
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 1g |
Purity: ≥98%
CAY10566 is a novel, orally bioavailable and selective SCD1 (stearoyl-CoA desaturase 1) inhibitor with IC50 values of 4.5 and 26 nM in mouse and human enzymatic assays, respectively. Cancer cell growth requires fatty acids to replicate cellular membranes. The kinase Akt is known to up-regulate fatty acid synthesis and desaturation, which is carried out by the oxygen-consuming enzyme stearoyl-CoA desaturase (SCD)1.
| Targets |
Stearoyl-CoA desaturase 1 (SCD1) (IC50 = 4 nM, determined by enzyme activity assay) [2]
- Stearoyl-CoA desaturase 2 (SCD2) (IC50 = 250 nM, determined by enzyme activity assay) [2] - Fatty acid desaturase 1 (FADS1), FADS2 (IC50 > 10000 nM, no significant inhibition) [2] |
|---|---|
| ln Vitro |
Swiss 3T3 cell proliferation is concentration-dependently inhibited by CAY10566 (0.0001-10 μM; 24 hours) [3].
Potently and selectively inhibited human SCD1 activity, with 62.5-fold selectivity over SCD2 and negligible activity against FADS1/FADS2 [2] - Reduced palmitoleic acid (16:1n-7) and oleic acid (18:1n-9) production (products of SCD1) in HepG2 cells: 100 nM CAY10566 decreased 16:1n-7 and 18:1n-9 levels by ~80% and ~75%, respectively, with increased saturated fatty acid (16:0, 18:0) accumulation [2] - Inhibited stearate-induced vascular smooth muscle cell (VSMC) calcification: 1 μM CAY10566 reduced calcium deposition by ~65% and downregulated calcification-related genes (Runx2, BMP2) by ~50% and ~45%, respectively [1] - Suppressed activating transcription factor 4 (ATF4) expression in stearate-treated VSMCs: 1 μM concentration reduced ATF4 protein levels by ~60%, blocking the ATF4-mediated calcification pathway [1] - Did not affect HepG2 or VSMC viability at concentrations up to 5 μM (MTT assay, cell survival rate > 90%) [1, 2] |
| ln Vivo |
Once the tumors became stable, the mice were given either vehicle or SCD1 complementation (2.5 mg/kg CAY10566, administered at least twice a day). SCD1 inhibition was more effective in suppressing Akt-driven tumors than Ras-driven tumors; mean tumor volumes on day 13 or 14 post-treatment were 0.5 ± 0.04 and 0.67 ± 0.05, respectively, compared with untreated tumors (P=0.01 Compared with Ras-Akt, passed two-tailed t test)[4].
|
| Enzyme Assay |
SCD1 enzyme activity assay: Recombinant human SCD1 protein was incubated with stearoyl-CoA (substrate), NADPH, and various concentrations of CAY10566 (0.1-1000 nM) in reaction buffer. After incubation at 37°C for 60 minutes, the reaction was stopped by adding hydrochloric acid-methanol. The product (oleoyl-CoA) was converted to free fatty acid and quantified by gas chromatography-mass spectrometry (GC-MS). IC50 was calculated based on the inhibition of product formation [2]
- SCD2/FADS selectivity assay: Recombinant human SCD2, FADS1, and FADS2 proteins were individually incubated with their respective acyl-CoA substrates, cofactors, and CAY10566 (0.1-10000 nM). Reaction products were detected by GC-MS, and IC50 values were determined to assess selectivity [2] |
| Cell Assay |
Cell Proliferation Assay[3]
Cell Types: Swiss 3T3 cells Tested Concentrations: 0.0001, 0.001, 0.01, 0.1, 1, 10 μM Incubation Duration: 24 hrs (hours) Experimental Results: Swiss 3T3 cell proliferation diminished in a concentration-dependent manner. HepG2 fatty acid composition assay: HepG2 cells were seeded in 6-well plates and treated with CAY10566 (0.1-100 nM) for 24 hours. Cells were harvested, and total lipids were extracted. Fatty acids were methylated and analyzed by GC-MS to quantify the levels of saturated (16:0, 18:0) and monounsaturated (16:1n-7, 18:1n-9) fatty acids [2] - VSMC calcification assay: VSMCs were isolated from rat aorta and seeded in 24-well plates. Cells were pre-treated with CAY10566 (0.1-1 μM) for 1 hour, then stimulated with stearate (200 μM) for 7 days. Culture medium was replaced every 2 days. Calcium deposition was measured by o-cresolphthalein complexone method. Runx2 and BMP2 mRNA levels were quantified by RT-PCR, and ATF4 protein expression was detected by western blot [1] |
| ADME/Pharmacokinetics |
The oral bioavailability in mice was approximately 30% (single dose of 10 mg/kg); the peak plasma concentration (Cmax) 1 hour after administration was 0.5 μg/mL [2]; the plasma half-life (t1/2) in mice was 3.2 hours; it was distributed in the liver and adipose tissue, with tissue/plasma concentration ratios of approximately 2.0 (liver) and 1.8 (adipose tissue) [2]
|
| Toxicity/Toxicokinetics |
In vitro cytotoxicity: CC50 > 5 μM in HepG2 cells and vascular smooth muscle cells (VSMCs); no significant cell death was observed at concentrations up to 5 μM [1, 2]
- Acute toxicity: Oral LD50 > 100 mg/kg in mice; no death or significant adverse reactions were observed at doses up to 100 mg/kg [2] - Plasma protein binding was approximately 90% (mice) [2] |
| References |
|
| Additional Infomation |
CAY10566 is a potent, selective, and orally bioavailable inhibitor of stearoyl-CoA desaturase 1 (SCD1). SCD1 is a key enzyme that catalyzes the conversion of saturated fatty acids into monounsaturated fatty acids [2]. Its core mechanism involves inhibiting SCD1-mediated fatty acid desaturation, thereby altering cellular fatty acid composition and blocking downstream signaling pathways involved in pathological processes (e.g., ATF4) [1, 2]. Based on its ability to regulate fatty acid metabolism and inhibit the expression of calcification-related genes, potential therapeutic applications include metabolic disorders (dyslipidemia, obesity) and vascular calcification [1, 2]. Compared to other fatty acid desaturases, CAY10566 exhibits high selectivity for SCD1, thereby minimizing off-target effects on fatty acid metabolism [2].
|
| Molecular Formula |
C18H17CLFN5O2
|
|---|---|
| Molecular Weight |
389.8113
|
| Exact Mass |
389.105
|
| Elemental Analysis |
C, 55.46; H, 4.40; Cl, 9.09; F, 4.87; N, 17.97; O, 8.21
|
| CAS # |
944808-88-2
|
| PubChem CID |
16732433
|
| Appearance |
Off-white to yellow solid powder
|
| Density |
1.4±0.1 g/cm3
|
| Boiling Point |
600.2±65.0 °C at 760 mmHg
|
| Flash Point |
316.8±34.3 °C
|
| Vapour Pressure |
0.0±1.7 mmHg at 25°C
|
| Index of Refraction |
1.592
|
| LogP |
1.88
|
| Hydrogen Bond Donor Count |
0
|
| Hydrogen Bond Acceptor Count |
8
|
| Rotatable Bond Count |
4
|
| Heavy Atom Count |
27
|
| Complexity |
485
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
FC1C=C(OC2CCN(C3C=CC(C4OC(C)=NN=4)=NN=3)CC2)C(Cl)=CC=1
|
| InChi Key |
WFOFPVXMPTVOTJ-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C18H17ClFN5O2/c1-11-21-24-18(26-11)15-4-5-17(23-22-15)25-8-6-13(7-9-25)27-16-10-12(20)2-3-14(16)19/h2-5,10,13H,6-9H2,1H3
|
| Chemical Name |
3-[4-(2-chloro-5-fluorophenoxy)-1-piperidinyl]-6-(5-methyl-1,3,4-oxadiazol-2-yl)-pyridazine
|
| Synonyms |
CAY10566; CAY-10566; CAY 10566
|
| HS Tariff Code |
2934.99.03.00
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO : ~25 mg/mL (~64.13 mM)
|
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.41 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.41 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (5.34 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5654 mL | 12.8268 mL | 25.6535 mL | |
| 5 mM | 0.5131 mL | 2.5654 mL | 5.1307 mL | |
| 10 mM | 0.2565 mL | 1.2827 mL | 2.5654 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
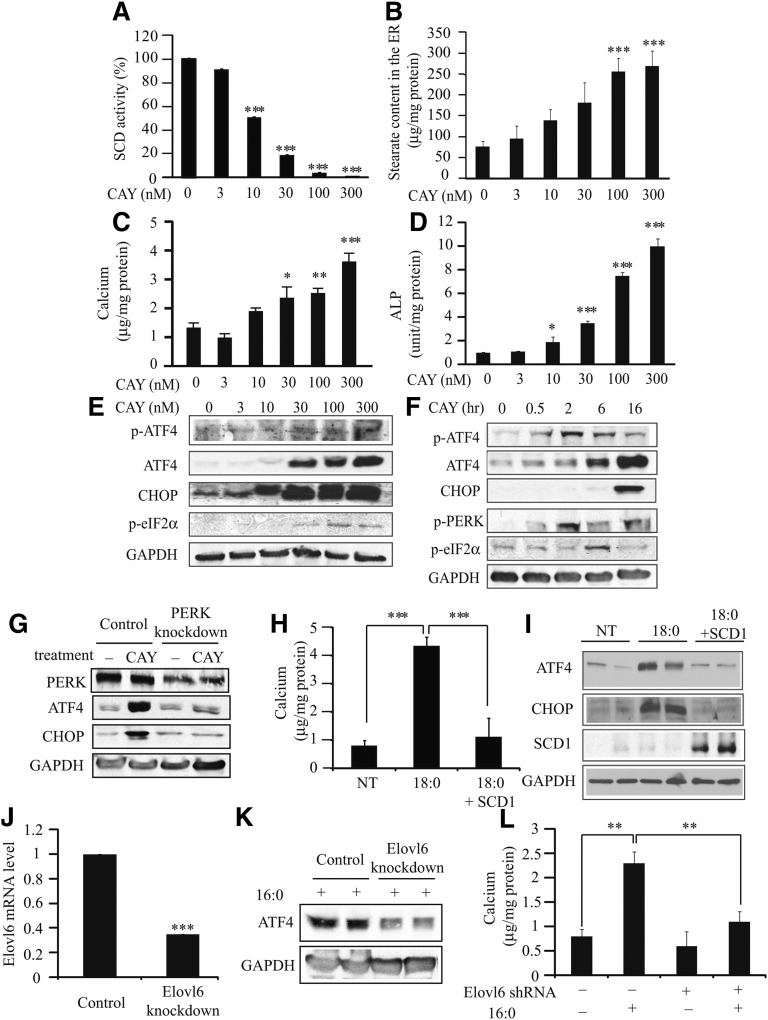 SCD activity and ER stearate levels regulate ATF4 expression through PERK-eIF2α pathway, mineralization, and osteoblastic differentiation. |
|---|
CD inhibition induces the expression of ER stress and osteogenic markers.J Lipid Res.2012 Aug;53(8):1543-52. |
ATF4 expression alters mineralization and osteoblastic differentiation of VSMCs induced by stearate and SCD inhibitor CAY10566.J Lipid Res.2012 Aug;53(8):15 |