
| Size | Price | Stock | Qty |
|---|---|---|---|
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| Other Sizes |
Purity: ≥98%
Bicalutamide (formerly known as ICI-176334; CDX; trade names: Casodex in the USA and Cosudex in other countries), a marketed drug for treating PC (prostate cancer), is a potent, synthetic, nonsteroidal androgen receptor (AR) antagonist with potential anticancer activity. It inhibits AR with an IC50 of 0.16 μM in LNCaP/AR(cs)cell line. Bicalutamide inhibits the growth of prostate cancer cells which overexpress androgen receptor by directly binding to AR then mediates androgen-mediated gene transcription. Bicalutamide directly binds to androgen receptor with Ki value of 12.5μM. In prostate cancer cells, bicalutamide impairs DNA binding and nuclear localization. Bicalutamide and MDV3100 significantly inhibited R1881-induced VP16-AR–mediated transcription in HepG2 cells with an IC50 value of 0.2 μM.
| Targets |
AR/androgen receptor
Androgen Receptor (AR): Bicalutamide binds to human AR as a competitive antagonist, with a Ki value of 1.9 nM; this affinity is ~13-fold lower than that of ARN-509 (Ki=0.14 nM, used as reference in [1]) [1] |
|---|---|
| ln Vitro |
Bicalutamide has an IC50 of 160 nM and competes with androgens for binding to AR in a whole-cell binding experiment (LNCaP/AR (cs) cells) [1]. Bicalutamide partially counteracts the effects of R1881, a synthetic androgen, while inducing VCaP cell proliferation in a dose-dependent manner [1].
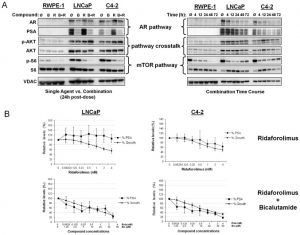
1. Antiproliferative Activity in Prostate Cancer Cells ([1]): Bicalutamide (1–50 μM) was tested as a control for ARN-509 in LNCaP (androgen-dependent) and C4-2 (castration-resistant prostate cancer, CRPC) cells. After 72-hour treatment: - LNCaP cells: Bicalutamide inhibited proliferation with an IC50 of 12.5 μM (MTT assay), while ARN-509 had an IC50 of 0.5 μM. - C4-2 cells (ARN-resistant): Bicalutamide showed weak activity, with only 30% proliferation inhibition at 50 μM, compared to 80% inhibition by ARN-509 (10 μM). At 20 μM, Bicalutamide downregulated AR target gene PSA (prostate-specific antigen) mRNA by 40% (real-time PCR) in LNCaP cells, but only by 15% in C4-2 cells [1] 2. AR Nuclear Translocation Inhibition ([1]): Immunofluorescence staining of LNCaP cells showed Bicalutamide (20 μM) reduced nuclear AR accumulation by 55% vs. vehicle control. In contrast, ARN-509 (5 μM) reduced nuclear AR by 90%. No significant inhibition of nuclear AR was observed in C4-2 cells treated with Bicalutamide (50 μM) [1] |
| ln Vivo |
In a mouse model of prostate cancer, belicutamide (10 mg/kg; ig; daily; for 28 days) shows anticancer efficacy [1].
Antitumor Efficacy in CRPC Xenografts (as Control) ([1]): Male nude mice (6–8 weeks old) were subcutaneously inoculated with 5×10⁶ C4-2 cells. When tumors reached 100 mm³, mice were orally administered Bicalutamide (50 mg/kg/day) or ARN-509 (10 mg/kg/day) for 28 days: - Bicalutamide group: Tumor volume was reduced by 25% vs. vehicle control; no significant change in tumor weight was observed. - ARN-509 group: Tumor volume and weight were reduced by 75% and 70%, respectively. Mice in the Bicalutamide group showed no weight loss, but serum PSA levels (AR activity marker) only decreased by 30%, compared to 85% reduction in the ARN-509 group [1] |
| Enzyme Assay |
Ligand-binding studies were performed either in a whole-cell assay (LNCaP/AR(cs)), using whole-cell extracts (MDA-MB-453), or in vitro with purified receptor. Proliferation assays (VCaP) were performed in either agonist- or antagonist-mode (without/with R1881). RNA was isolated from LNCaP/AR cells for RT-PCR analysis with primers (Suppl. Table 1) specific for AR target-genes. Fluorescence microscopy was performed in LNCaP cells transfected with AR-EYFP as previously described. AR antibody PG-21 was used for chromatin immunoprecipitation (ChIP) experiments (LNCaP/AR(cs)) together with primers for PSA and TMPRSS2 enhancers. Luciferase reporter-gene assays were performed in LNCaP/AR-luc or Hep-G2 cells expressing VP16-AR[1].
Ligand binding studies [1] Whole cell LNCaP/AR: Whole-cell competitive binding assays were performed in LNCaP/AR(codon-switch) (LNCaP/AR(cs)) (harbors a mixture of exogenous wild-type AR and endogenous mutant AR (T877A)) and cells propagated in Iscove’s or RPMI media supplemented with 10% fetal bovine serum (FBS), or during the assay with 10% charcoal-stripped, dextran-treated fetal bovine serum (CSS). Cells were pre-incubated with 18F-FDHT, increasing concentrations (1pM to 1μM) of cold competitor were added, and the assay was performed according to published procedures to measure specific uptake of 18F-FDHT. AR Competitive Binding Assay ([1]): 1. Recombinant AR Preparation: Human AR ligand-binding domain (LBD) was expressed in Sf9 insect cells and purified via nickel-chelate chromatography. 2. Reaction System: 200 μL mixture contained 50 mM Tris-HCl (pH 7.4), 10% glycerol, 0.5 nM [³H]-dihydrotestosterone (DHT, AR agonist), 100 ng AR-LBD, and Bicalutamide (0.1–50 nM, cold competitor). 3. Incubation & Separation: Incubated at 4°C for 2 hours; unbound [³H]-DHT was removed by adding dextran-coated charcoal (1% charcoal, 0.1% dextran) and centrifuging at 3,000×g for 10 minutes. 4. Detection & Calculation: Radioactivity of the supernatant was measured via liquid scintillation counter; the Ki value of Bicalutamide (1.9 nM) was calculated using the Cheng-Prusoff equation, with ARN-509 (Ki=0.14 nM) as a reference [1] |
| Cell Assay |
Proliferation assays[1]
Trypsinized VCaP cells were adjusted to a concentration of 100,000 cells per mL in phenol-red-free RPMI 1640 (with 5% CSS), and dispensed in 16 µL aliquots into CellBIND 384 well plates. Cells were incubated for 48 hours, after which ligand was added in a 16 µL volume to the RPMI culture medium. 1. Prostate Cancer Cell Proliferation Assay ([1]): - Cell Culture: LNCaP and C4-2 cells were cultured in RPMI 1640 medium (10% fetal bovine serum) and seeded in 96-well plates (5×10³ cells/well). - Drug Treatment: Cells were treated with Bicalutamide (1–50 μM) or ARN-509 (0.1–10 μM) for 72 hours; vehicle control (0.1% DMSO) was included. - Detection: MTT reagent (10 μL/well) was added for the final 4 hours of incubation; absorbance was measured at 570 nm, and IC50 values were calculated using GraphPad Prism software [1] 2. AR Target Gene and Nuclear Translocation Assays ([1]): - Gene Expression: LNCaP/C4-2 cells (2×10⁵ cells/6-well plate) were treated with Bicalutamide (20–50 μM) for 24 hours; total RNA was extracted, and PSA mRNA levels were measured via real-time PCR (GAPDH as internal control). - Nuclear AR: Cells were fixed with 4% paraformaldehyde, stained with anti-AR primary antibody and Alexa Fluor 488-conjugated secondary antibody; nuclear fluorescence intensity was quantified using ImageJ software [1] |
| Animal Protocol |
Animal/Disease Models: Castrate male mice, bearing LNCaP/AR(cs) xenograft tumors[1]
Doses: 10 mg/kg Route of Administration: po (oral gavage), daily, for 28 days Experimental Results: Inhibited tumor growth. C4-2 CRPC Xenograft Protocol ([1]): 1. Animal Selection: 6–8 weeks old male BALB/c nude mice (n=6/group) were randomized to vehicle control, Bicalutamide (50 mg/kg), and ARN-509 (10 mg/kg) groups. 2. Model Induction: 5×10⁶ C4-2 cells were suspended in 0.2 mL PBS + 50% Matrigel and subcutaneously injected into the right flank of mice. 3. Drug Preparation: Bicalutamide was suspended in 0.5% carboxymethylcellulose (CMC) + 0.1% Tween 80 to a concentration of 5 mg/mL (to deliver 50 mg/kg via 10 mL/kg oral gavage). 4. Administration: Oral gavage once daily for 28 days; vehicle control received 0.5% CMC + 0.1% Tween 80. 5. Detection: Tumor volume was measured twice weekly (volume = length × width² / 2); mice were euthanized on day 28, and serum PSA levels were measured via enzyme-linked immunosorbent assay (ELISA) [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Bicalutamide is well absorbed after oral administration, but its absolute bioavailability is unclear. Apparent oral clearance (cl) = 0.32 L/h [Normal male] Bicalutamide is well absorbed after oral administration, but its absolute bioavailability is unclear. Co-administration of bicalutamide with food has no clinically significant effect on the rate or extent of absorption. Bicalutamide has a high protein binding rate (96%). …Bicalutamide metabolites are excreted in almost equal amounts in urine and feces, with little or no excretion of the parent drug in urine; conversely, the parent drug is predominantly excreted in plasma. Fecal bicalutamide is thought to originate from the hydrolysis of bicalutamide glucuronide and unabsorbed drug. … …15 healthy male volunteers participated in a three-cycle, three-treatment, randomized crossover trial with a 9-week washout period, receiving a single oral dose of bicalutamide (50 mg) after meals and on an empty stomach. After fasting, the plasma concentration of (R)-bicalutamide was significantly higher than that of (S)-bicalutamide; the mean Cmax of the (R)-enantiomer (734 ng mL⁻¹) was approximately nine times that of the (S)-enantiomer (84 ng mL⁻¹). The corresponding tmax values for (R)-bicalutamide and (S)-bicalutamide were 19 hours and 3 hours, respectively. Elimination of (R)-bicalutamide from plasma exhibited a single exponential decay and was slow (t¹/² = 5.8 days). Elimination of (S)-bicalutamide was biphasic in some volunteers, while it was monophasic in others (terminal t¹/² = 1.2 days; n = 11). Food had no significant effect on the AUC, tmax, or t¹/² data for either enantiomer. When taken with food, the Cmax values of (R)-bicalutamide (14%) and (S)-bicalutamide (19%) increased slightly and statistically significantly. ... For more complete data on the absorption, distribution, and excretion of bicalutamides (9 in total), please visit the HSDB record page. Metabolism/Metabolites Bicalutamide undergoes stereoselective metabolism. The S (inactive) isomer is primarily metabolized via glucuronidation. The R (active) isomer also undergoes glucuronidation, but is primarily oxidized to the inactive metabolite before glucuronidation. Bicalutamide undergoes stereoselective metabolism. The S (inactive) isomer is primarily metabolized via glucuronidation. The R (active) isomer also undergoes glucuronidation, but is primarily oxidized to the inactive metabolite before glucuronidation. Both the parent drug and the glucuronides of the metabolites are excreted in urine and feces. The S-enantiomer is cleared faster than the R-enantiomer, which accounts for approximately 99% of the total steady-state plasma concentration. Bicalutamide undergoes stereoselective metabolism. The S (inactive) isomer is primarily metabolized via glucuronidation. The R (active) isomer also undergoes glucuronidation, but is mainly oxidized to the inactive metabolite, followed by further glucuronidation. Half-life: 5.9 days. Biological half-life 5.9 days... In some volunteers, elimination of (S)-bicalutamide was biphasic, while in others it was monophasic (terminal t1/2 = 1.2 days; n = 11). ... The apparent plasma elimination half-life observed after repeated dosing was 8.4 +/- 1.1 days. Oral absorption: Bicalutamide has an oral bioavailability of approximately 70% in humans (cited in [1] as a reference for the comparison of ARN-509); peak plasma concentration (Cmax) of 1.2 μg/mL is reached 2–3 hours after oral administration of 50 mg [1] - Metabolism: Primarily metabolized in the liver by CYP3A4 to the active metabolite (hydroxybicalutamide), which has approximately twice the affinity for the androgen receptor (AR) of the parent drug [1] - Plasma half-life: The elimination half-life of bicalutamide in humans is approximately 6 days, and the half-life of its active metabolite is approximately 7 days [1] - Plasma protein binding: The binding rate to human plasma albumin is >99% [1] |
| Toxicity/Toxicokinetics |
Toxicity Summary
Bicalutamide competes with androgens for androgen receptors, thereby blocking the effects of androgens from the adrenal glands and testes, which stimulate the growth of both normal and malignant prostate tissue. Organic nitrile compounds can decompose into cyanide ions both in vivo and in vitro. Therefore, the primary mechanism of toxicity for organic nitrile compounds is the production of toxic cyanide ions, or hydrogen cyanide. Cyanide ions are inhibitors of cytochrome c oxidase in the fourth electron transport chain complex (located on the mitochondrial membrane of eukaryotic cells). It forms a complex with the ferric atom in this enzyme. The binding of cyanide ions to this cytochrome prevents electrons from being transferred from cytochrome c oxidase to oxygen. As a result, the electron transport chain is disrupted, and the cell can no longer perform aerobic ATP energy production. Tissues that primarily rely on aerobic respiration, such as the central nervous system and the heart, are particularly vulnerable. Cyanide can also exert some toxic effects by binding to catalase, glutathione peroxidase, methemoglobin, hydrocobalamin, phosphatase, tyrosinase, ascorbic acid oxidase, xanthine oxidase, succinate dehydrogenase, and copper/zinc superoxide dismutase. Cyanide binds to the iron ions in methemoglobin to form inactive cyanogenic methemoglobin. (L97) Hepatotoxicity Bicalutamide treatment is associated with mild, asymptomatic, and transient increases in serum transaminase levels in approximately 6% of patients. Compared to flutamide, bicalutamide appears to cause ALT elevations less frequently and to a lesser extent. Similarly, there are a few case reports of clinically significant liver injury caused by bicalutamide, but the incidence is lower than that of flutamide. In a pharmacovigilance study in Spain, 11 cases of bicalutamide hepatotoxicity were reported, but none resulted in death. On the other hand, the bicalutamide product information mentions a few cases of fatal liver failure. The clinical presentation of liver injury caused by bicalutamide appears similar to that of flutamide. The latency period is typically 2 to 3 months, but may shorten with re-administration, and occasionally occurs 4 to 6 months after initiation. Elevated serum enzymes are typically hepatocellular, and severe fulminant cases have been reported. Rash, fever, and eosinophilia are uncommon, and autoantibody formation has not been reported. Probability score: B (likely the cause of clinically significant liver injury). Protein binding: 96% protein binding studies indicate that bicalutamide can displace coumarin anticoagulants from their binding sites. Prothrombin time should be closely monitored in patients already taking coumarin anticoagulants who begin bicalutamide treatment. In vitro studies show that R-bicalutamide is an inhibitor of CYP 3A4, with weaker inhibitory activity against CYP 2C9, 2C19, and 2D6. Clinical studies have shown that when used in combination with midazolam (a CYP 3A4 substrate), the mean plasma concentration (Cmax) of midazolam may increase by 1.5 times and the AUC may increase by 1.9 times. Therefore, caution should be exercised when using bicalutamide in combination with CYP 3A4 substrate. 1. In vitro toxicity ([1]): Bicalutamide (1–50 μM) has no cytotoxicity on normal human prostate epithelial cells (RWPE-1), with cell viability >90% (MTT method), compared with the solvent control group [1] 2. In vivo toxicity ([1]): After mice were treated with bicalutamide (50 mg/kg/day, 28 days), there were no significant changes in liver function (ALT/AST) or kidney function (BUN/creatinine) compared with the solvent control group. No histopathological abnormalities were observed in the liver and kidney tissues [1] 3. Clinical toxicity (cited from [1]): Common side effects of CRPC patients taking bicalutamide (50 mg/day) include hot flashes (45%), breast tenderness (30%), and mild elevation of liver enzymes (15%); serious hepatotoxicity is rare (<2%) [1] |
| References | |
| Additional Infomation |
Therapeutic Uses
Androgen antagonist; antitumor drug. Bicalutamide 50 mg/day is indicated for use in combination with a luteinizing hormone-releasing hormone (LHRH) analogue for the treatment of stage D2 metastatic prostate cancer. /US product label contains/ Bicalutamide 150 mg/day is not approved for use alone or in combination with other therapies. /US product label contains/ Drug Warnings Bicalutamide is contraindicated in patients with a known hypersensitivity to any component of this drug or tablet. Bicalutamide is not indicated for use in women, especially for non-serious or non-life-threatening conditions. FDA Pregnancy Risk Category: X / Contraindicated during pregnancy. Animal and/or human studies, or investigational reports or post-marketing reports, have demonstrated a risk of fetal malformation or other adverse events that significantly outweigh any potential benefit to the patient. / It is currently unknown whether bicalutamide is excreted into human breast milk. Because many drugs are excreted into human breast milk, caution should be exercised when breastfeeding women take bicalutamide. For more complete data on drug warnings for bicalutamide (10 in total), please visit the HSDB record page. Pharmacodynamics Bicalutamide is an anti-tumor hormone primarily used to treat prostate cancer. Bicalutamide is a pure nonsteroidal anti-androgen with an affinity for androgen receptors (but no affinity for progesterone receptors, estrogen receptors, or glucocorticoid receptors). Therefore, bicalutamide blocks the effects of adrenal and testicular androgens that stimulate the growth of normal and malignant prostate tissue. Most prostate cancers are androgen-dependent and can be treated with surgical or medical castration. To date, anti-androgen monotherapy has not been shown to be as effective as castration therapy. 1. Drug Background ([1]): Bicalutamide is a first-generation nonsteroidal anti-androgen (NSAA) approved for the treatment of advanced prostate cancer, usually in combination with castration therapy. It functions as a competitive androgen receptor antagonist, but its efficacy is limited due to castration-resistant prostate cancer caused by androgen receptor mutations or overexpression[1]. 2. Mechanism of action([1]): Bicalutamide binds to the androgen receptor ligand-binding domain (LBD), competes with endogenous androgens (e.g., DHT) to inhibit androgen receptor nuclear translocation, and subsequently binds to androgen response elements (AREs) in target genes (e.g., PSA). However, compared with second-generation nonsteroidal anti-inflammatory drugs (NSAAs) such as ARN-509, bicalutamide has a lower affinity for androgen receptors (AR) and is less active in castration-resistant prostate cancer (CRPC) cells[1]. 3. Treatment limitations ([1]): Bicalutamide is often poorly effective in CRPC patients due to the presence of AR mutants (e.g., T877A), which convert bicalutamide from an antagonist to a partial agonist, while ARN-509 retains its antagonistic activity against these mutants [1]. |
| Molecular Formula |
C18H14F4N2O4S
|
|---|---|
| Molecular Weight |
430.37
|
| Exact Mass |
430.061
|
| Elemental Analysis |
C, 50.23; H, 3.28; F, 17.66; N, 6.51; O, 14.87; S, 7.45
|
| CAS # |
90357-06-5
|
| Related CAS # |
(R)-Bicalutamide;113299-40-4;Bicalutamide-d4;1185035-71-5
|
| PubChem CID |
2375
|
| Appearance |
White to off-white solid powder
|
| Density |
1.5±0.1 g/cm3
|
| Boiling Point |
650.3±55.0 °C at 760 mmHg
|
| Melting Point |
191-193°C
|
| Flash Point |
347.1±31.5 °C
|
| Vapour Pressure |
0.0±2.0 mmHg at 25°C
|
| Index of Refraction |
1.578
|
| LogP |
4.94
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
9
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
29
|
| Complexity |
750
|
| Defined Atom Stereocenter Count |
0
|
| InChi Key |
LKJPYSCBVHEWIU-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C18H14F4N2O4S/c1-17(26,10-29(27,28)14-6-3-12(19)4-7-14)16(25)24-13-5-2-11(9-23)15(8-13)18(20,21)22/h2-8,26H,10H2,1H3,(H,24,25)
|
| Chemical Name |
N-(4-cyano-3-(trifluoromethyl)phenyl)-3-((4-fluorophenyl)sulfonyl)-2-hydroxy-2-methylpropanamide
|
| Synonyms |
ICI-176334; ICI 176334; ICI176334; CDX. US trade name: Casodex; Cosudex. Calutide;
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.81 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.81 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (5.81 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3236 mL | 11.6179 mL | 23.2358 mL | |
| 5 mM | 0.4647 mL | 2.3236 mL | 4.6472 mL | |
| 10 mM | 0.2324 mL | 1.1618 mL | 2.3236 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
 |
|---|
|
|