
| Size | Price | Stock | Qty |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g | |||
| Other Sizes |
Purity: ≥98%
Clemastine Fumarate (HS-592; HS 592 fumarate; Meclastine), the fumarate salt of clemastine, is a first-generation H1 histamine antagonist with anticholinergic and sedative adverse effects. It has an IC50 of 3 nM for H1 histamine receptor inhibition. Clemastine is used as the hydrogen fumarate in hay fever, rhinitis, allergic skin conditions, and pruritus. It also relieves sneezing, runny nose, and red, itchy, and tearing eyes.
| Targets |
mTOR; Histamine H1 receptor ( IC50 = 3 nM )
- Histamine H₁ receptor (antagonist activity, no IC₅₀/Ki provided)[1,3,12] - Human Ether-à-go-go-Related Gene (hERG) K⁺ channel (IC₅₀ = 12 nM for blocking IHERG current)[7] - P2X₇ receptor (allosteric sensitizer, enhances ATP-induced cation entry)[8] |
|---|---|
| ln Vitro |
In vitro activity: Clemastine Fumarate inhibits the increase in [Ca2+]i caused by histamine in HL-60 cells, with an IC50 of 3 nM, whereas the IC50 values of chlorpheniramine and diphenhydramine are 20 nM and 100 nM, respectively.[1]
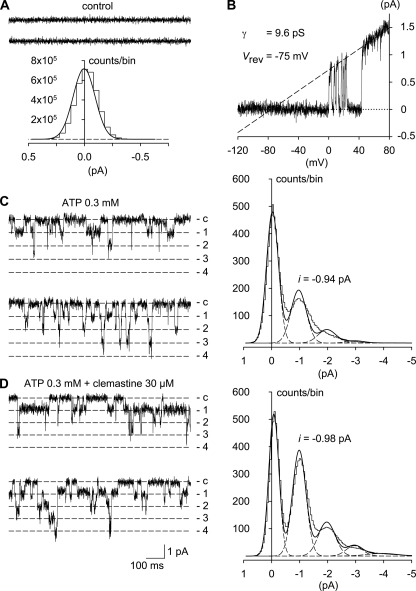
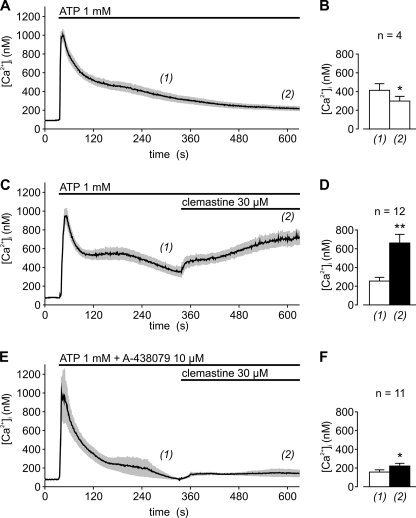
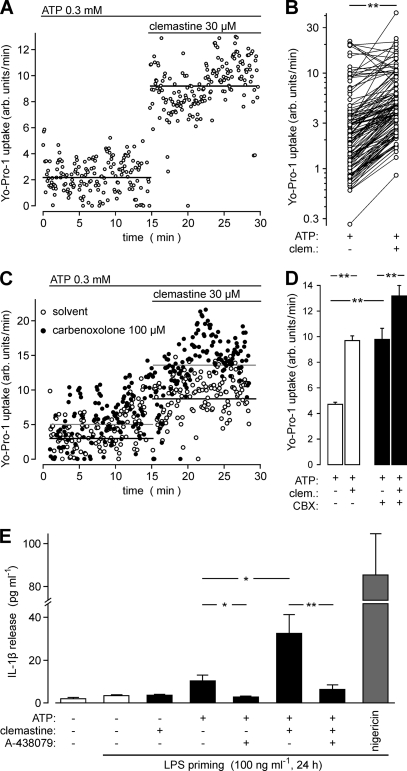
Clemastine fumarate dramatically suppresses lymphocyte NK and ADCC responses against human erythroleukemia cell line K562 and human B-lymphoblast cell line SB, respectively, at concentrations of ≥25 μM.[2] Clemastine Fumarate inhibits the contraction of the guinea pig ileum caused by histamine with an IC50 of 231 nM.[3] In HEK 293 cells that consistently express HERG channels, clemastine fumarate potently inhibits the HERG K+ channel in a concentration-dependent manner with an IC50 of 12 nM. This effect can be mitigated by the Y652A or F656A mutation of HERG.[4] Clemastine Fumarate augments the release of IL-1β from LPS-induced human macrophages and considerably amplifies the ATP-induced rise in [Ca2+]i in HEKhP2X7 cells, without depending on histamine receptor blockage but rather on sensitizing P2X7 receptor in a concentration-dependent manner with an EC50 of 10 μM.[5] - Antihistamine activity: Clemastine competitively blocks H₁ receptors on effector cells (e.g., vascular endothelium, airway smooth muscle), reducing histamine-mediated vasodilation and bronchoconstriction. This effect was confirmed in isolated guinea pig tracheal strips and rat mesenteric artery assays[3] - hERG channel inhibition: In HEK293 cells expressing hERG channels, clemastine (12 nM) significantly reduced peak IHERG current by ~50%, with voltage-dependent binding to the channel pore cavity. Mutations Y652A/F656A in the S6 helix attenuated this effect[7] - P2X₇ receptor modulation: In HEK293 cells stably expressing P2X₇ receptors, clemastine (1-10 μM) enhanced ATP-induced Ca²⁺ influx, accelerated pore dilation (Yo-Pro-1 uptake), and increased fractional permeability to NMDG⁺. Similar effects were observed in human monocyte-derived macrophages and murine bone marrow-derived macrophages[8] - Autophagy promotion: In LPS-stimulated H9c2 cardiomyocytes, clemastine (10-50 μM) increased LC3-II/LC3-I ratio, Beclin-1 expression, and autophagosome formation, which was abolished by the autophagy inhibitor 3-methyladenine. |
| ln Vivo |
Clemastine Fumarate treatment significantly lowers the innate immune responses of mice to Listeria monocytogenes by interfering with the production of proinflammatory cytokines like TNF-α and IL-6 by extracellular signal-regulated kinase (ERK). This effect is surprising because it is not dependent on blocking the histamine H1 receptor. The result is a significant increase in mortality.[7] Rats that are given Clemastine Fumarate (5–20 mg/kg) show a dose-dependent, significant inhibition of both zymosan paw oedema and croton oil ear oedema simultaneously. At 20 mg/kg, the inhibition is 53.6% and 46.8%, respectively, with ID50 values of 18.0 mg/kg and 20.5 mg/kg, respectively. [6]
Allergic rhinitis model: Oral clemastine (1 mg) administered 4-6 hours before allergen challenge significantly reduced sneezing frequency (p < 0.01) and rhinorrhea severity in 20 allergic subjects. The effect was dose-dependent and lasted ≥12 hours[6] - Sepsis-induced myocardial injury: In CLP-induced septic rats, intraperitoneal clemastine (30-50 mg/kg) improved 7-day survival rate (from 30% to 60%), reduced serum cTnI levels, preserved left ventricular ejection fraction, and attenuated mitochondrial fragmentation. Similar protective effects were observed in LPS-stimulated H9c2 cells - Optic neuritis model: Oral clemastine (1 mg twice daily for 90 days) in 16 patients with acute optic neuritis preserved retinal nerve fiber layer (RNFL) thickness in temporal/superotemporal quadrants and enhanced P100 wave amplitude recovery in visual evoked potentials compared to placebo. |
| Enzyme Assay |
In a buffer containing 138 mM NaCl, 6 mM KC1, 1 mM MgSO4, 1 mM Na2HPO4, 5 mM NaHCO33, 5.5 mM glucose, and 20 mM HEPES-NaOH, pH 7.4, HL-60 cells are suspended at a density of 1×107 cells/mL. The buffer is further enhanced with 0.1% (w/v) bovine serum albumin. Following a 10-minute incubation period at 37 °C, 4 μM of the dye fura-2/AM is added to the cells. After being diluted with the previously mentioned buffer to a concentration of 5×106 cells/mL, the cells are incubated at 37 °C for 45 minutes. Following this, cells are diluted using the previously mentioned buffer until they reach a final concentration of 0.5 × 106 cells/mL, and they are centrifuged at 250 g for 10 minutes at ambient temperature. In the previously mentioned buffer, cells are suspended at 1.0 × 106 cells/mL and stored at 20 °C until measurement. After loading with fura-2/AM, HL-60 cells are suspended in 2 mL of the previously mentioned buffer for a maximum of 4 hours using acryl fluorescence cuvettes. Prior to the addition of histamine (100 μM), HL-60 cells are incubated for 3 minutes at 37 °C with 1 mM Ca2+ and different concentrations of clemastine fumarate. With the cells constantly stirred at 1×103 rpm and at 37 °C, fluorescence is measured with a Ratio II spectrofluorometer. For one minute, the basal fluorescence, or basal [Ca2+]i, is measured. To find the increase in [Ca2+]i, the corresponding peak [Ca2+]i values are subtracted from the basal [Ca2+]i values. The wavelengths of excitation and emission are 500 nm and 340 nm, respectively. The competitive curve is used to calculate the IC50 value.
- hERG channel electrophysiology: Whole-cell patch-clamp recordings were performed on HEK293 cells expressing hERG channels. Cells were perfused with clemastine (1-100 nM) in Tyrode's solution at 37°C. IHERG tail currents were measured at -40 mV after depolarization to +20 mV. IC₅₀ was calculated using nonlinear regression[7] - P2X₇ receptor calcium flux assay: HEK293-P2X₇ cells were loaded with Fluo-4 AM and treated with clemastine (1-10 μM) followed by ATP (100 μM). Calcium transients were recorded using fluorescence microscopy. Yo-Pro-1 uptake was quantified by flow cytometry to assess pore formation[8] |
| Cell Assay |
- H₁ receptor antagonism assay: Guinea pig tracheal ring segments were incubated with clemastine (0.1-10 μM) and challenged with histamine (1 μM). Isometric tension changes were recorded to determine antagonist potency[3]
- Autophagy detection: H9c2 cardiomyocytes were treated with clemastine (10-50 μM) and LPS (1 μg/mL). LC3-II/LC3-I ratio and Beclin-1 expression were analyzed by Western blot. Autophagosomes were visualized by transmission electron microscopy. |
| Animal Protocol |
Male Wistar rats with paw oedema induced by subplantar injection of zymosan and ear oedema induced by croton oil
5-20 mg/kg Intraperitoneally - Sepsis model: Male Sprague-Dawley rats (250-300 g) underwent cecal ligation and puncture (CLP). Clemastine (10-50 mg/kg) was dissolved in 0.9% saline and administered intraperitoneally 30 minutes post-CLP. Survival was monitored for 7 days, and cardiac function was assessed by echocardiography on day 3 - Allergic rhinitis model: Human subjects received oral clemastine (1 mg) or placebo in a double-blind crossover design. Nasal allergen challenges were performed 1, 4, and 6 hours post-dose. Sneeze counts and nasal secretion weights were recorded[6] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Rapidly absorbed via the gastrointestinal tract. Primarily excreted in the urine. Metabolism/Metabolites Antichristins are primarily metabolized in the liver via monodemethylation, didemethylation, and glucuronidation. Absorption: Rapidly absorbed orally, with a bioavailability of approximately 40%. Peak plasma concentration (Cmax) is 1-2 ng/mL, reached within 2-4 hours. Distribution: Widely distributed in tissues (volume of distribution approximately 800 L), and can cross the blood-brain barrier. Plasma protein binding is approximately 95%. Metabolism: Extensively metabolized in the liver via O-dealkylation, oxidation, and glucuronidation. Major metabolites include demethylcrimetine and its hydroxylated derivatives. Excretion: Approximately 42% is excreted in the urine (primarily as metabolites), and 27% in the feces. The terminal half-life is approximately 21 hours. |
| Toxicity/Toxicokinetics |
Hepatotoxicity
Despite widespread use, first-generation antihistamines (such as chlorpheniramine) are rarely associated with abnormal liver function or clinically significant liver injury. Their safety may be related to their low daily dose and limited duration of use. Likelihood Score: E (Unlikely to cause clinically significant liver injury). References on the safety and potential hepatotoxicity of antihistamines are listed after the "Antihistamines Overview" section. Drug Category: Antihistamines Pregnancy and Lactation Effects ◉ Overview of Use During Lactation Occasional use of low doses of chlorpheniramine during lactation is acceptable. Higher doses or longer use may cause infant drowsiness and other adverse reactions, or reduce milk production, especially when used in combination with sympathomimetic drugs (such as pseudoephedrine) or before lactation is fully established. For many women, a single dose after the last feeding at bedtime may be sufficient and minimize any adverse effects of the drug. Non-sedating antihistamines are a better alternative. ◉ Effects on Breastfed Infants A 10-week-old breastfed infant born to a mother taking chlorpheniramine, phenytoin sodium, and carbamazepine experienced lethargy, refusal to feed, irritability, and loud crying. These side effects may be due to chlorpheniramine in breast milk, but other medications may also play a role. In a telephone follow-up study, mothers reported that 10% of infants exposed to various antihistamines experienced irritability and colic, and 1.6% experienced lethargy. None of these reactions required medical attention. ◉ Effects on Lactation and Breast Milk Injected relatively high doses of antihistamines can lower basal serum prolactin levels in non-lactating women and early postpartum women. However, pre-administration of antihistamines by postpartum mothers does not affect suckling-induced prolactin secretion. Whether lower oral doses of antihistamines have the same effect on serum prolactin, and whether these effects have any consequences for breastfeeding success, has not yet been studied. For mothers who have established lactation, prolactin levels may not affect their ability to breastfeed. Non-human toxicity values Oral LD50 in mice: 730 mg/kg Intravenous LD50 in mice: 43 mg/kg Oral LD50 in rats: 3550 mg/kg Intravenous LD50 in rats: 82 mg/kg. - Acute toxicity: LD₅₀ in mice >100 mg/kg (oral). Common adverse reactions include sedation, dry mouth, blurred vision, and urinary retention (caused by anticholinergic activity). - Cardiotoxicity: At supertherapeutic concentrations (≥1 μM), cristatin prolongs the QT interval in isolated cat hearts, but no clinical QT interval prolongation has been reported at therapeutic doses (1-6 mg/day) [7] - Drug interactions: Concomitant use with alcohol, opioids, or benzodiazepines may enhance central nervous system depression. Due to the risk of hypertensive crisis, it is contraindicated for use in combination with monoamine oxidase inhibitors. |
| References |
[1]. Mol Pharmacol . 1992 Aug;42(2):227-34. [2]. Cell Immunol . 1983 Oct 1;81(1):45-60. [3]. J Pharmacol Exp Ther . 1997 Jan;280(1):114-21. [4].J Mol Cell Cardiol . 2006 Jan;40(1):107-18. [5]. J Biol Chem . 2011 Apr 1;286(13):11067-81. [6]. J Pharmacol Exp Ther . 1997 Jan;280(1):114-21. |
| Additional Infomation |
Clemastine is 2-[(2R)-1-methylpyrrolidone-2-yl]ethanol, wherein the hydrogen on the hydroxyl group is replaced by 1-(4-chlorophenyl)-1-phenethyl (R configuration). It is a drug with antihistamine, antimuscarinic, and moderate sedative effects; its fumarate is used to relieve symptoms of allergic diseases such as rhinitis, urticaria, conjunctivitis, and pruritic dermatitis. It acts as an H1 receptor antagonist, antihistamine, muscarinic receptor antagonist, and antipruritic. It is an N-alkylpyrrolidine compound belonging to the monochlorobenzene class. It is an ethanolamine derivative and a first-generation histamine H1 receptor antagonist used to treat hay fever, rhinitis, allergic dermatitis, and pruritus. It can cause drowsiness. Clemastine is a first-generation antihistamine used to treat symptoms of allergic rhinitis and the common cold. Clemastine has not been found to be associated with clinically significant cases of acute liver injury.
Cemastine is a synthetic ethanolamine with anticholinergic, sedative, and histamine H1 receptor antagonistic effects. After administration, caemastine blocks H1 histamine receptors, thereby preventing symptoms caused by histamine acting on capillary, bronchial, and gastrointestinal smooth muscle, including vasodilation, increased capillary permeability, bronchoconstriction, and gastrointestinal smooth muscle spasms. Caemastine can also prevent histamine-induced mucosal pain and itching. A histamine H1 receptor antagonist, in the form of hydrogen fumarate, is used to treat hay fever, rhinitis, allergic dermatitis, and pruritus. It can cause drowsiness. See also: Caemastine fumarate (in salt form). Indications For relief of symptoms associated with allergic rhinitis, such as sneezing, runny nose, itching, and irritation. Also used to treat mild, uncomplicated allergic skin manifestations, such as urticaria and angioedema. Can be used as self-medication for common cold-related symptoms to provide temporary relief. Mechanism of Action Ceratine is a selective histamine H1 receptor antagonist that binds to histamine H1 receptors. This product blocks the action of endogenous histamine, thereby temporarily relieving histamine-induced adverse symptoms. Therapeutic Use For the treatment of allergic rhinitis symptoms in adults and children aged 12 years and older… For the treatment of mild allergic urticaria and angioedema symptoms in adults and children aged 12 years and older… This multicenter, double-blind, randomized parallel-group study compared the efficacy of outpatients with active perennial allergic rhinitis treated with loratadine (Claritin) 10 mg once daily, clematine (Dabigatran) 1 mg twice daily, and placebo for 3 weeks. A total of 155 patients were evaluated for efficacy and safety. On days 6, 13, and 20 of treatment, four nasal symptoms and three non-nasal symptoms, rhinoscopy results, and treatment response were scored. Patients were required to record daily symptoms and possible adverse reactions in a diary, noting when active rhinitis symptoms subsided. Based on assessments of nasal and ocular symptoms, diary scores, rhinoscopy results, and symptom relief time, loratadine and chlorpheniramine were significantly superior to placebo throughout the study period (p < 0.05). The loratadine group showed a significantly faster onset of symptom relief compared to the chlorpheniramine group (p < 0.05). Regarding nasal congestion, loratadine was significantly superior to chlorpheniramine after one week of treatment (p < 0.05). Adverse reaction reports showed a significantly higher proportion of patients experiencing sedation in the chlorpheniramine group than in the loratadine group (p < 0.05). There were no statistically significant differences among the three treatment groups in other adverse reactions and laboratory test results (p < 0.05). This study demonstrates that both loratadine and chlorpheniramine effectively relieve nasal and ocular symptoms in patients with perennial allergic rhinitis compared to placebo. Loratadine has a good safety profile and is well-tolerated, with significantly less sedative effect than loratadine; therefore, loratadine may have an advantage in the clinical treatment of perennial allergic rhinitis. A randomized, double-blind, placebo-controlled study compared the effects of sodium cromoglycate (sodium cromoglycate), loratadine, and ketotifen administered to the nasal mucosa of patients with seasonal and perennial allergic rhinitis 30 minutes before histamine and allergen provocation tests. Crimethalin and sodium cromoglycate (but not ketotifen) significantly inhibited the nasal response to progressively increasing concentrations of histamine. At the concentrations used, none of the drugs significantly inhibited the nasal response to allergens. Drug Warnings This drug is contraindicated during breastfeeding, including… loratadine. There are currently no adequate and well-controlled studies evaluating the safety of loratadine fumarate alone or in combination with phenylpropanolamine in pregnant women; therefore, this drug should only be used during pregnancy when clearly necessary. Because chlorpheniramine may cause serious adverse reactions in breastfeeding infants, the importance of the drug to the woman should be weighed when deciding whether to discontinue breastfeeding or discontinue the medication. The following drugs are associated with significant adverse reactions in some breastfeeding infants and should be used with caution in breastfeeding women: Chlorpheniramine: drowsiness, irritability, refusal to eat, high-pitched crying, neck stiffness (1 case). /Excerpt from Table 5/ |
| Molecular Formula |
C25H30CLNO5
|
|
|---|---|---|
| Molecular Weight |
459.96
|
|
| Exact Mass |
459.181
|
|
| Elemental Analysis |
C, 65.28; H, 6.57; Cl, 7.71; N, 3.05; O, 17.39
|
|
| CAS # |
14976-57-9
|
|
| Related CAS # |
Clemastine; 15686-51-8; Clemastine-d5 fumarate
|
|
| PubChem CID |
26987
|
|
| Appearance |
White to light yellow crystalline powder
|
|
| Density |
1.097 g/cm3
|
|
| Boiling Point |
116 °C / 24mmHg
|
|
| Melting Point |
61 °C
|
|
| Flash Point |
211ºC
|
|
| Vapour Pressure |
1.94E-07mmHg at 25°C
|
|
| Index of Refraction |
1.553
|
|
| LogP |
4.754
|
|
| Hydrogen Bond Donor Count |
0
|
|
| Hydrogen Bond Acceptor Count |
2
|
|
| Rotatable Bond Count |
6
|
|
| Heavy Atom Count |
24
|
|
| Complexity |
376
|
|
| Defined Atom Stereocenter Count |
2
|
|
| SMILES |
ClC1C([H])=C([H])C(=C([H])C=1[H])[C@@](C([H])([H])[H])(C1C([H])=C([H])C([H])=C([H])C=1[H])OC([H])([H])C([H])([H])[C@@]1([H])C([H])([H])C([H])([H])C([H])([H])N1C([H])([H])[H].O([H])C(/C(/[H])=C(\[H])/C(=O)O[H])=O
|
|
| InChi Key |
PMGQWSIVQFOFOQ-YKVZVUFRSA-N
|
|
| InChi Code |
InChI=1S/C21H26ClNO.C4H4O4/c1-21(17-7-4-3-5-8-17,18-10-12-19(22)13-11-18)24-16-14-20-9-6-15-23(20)2;5-3(6)1-2-4(7)8/h3-5,7-8,10-13,20H,6,9,14-16H2,1-2H3;1-2H,(H,5,6)(H,7,8)/b;2-1+/t20-,21-;/m1./s1
|
|
| Chemical Name |
(E)-but-2-enedioic acid;(2R)-2-[2-[(1R)-1-(4-chlorophenyl)-1-phenylethoxy]ethyl]-1-methylpyrrolidine
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.43 mg/mL (3.11 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 14.3 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.43 mg/mL (3.11 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 14.3 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 1.43 mg/mL (3.11 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 30% propylene glycol, 5% Tween 80, 65% D5W: 5mg/mL Solubility in Formulation 5: 1.43 mg/mL (3.11 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1741 mL | 10.8705 mL | 21.7410 mL | |
| 5 mM | 0.4348 mL | 2.1741 mL | 4.3482 mL | |
| 10 mM | 0.2174 mL | 1.0871 mL | 2.1741 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT06039137 | Active Recruiting |
Drug: Cetirizine | Solid Tumor | Erasmus Medical Center | June 1, 2022 | N/A |
| NCT03109288 | Recruiting | Drug: Pioglitazone Drug: clemastine fumarate Drug: Dantrolene Drug: Pirfenidone |
Multiple Sclerosis | National Institute of Allergy and Infectious Diseases (NIAID) |
August 11, 2017 | Phase 1 Phase 2 |
| NCT02521311 | Recruiting | Drug: Clemastine Drug: Placebo |
Optic Neuritis | University of California, San Francisco |
February 28, 2017 | Phase 2 |
| NCT05359653 | Recruiting | Drug: Clemastine Fumarate Drug: Placebo |
Multiple Sclerosis (MS) Multiple Sclerosis Relapse Multiple Sclerosis Benign |
University of California, San Francisco |
August 1, 2023 | Phase 1 Phase 2 |
| NCT06065670 | Not yet recruiting | Drug: Clemastine Fumarate Drug: Placebo |
Demyelinating Diseases Multiple Sclerosis Brain Lesion |
University of California, San Francisco |
November 1, 2023 | Phase 1 Phase 2 |
 |
|---|
 |
 |