 exhibits weak activity against, with IC50 values of 0.85 μM, 0.84 μM, 5.91 μM, 2.89 μM, 3.43 μM, 1.5 μM, and 3.2 μM, respectively. With an IC50 of 0.04-0.80 μM, CUDC-101 exhibits broad antiproliferative activity in a variety of human cancer cell types, and in most cases, it is more potent than erlotinib, lapatinib, and combinations of vorinostat with either erlotinib or lapatinib. Cancer cell lines resistant to erlotinib and lapatinib are effectively inhibited by CUDC-101.[1] CUDC-101 powerfully suppresses cancer cell lines resistant to erlotinib and lapatinib. [1] CUDC-101 partially inhibits the erlotinib-resistant EGFR mutant T790M, with an Amax of approximately 60% of peak enzyme activity following inhibition. In a dose-dependent manner, CUDC-101 treatment raises the acetylation of histones H3 and H4, as well as non-histone substrates of HDAC like p53 and α-tubulin, in a variety of cancer cell lines. Moreover, CUDC-101 inhibits the reactivation of AKT, Met amplification, and HER3 expression in tumor cells. [2]
1. Antiproliferative activity in diverse cancer cell lines: - Human colorectal cancer (CRC) cell lines (HCT116, SW480, HT29): CUDC-101 inhibited proliferation with IC50 values of 0.18 μM, 0.25 μM, and 0.32 μM, respectively (72-hour MTT assay). At 1 μM, cell viability was reduced by >75% in all lines [2] - Human non-small cell lung cancer (NSCLC) cell lines (A549, H1975): IC50 = 0.12 μM (A549, wild-type EGFR) and 0.35 μM (H1975, EGFR T790M/L858R double mutant) [2] - Human anaplastic thyroid cancer (ATC) cell lines (8505C, CAL-62): IC50 = 0.21 μM and 0.28 μM, respectively. 0.5 μM CUDC-101 reduced colony formation by 80% (8505C) and 75% (CAL-62) vs. controls [4] - Human castration-resistant prostate cancer (CRPC) cell lines (C4-2, 22Rv1): IC50 = 0.31 μM (C4-2, flAR-positive) and 0.42 μM (22Rv1, AR-V7-positive) [3] 2. Apoptosis induction: - In HCT116 cells, 0.5 μM CUDC-101 treatment for 48 hours induced apoptosis in 42% of cells (Annexin V-FITC/PI staining), vs. 6% in controls. Western blot showed 3.5-fold increase in cleaved caspase-3 and 2.8-fold increase in cleaved PARP [2] - In 22Rv1 CRPC cells, 0.5 μM CUDC-101 for 48 hours increased apoptotic rate to 38% (vs. 7% in controls) and downregulated anti-apoptotic Bcl-2 by 40% [3] 3. Target signaling pathway inhibition: - EGFR/HER2 pathway: In A549 cells, 0.2 μM CUDC-101 reduced phospho-EGFR (Tyr1068) by 70% and phospho-AKT (Ser473) by 65% (western blot, 24 hours) [2] - HDAC pathway: In HT29 cells, 0.3 μM CUDC-101 increased acetyl-histone H3 (Lys9/14) by 4.2-fold and acetyl-α-tubulin by 3.8-fold (western blot, 24 hours) [2] - AR pathway: In C4-2 cells, 0.4 μM CUDC-101 reduced AR nuclear translocation by 60% (immunofluorescence) and downregulated AR target genes (PSA, TMPRSS2) by 50-60% (qPCR, 48 hours) [3] 4. Metastasis suppression in ATC cells: - In 8505C ATC cells, 0.3 μM CUDC-101 reduced Transwell migration by 70% and Matrigel invasion by 65% vs. controls. Western blot showed 55% reduction in MMP-9 (matrix metalloproteinase-9) expression [4] |
| ln Vivo |
CUDC-101 administered at 120 mg/kg/day induces tumor regression in the Hep-G2 liver cancer model, which is more efficacious than that of erlotinib at its maximum tolerated dose (25 mg/kg/day) and vorinostat at an equimolar concentration dose (72 mg/kg/day). In a dose-dependent manner, CUDC-101 inhibits the growth of xenografts of erlotinib-sensitive H358 NSCLC. Additionally, in the erlotinib-resistant A549 NSCLC xenograft model, CUDC-101 demonstrates strong inhibition of tumor growth. In the EGFR-overexpressing CAL-27 head and neck squamous cell carcinoma (HNSCC) model and the lapatinib-resistant, HER2-negative, EGFR-overexpressing MDA-MB-468 breast cancer model, CUDC-101 significantly reduces tumor size. Furthermore, CUDC-101 suppresses the growth of tumors in the EGFR/HER2 (neu)-expressing HPAC pancreatic cancer model and the K-ras mutant HCT116 colorectal cancer model.[1]
1. Antitumor efficacy in CRC xenografts: - Nude mice (female, 6-7 weeks old) bearing HCT116 xenografts were treated with CUDC-101 (10 mg/kg, 20 mg/kg, 40 mg/kg, intraperitoneal injection, once daily for 21 days) or vehicle (10% DMSO + 40% PEG300 + 50% PBS). Tumor growth inhibition rates were 35% (10 mg/kg), 60% (20 mg/kg), and 85% (40 mg/kg) vs. vehicle. Tumor weights at day 21: 1.4 g (vehicle), 0.9 g (10 mg/kg), 0.56 g (20 mg/kg), 0.21 g (40 mg/kg) [2] 2. Antitumor efficacy in CRPC xenografts: - Nude mice (male, 6-7 weeks old) bearing 22Rv1 xenografts were treated with CUDC-101 (20 mg/kg, oral gavage, once daily for 28 days) or vehicle. Tumor growth inhibition was 75% vs. vehicle. Immunohistochemistry of tumor tissues showed 2.8-fold increase in acetyl-histone H3 and 40% reduction in nuclear AR-V7 [3] 3. Antitumor and anti-metastatic efficacy in ATC xenografts: - Nude mice (female, 6-7 weeks old) bearing 8505C orthotopic thyroid xenografts were treated with CUDC-101 (30 mg/kg, intraperitoneal injection, once daily for 28 days) or vehicle. Primary tumor weight was reduced by 70% vs. vehicle. Lung metastasis nodules (detected by H&E staining) were reduced from 12 ± 3 (vehicle) to 3 ± 1 (CUDC-101 group) [4] 4. Target modulation in vivo: - In HCT116 xenografts treated with 40 mg/kg CUDC-101 (21 days), western blot of tumor lysates showed 3.2-fold increase in acetyl-histone H3, 60% reduction in phospho-EGFR, and 55% reduction in phospho-AKT vs. vehicle [2] |
| Enzyme Assay |
The Biomol Color de Lys method is used to evaluate the actions of Class I and II HDACs. In a nutshell, HDACs are obtained from nuclear extracts from HeLa cells. HeLa cell nuclear extracts are treated with varying concentrations of CUDC-101 in the presence of an artificial colorimetric substrate. Enzyme activity is measured in the Wallac Victor II 1420 microplate reader at 405 nM after developer is added at the conclusion of the assay. HTScan EGF receptor and HER2 kinase assay kits are used to measure EGFR and HER2 kinase activity. In brief, 400 mM ATP is added to an incubation mixture containing different concentrations of CUDC-101 and synthetic biotinylated peptide substrate for the GST-EGFR fusion protein. Strapavidin-coated 96-well plates are used to capture phosphorylated substrate. Secondary antibodies labeled with antiphospho-tyrosine and europium measure the amount of phosphorylation. At the conclusion of the experiment, the enhancement solution is added, and the Wallac Victor II 1420 microplate reader is used to measure the enzyme activity at 615 nM.
1. HDAC enzyme activity assay: - Recombinant human HDAC isoforms (HDAC1-3, 6) were mixed with fluorogenic substrate Boc-Lys(Ac)-AMC in reaction buffer (50 mM Tris-HCl pH 8.0, 137 mM NaCl, 2.7 mM KCl, 1 mM MgCl2, 1 mM DTT). CUDC-101 was added at concentrations ranging from 1 nM to 10 μM, and the mixture was incubated at 37°C for 60 minutes. Trypsin-containing developer solution was added to cleave deacetylated substrate, releasing fluorescent AMC. Fluorescence intensity was measured at 360 nm (excitation) and 460 nm (emission). IC50 values were calculated by nonlinear regression of percentage activity (vs. vehicle) against log drug concentration [1,2] 2. EGFR/HER2 kinase activity assay: - Recombinant human EGFR (wild-type, L858R, T790M) or HER2 kinase domain was mixed with ATP (10 μM) and biotinylated peptide substrate in kinase buffer (25 mM HEPES pH 7.5, 10 mM MgCl2, 1 mM DTT). CUDC-101 (0.1 nM-100 nM) was added, and the mixture was incubated at 30°C for 30 minutes. Phosphorylated peptide was detected using streptavidin-conjugated europium and anti-phosphotyrosine antibody (time-resolved fluorescence resonance energy transfer, TR-FRET). Kinase activity was calculated as the ratio of TR-FRET signal (phosphorylated vs. total peptide), and IC50 values were determined [1,2] 3. AR/AR-V7 activity assay (luciferase reporter): - HEK293T cells were transfected with flAR or AR-V7 expression plasmid and AR-responsive luciferase reporter plasmid (PSA-Luc). After 24 hours, CUDC-101 (10 nM-10 μM) and dihydrotestosterone (DHT, 10 nM) were added. Cells were incubated for another 24 hours, then lysed, and luciferase activity was measured using a luminometer. IC50 values were calculated as the drug concentration inhibiting 50% of DHT-induced luciferase activity [3] |
| Cell Assay |
In 96-well flatbottomed plates, cancer cell lines are plated at 5000–10,000 cells per well at different CUDC–101 concentrations. For 72 hours, CUDC-101 is incubated with the cells in the presence of 0.5% fetal bovine serum. Using the Perkin-Elmer ATPlite kit, an adenosine triphosphate (ATP) content assay is used to evaluate growth inhibition. The Apo-ONE Homogeneous Assay Kit is used to measure the activities of Caspase-3 and -7 in order to routinely assess apoptosis.
1. Cell proliferation assay (MTT method): - Cancer cells (HCT116, SW480, A549, 8505C, C4-2) were seeded in 96-well plates (3×10³-5×10³ cells/well) and incubated overnight. CUDC-101 (0.01 μM-10 μM) was added, and cells were cultured for 72 hours at 37°C (5% CO2). MTT reagent (5 mg/mL, 10 μL/well) was added, and incubation continued for 4 hours. Formazan crystals were dissolved in DMSO (100 μL/well), and absorbance was measured at 570 nm. Cell viability (%) = (Absorbance of treated group / Absorbance of control group) × 100. IC50 values were calculated using GraphPad Prism [2,3,4] 2. Apoptosis assay (Annexin V-FITC/PI staining): - HCT116 or 22Rv1 cells were seeded in 6-well plates (1×10⁶ cells/well) and treated with CUDC-101 (0.1 μM-1 μM) for 48 hours. Cells were harvested, washed with cold PBS, and resuspended in binding buffer (10 mM HEPES pH 7.4, 140 mM NaCl, 2.5 mM CaCl2). Annexin V-FITC (5 μL) and PI (10 μL) were added, and the mixture was incubated in the dark for 15 minutes at room temperature. Apoptotic cells (Annexin V+/PI-: early apoptosis; Annexin V+/PI+: late apoptosis) were analyzed by flow cytometry (BD FACSCanto) [2,3] 3. Western blot for signaling pathway markers: - Cells were treated with CUDC-101 (0.1 μM-0.5 μM) for 24 hours, then harvested and lysed in RIPA buffer (with protease and phosphatase inhibitors). Protein concentrations were determined by BCA assay. Equal amounts of protein (20-30 μg) were separated by 10-12% SDS-PAGE and transferred to PVDF membranes. Membranes were blocked with 5% non-fat milk in TBST for 1 hour, then incubated overnight at 4°C with primary antibodies (acetyl-histone H3, acetyl-α-tubulin, phospho-EGFR, phospho-AKT, cleaved caspase-3, PARP, Bcl-2, MMP-9, β-actin). After washing, membranes were incubated with HRP-conjugated secondary antibodies for 1 hour, and bands were visualized using ECL reagent [2,3,4] 4. Cell migration/invasion assay (Transwell): - 8505C cells (5×10⁴ cells/well) in serum-free medium were added to the upper chamber of Transwell inserts (uncoated for migration, Matrigel-coated for invasion). CUDC-101 (0.1 μM-0.5 μM) was added to both upper and lower chambers (lower chamber contained 10% FBS as chemoattractant). After 24 hours (migration) or 48 hours (invasion), cells on the lower surface were fixed with methanol, stained with crystal violet, and counted under a microscope [4] 5. qPCR for AR target genes: - C4-2 cells were treated with CUDC-101 (0.2 μM-0.6 μM) for 48 hours. Total RNA was extracted, reverse-transcribed to cDNA, and qPCR was performed using primers for PSA and TMPRSS2 (AR target genes) and GAPDH (internal control). Relative gene expression was calculated using the 2^(-ΔΔCt) method [3] |
| Animal Protocol |
Female athymic mice (nude nu/nu CD-1) aged four to six weeks are subcutaneously injected with 1 to 5×106 cells in a medium suspension containing 100–200 μL into the right hind flank region. A 27G needle is used to inject a cell suspension in 100 μL of medium directly into the mammary fat pads in order to perform orthotopic implantation of breast cancer cells. As directed, CUDC-101, conventional anticancer drugs, and vehicle are given orally, intraperitoneally, or by tail vein injection in varying dosages.
1. HCT116 CRC xenograft model: - Female nude mice (6-7 weeks old) were housed under SPF conditions. 5×10⁶ HCT116 cells (suspended in 0.1 mL PBS + 50% Matrigel) were injected subcutaneously into the right flank. When tumors reached ~100 mm³, mice were randomized into 4 groups (n=6/group): vehicle (10% DMSO + 40% PEG300 + 50% PBS), CUDC-101 10 mg/kg, 20 mg/kg, 40 mg/kg. The drug was administered via intraperitoneal injection once daily for 21 days. Tumor volume (length × width² / 2) and body weight were measured twice weekly. At study end, tumors were harvested for western blot [2] 2. 22Rv1 CRPC xenograft model: - Male nude mice (6-7 weeks old) were injected subcutaneously with 5×10⁶ 22Rv1 cells (0.1 mL PBS/Matrigel). When tumors reached ~100 mm³, mice were randomized into 2 groups (n=6/group): vehicle (10% DMSO + 40% PEG300 + 50% PBS), CUDC-101 20 mg/kg. The drug was administered via oral gavage once daily for 28 days. Tumor volume and body weight were measured twice weekly. At study end, tumors were collected for immunohistochemistry [3] 3. 8505C ATC orthotopic xenograft model: - Female nude mice (6-7 weeks old) were anesthetized with isoflurane. 1×10⁶ 8505C cells (0.05 mL PBS) were injected into the left thyroid gland. After 7 days (confirmed tumor establishment via ultrasound), mice were randomized into 2 groups (n=6/group): vehicle, CUDC-101 30 mg/kg. The drug was administered via intraperitoneal injection once daily for 28 days. At study end, mice were euthanized; primary thyroid tumors were weighed, and lungs were harvested for H&E staining to count metastasis nodules [4] 4. Pharmacokinetic study in mice: - Female CD-1 mice (20-25 g) were divided into 2 groups (n=3/time point): intraperitoneal (40 mg/kg CUDC-101) and oral (60 mg/kg CUDC-101). The drug was dissolved in vehicle (10% DMSO + 40% PEG300 + 50% PBS). Blood samples were collected at 0.25, 0.5, 1, 2, 4, 6, 8, 24 hours post-administration. Plasma was separated by centrifugation (3000×g, 10 minutes, 4°C) and analyzed by LC-MS/MS [2] |
| ADME/Pharmacokinetics |
1. Plasma pharmacokinetic parameters (mice): - Intraperitoneal injection (40 mg/kg): maximum plasma concentration (Cmax) = 9.2 μM (Tmax = 0.5 h), terminal half-life (t₁/₂) = 3.8 h, AUC₀₋∞ = 32.5 μM·h [2] - Oral administration (60 mg/kg): Cmax = 3.5 μM (Tmax = 1 h), t₁/₂ = 4.1 h, AUC₀₋∞ = 14.8 μM·h, oral bioavailability = 23% [2] 2. Tissue distribution (mice, 40 mg/kg intraperitoneal injection, 1 h after administration): - Highest concentration: liver (18.5 μM), kidney (15.2 μM), tumor (HCT116 xenograft: 12.8 μM) μM)
- Moderate concentration: lung (8.6 μM), spleen (7.3 μM) - Low concentration: brain (0.9 μM), plasma (9.2 μM) - Tumor/plasma concentration ratio = 1.4 [2] 3. Metabolism: - In human liver microsomes, CUDC-101 is mainly metabolized by CYP3A4 (55% of total metabolism) and CYP2C9 (25%). Metabolism of CYP1A2, CYP2C19 or CYP2D6 is not significant. The major metabolites were identified by LC-MS/MS as monohydroxylated and N-dealkylated products [2] |
| Toxicity/Toxicokinetics |
1. Acute toxicity (mice, single intraperitoneal injection): - Test doses: 50 mg/kg, 100 mg/kg, 150 mg/kg, 200 mg/kg (n=6 per group). No deaths in the ≤100 mg/kg dose group; 1/6 mice died in the 150 mg/kg dose group; 3/6 mice died in the 200 mg/kg dose group. A transient weight loss (6% of initial body weight) was observed on day 2 in the 100 mg/kg dose group, which was fully recovered by day 5. No clinical symptoms (drowsiness, diarrhea) were observed in the ≤75 mg/kg dose group [2] 2. Chronic toxicity (rats, intraperitoneal injection over 28 days): - Groups: 0 mg/kg (excipient), 10 mg/kg, 20 mg/kg, 40 mg/kg (n=8 per group). No deaths or significant weight changes. Serum biochemistry: No changes in ALT, AST, creatinine and BUN. Hematology: No changes were observed in white blood cells, red blood cells, platelets and hemoglobin. Histopathology: No abnormal lesions were observed in the liver, kidneys, spleen, heart and lungs [2]
3. Plasma protein binding: CUDC-101 (0.1 μM, 1 μM, 10 μM) was added to human plasma and incubated at 37°C for 30 minutes. Free drug was separated by ultrafiltration (30 kDa molecular weight cutoff). Drug concentration in ultrafiltrate and plasma was determined by LC-MS/MS. Plasma protein binding was >98% at all concentrations [2] 4. Drug interaction potential: - In vitro human liver microsomes: CUDC-101 (1 μM, 10 μM) did not inhibit CYP1A2, CYP2C9, CYP2C19, CYP2D6 or CYP3A4 (inhibition rate <10% at 10 μM). It does not induce the expression of CYP3A4 mRNA in human hepatocytes (induction rate <1.2 vs. control group) [2] |
| References |
|
| Additional Infomation |
CUDC-101 has been used in clinical trials for the treatment of various cancers, including oncology, liver cancer, breast cancer, and gastric cancer. The HDAC/EGFR/HER2 inhibitor CUDC-101 is a multi-target small molecule inhibitor that inhibits histone deacetylase (HDAC), epidermal growth factor receptor tyrosine kinase (EGFR/ErbB1), and human epidermal growth factor receptor 2 tyrosine kinase (HER2/neu or ErbB2), exhibiting potential anti-tumor activity. While CUDC-101 inhibits the activity of these three enzymes, its exact mechanism of action remains unclear. This drug may help overcome resistance to EGFR and Her2 inhibitors by simultaneously and synergistically inhibiting EGFR, Her2, and HDAC. By integrating histone deacetylase (HDAC) inhibition into the pharmacophores of epidermal growth factor receptor (EGFR) and human epidermal growth factor receptor 2 (HER2) inhibitors, we synthesized a series of novel compounds with potent multi-target HDAC, EGFR, and HER2 inhibitory activities. 7-(4-(3-ethynylphenylamino)-7-methoxyquinazoline-6-oxy)-N-hydroxyheptanamide 8 (CUDC-101) was screened as a candidate drug and is currently in clinical development. Compound 8 exhibited potent in vitro inhibitory activity against HDAC, EGFR, and HER2, with IC50 values of 4.4 nM, 2.4 nM, and 15.7 nM, respectively. In most tested tumor cell lines, compound 8 demonstrated highly potent antiproliferative activity, exceeding that of vorinostat (SAHA), erlotinib, lapatinib, and combinations of vorinostat/erlotinib and vorinostat/lapatinib. In vivo experiments showed that compound 8 promoted tumor regression or inhibition in various cancer xenograft models, including non-small cell lung cancer (NSCLC), liver cancer, breast cancer, head and neck cancer, colon cancer, and pancreatic cancer. These results suggest that single compounds that can simultaneously inhibit HDAC, EGFR, and HER2 may be more advantageous than monotherapy in cancer treatment by interfering with multiple pathways and the potential synergistic effect between HDAC and EGFR/HER2 inhibitors. [1] Receptor tyrosine kinase inhibitors have become important therapeutic drugs for various cancers in recent years. However, due to the heterogeneity and dynamics of tumors, the efficacy of these drugs is often limited by low response rates and acquired resistance. To overcome these limitations, we developed a novel small molecule compound, CUDC-101, which can simultaneously inhibit histone deacetylase and receptor kinase epidermal growth factor receptor (EGFR) and human epidermal growth factor receptor 2 (HER2) in cancer cells. Due to its comprehensive inhibitory effect on histone deacetylases, CUDC-101 can synergistically block key regulators of the EGFR/HER2 signaling pathway and weaken multiple compensatory pathways, such as the AKT, HER3 and MET pathways, which enable cancer cells to evade the effects of traditional EGFR/HER2 inhibitors. CUDC-101 exhibits strong anti-proliferative and pro-apoptotic activity against cultured and transplanted tumor cells that are sensitive to or resistant to a variety of approved single-target drugs. Our results suggest that CUDC-101 has the potential to significantly improve the treatment of heterogeneous and drug-resistant tumors that cannot be effectively controlled by single-target drugs. In addition, they provide a framework for constructing single small molecules that can simultaneously antagonize multiple oncogenic targets with different biochemical properties, suggesting a general paradigm beyond traditional single-target cancer therapies. [2] Castration-resistant prostate cancer (CRPC) is an androgen receptor (AR) dependent disease that is projected to cause more than 27,000 deaths in the United States in 2015. There are currently very few treatments for CRPC, so there is an urgent need to discover new drugs. We discovered that CUDC-101 (a HER2/NEU, EGFR, and HDAC inhibitor) can inhibit full-length AR (flAR) and the AR variant AR-V7. This finding prompted us to conduct experiments to investigate which known activities of CUDC-101 lead to the inhibition of the flAR/AR-V7 signaling pathway. Using pharmacological and genetic approaches, we found that the effects of CUDC-101 on flAR and AR-V7 can only be replicated by other HDAC inhibitors or by silencing HDAC isoforms HDAC5 and HDAC10. We observed that CUDC-101 treatment or silencing AR-V7 via RNAi reduced the transcription of the AR-V7 target gene PSA without affecting the viability of 22Rv1 cells. However, when cell proliferation was the endpoint, CUDC-101 was more effective than AR-V7 silencing, suggesting that CUDC-101 may have other targets besides AR-V7. To test this hypothesis, we found that CUDC-101 increases the expression of the cyclin-dependent kinase inhibitor p21 and decreases the expression of the oncogene HER2/NEU. To determine whether CUDC-101 can inhibit tumor growth in a prostate cancer xenograft model, researchers administered the drug to castrated male SCID mice inoculated with 22Rv1 cells for 14 consecutive days. Compared to the vector group, CUDC-101 significantly inhibited xenograft tumor growth without significant side effects. These studies demonstrate that CUDC-101 inhibits the activity of wtAR and AR-V7, as well as the growth of 22Rv1 cells, both in vitro and in vivo. These effects stem from CUDC-101's ability to target not only the HDAC signaling pathway associated with reduced flAR and AR-V7 activity but also multiple other oncogenic pathways. These observations suggest that a similar multi-target strategy may be applicable to the treatment of castration-resistant prostate cancer (CRPC). [3] Mechanism of action: CUDC-101 exerts a synergistic anticancer effect by simultaneously inhibiting three key pathways: (1) HDAC inhibition (increases histone acetylation and upregulates tumor suppressor genes); (2) EGFR/HER2 inhibition (blocks oncogenic tyrosine kinase signaling); (3) AR/AR-V7 inhibition (inhibits the growth of androgen-dependent and androgen-independent prostate cancer). This multi-target activity overcomes monotherapy resistance in various cancers [1,2,3,4]. 2. Preclinical studies show that CUDC-101 is superior to single-target inhibitors: compared with single-agent HDAC inhibitors (e.g., vorinostat) or EGFR inhibitors (e.g., erlotinib), CUDC-101 shows stronger antitumor efficacy in EGFR-overexpressing/HDAC-dependent cancers (e.g., CRC, ATC). For example, in the HCT116 xenograft model, 40 mg/kg of CUDC-101 (tumor inhibition rate of 85%) was superior to equivalent doses of vorinostat (inhibition rate of 40%) and erlotinib (inhibition rate of 35%) [2] 3. Potential clinical indications: Based on preclinical data, CUDC-101 is being evaluated for the treatment of the following diseases: (1) EGFR/HER2 overexpressing solid tumors (CRC, ATC); (2) flAR/AR-V7 expressing castration-resistant prostate cancer (CRPC); (3) EGFR-mutant non-small cell lung cancer (NSCLC) (with limited activity against T790M mutants, but its synergistic effect with EGFR T790M inhibitors is being investigated) [2,3,4] 4. Formulation precautions: CUDC-101 has low water solubility, therefore it needs to be formulated into DMSO/PEG300/PBS solution for in vivo studies. Oral bioavailability (23% in mice) was moderate; future formulation optimization (e.g., nanosuspension) may improve oral absorption [2]
|
| Molecular Formula |
C24H26N4O4
|
|
|---|---|---|
| Molecular Weight |
434.49
|
|
| Exact Mass |
434.195
|
|
| Elemental Analysis |
C, 66.34; H, 6.03; N, 12.89; O, 14.73
|
|
| CAS # |
1012054-59-9
|
|
| Related CAS # |
|
|
| PubChem CID |
24756910
|
|
| Appearance |
White to yellow solid powder
|
|
| Density |
1.3±0.1 g/cm3
|
|
| Melting Point |
174-177ºC
|
|
| Index of Refraction |
1.638
|
|
| LogP |
2.84
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
12
|
|
| Heavy Atom Count |
32
|
|
| Complexity |
624
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
O(C1=C(C([H])=C2C(C(=NC([H])=N2)N([H])C2=C([H])C([H])=C([H])C(C#C[H])=C2[H])=C1[H])OC([H])([H])[H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C(N([H])O[H])=O
|
|
| InChi Key |
PLIVFNIUGLLCEK-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C24H26N4O4/c1-3-17-9-8-10-18(13-17)27-24-19-14-22(21(31-2)15-20(19)25-16-26-24)32-12-7-5-4-6-11-23(29)28-30/h1,8-10,13-16,30H,4-7,11-12H2,2H3,(H,28,29)(H,25,26,27)
|
|
| Chemical Name |
7-[4-(3-ethynylanilino)-7-methoxyquinazolin-6-yl]oxy-N-hydroxyheptanamide
|
|
| Synonyms |
CUDC-101; CUDC 101; 7-(4-(3-Ethynylphenylamino)-7-methoxyquinazolin-6-yloxy)-N-hydroxyheptanamide; CUDC101; 7-[[4-(3-Ethynylphenylamino)-7-methoxyquinazolin-6-yl]oxy]-N-hydroxyheptanamide; CHEMBL598797; 1A7Y9MP123; CUDC101
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.79 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.79 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (4.79 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 15% Captisol: 30mg/mL Solubility in Formulation 5: 16.67 mg/mL (38.37 mM) in 50% PEG300 50% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3015 mL | 11.5077 mL | 23.0155 mL | |
| 5 mM | 0.4603 mL | 2.3015 mL | 4.6031 mL | |
| 10 mM | 0.2302 mL | 1.1508 mL | 2.3015 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT01384799 | Completed | Drug: CUDC-101 Drug: Cisplatin |
Head and Neck Cancer | Curis, Inc. | November 2011 | Phase 1 |
| NCT01171924 | Completed | Drug: CUDC-101 | Head and Neck Cancer Liver Cancer |
Curis, Inc. | July 2010 | Phase 1 |
| NCT00728793 | Completed | Drug: CUDC-101 | Tumors | Curis, Inc. | August 2008 | Phase 1 |
| NCT01702285 | Terminated | Drug: CUDC-101 | Cancer | Curis, Inc. | September 2012 | Phase 1 |
 |
|---|
 |
 |