
| Size | Price | Stock | Qty |
|---|---|---|---|
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
Bepotastine besilate (bepotastine besylate; TAU-284; TAU284; Bepreve; Talion), the besylate salt of Bepotastine, is a 2nd generation, non-sedating, and selective antagonist of histamine 1 (H1) receptor with a pIC50 of 5.7. Bepostatine besilate has been given approval to treat pruritus, urticaria, and allergic rhinitis. By blocking histamine H1 receptors, antagonistically opposing the vasoconstrictor and, to a lesser degree, the vasodilator effects of histamine, bepotastine besilate inhibits the action of histamine. Mast cell stabilisers work by stopping the regular flow of intracellular signals, which inhibits degranulation and the subsequent release of histamine. Talion is the brand name for bepostatine besilate.
| Targets |
Histamine H1 receptor ( pIC50 = 5.7 )
Histamine H1 receptor (H1R) (human H1R, Ki=0.13 nM; rat H1R, Ki=0.16 nM; guinea pig H1R, Ki=0.21 nM) [4] Histamine H2/H3/H4 receptors, muscarinic receptors, adrenergic receptors (Ki>1000 nM, negligible affinity) [4] |
|---|---|
| ln Vitro |
In vitro activity: Bepotastine besilate (10, 100, 1000 µM; preincubates for 120 min) reaches a statistically significant reduction in the amount of histamine released as a result of treatment with A23187 at 1000 µM[1].
Bepotastine besilate (50 µM; 1 h) inhibits NGF mRNA expression in NHEKs[2]. Human umbilical vein endothelial cells (HUVECs) were stimulated with histamine (10 μM) to induce vascular hyperpermeability. Bepotastine Besilate (Bepreve) (0.1 nM-1 μM) dose-dependently inhibited this effect, with 82% inhibition at 1 μM, via H1R antagonism [1] - Isolated human eosinophils were activated with eotaxin (10 ng/mL). Treatment with Bepotastine Besilate (Bepreve) (1 nM-100 nM) suppressed eosinophil chemotaxis by 65% at 100 nM and reduced release of pro-inflammatory cytokines (IL-5, IL-8) by 48% and 53% respectively [1] - Normal human epidermal keratinocytes (NHEKs) were cultured and treated with Bepotastine Besilate (Bepreve) (1 μM-50 μM) for 24 hours. The drug downregulated mRNA and protein expression of nerve elongation factors (NEF-H, NEF-M) by 35-42% at 50 μM, without affecting cell viability [2] - Radioligand binding assay confirmed Bepotastine Besilate (Bepreve) as a highly selective H1R antagonist, with no significant binding to other histamine receptor subtypes or neurotransmitter receptors [4] |
| ln Vivo |
Bepotastine besilate (10 g/L; eye drop; 3 times at intervals of 20 min in one eye) significantly inhibits the infiltration of conjunctival eosinophils induced by PAF[1].
Bepotastine besilate (3 mg/kg; p.o.; once) suppresses scratching behavior to a frequency of 59.0 and a duration of 14.57 seconds, near identical levels to the control[3]. Bepotastine besilate (10 mg/kg; p.o.; once) (10 mg/kg; p.o.; once) considerably reduces serum LTB 4 levels to 711.3 pg/mL at 1 h and 858.8 pg/mL at 2 h in NC/Nga mice with a rash[3]. Experimental allergic conjunctivitis (EAC) model in rats: Topical administration of Bepotastine Besilate (Bepreve) (0.1%, 0.3%, 1% eye drops) twice daily for 7 days reduced conjunctival redness, edema, and eosinophil infiltration by 45-70% in a dose-dependent manner. Oral administration (1 mg/kg, 3 mg/kg) also inhibited vascular hyperpermeability by 58% and 72% respectively [1] - Atopic dermatitis (AD) model in NC/Nga mice: Oral gavage of Bepotastine Besilate (Bepreve) (1 mg/kg/day, 3 mg/kg/day, 10 mg/kg/day) for 4 weeks dose-dependently suppressed scratching behavior (reduced by 32%, 55%, 78% respectively) and improved skin lesions (eczema score reduced by 28%, 46%, 68% respectively) [3] - Guinea pig passive cutaneous anaphylaxis (PCA) model: Intraperitoneal injection of Bepotastine Besilate (Bepreve) (0.3 mg/kg, 1 mg/kg) 1 hour before antigen challenge inhibited skin wheal formation by 52% and 75% respectively [4] |
| Enzyme Assay |
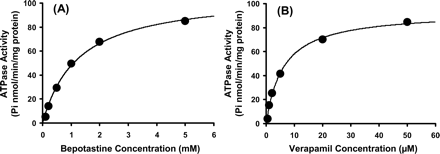
H1R binding assay: Prepare membrane fractions from HEK293 cells expressing human/rat/guinea pig H1R or animal brain tissue. Incubate membranes with [3H]-pyrilamine (0.5 nM) and various concentrations of Bepotastine Besilate (Bepreve) (0.01 nM-100 nM) at 25°C for 60 minutes. Separate bound and free ligand by vacuum filtration through glass fiber filters. Measure radioactivity with a liquid scintillation counter and calculate Ki values using the Cheng-Prusoff equation [4]
|
| Cell Assay |
Cell Line: NHEKs
Concentration: 50 µM (preincubation) Incubation Time: 1 h Result: Suppressed the expression of NGF mRNA in NHEKs. Endothelial cell permeability assay: Seed HUVECs on Transwell inserts and culture to confluence. Pre-treat cells with Bepotastine Besilate (Bepreve) (0.1 nM-1 μM) for 30 minutes, then stimulate with histamine (10 μM) for 1 hour. Add FITC-dextran (4 kDa) to the upper chamber, and measure fluorescence intensity in the lower chamber to quantify permeability [1] - Eosinophil chemotaxis assay: Isolate human eosinophils via density gradient centrifugation. Suspend cells in chemotaxis buffer and add to the upper chamber of transwell plates. Bepotastine Besilate (Bepreve) (1 nM-100 nM) and eotaxin (10 ng/mL) are added to the lower chamber. Incubate at 37°C for 2 hours, then count migrated eosinophils in the lower chamber [1] - Keratinocyte gene expression assay: Seed NHEKs in 6-well plates and incubate until 80% confluent. Treat cells with Bepotastine Besilate (Bepreve) (1 μM-50 μM) for 24 hours. Extract total RNA and protein to detect NEF-H/NEF-M expression via RT-PCR and Western blot; assess cell viability via CCK-8 assay [2] |
| Animal Protocol |
Guinea pigs (6-week-old)
10 g/L (1.0% (w/v)) for 10 µL Eye drop; 3 times at intervals of 20 min (in one eye). EAC rat model: Male Wistar rats (150-200 g) were sensitized with ovalbumin (100 μg) + aluminum hydroxide (2 mg) via intraperitoneal injection on days 0 and 7. From day 14, topical ovalbumin (1% eye drops) was administered to induce EAC. Bepotastine Besilate (Bepreve) eye drops (0.1%, 0.3%, 1%) were applied twice daily, or the drug was dissolved in physiological saline and administered via oral gavage (1 mg/kg, 3 mg/kg) once daily for 7 days. Conjunctival tissues were collected for histopathological analysis and eosinophil counting [1] - AD NC/Nga mouse model: Female NC/Nga mice (6-8 weeks old) were housed in a specific pathogen-free environment and induced with house dust mite extract to develop AD-like lesions. Bepotastine Besilate (Bepreve) was dissolved in 0.5% carboxymethylcellulose sodium and administered via oral gavage (1 mg/kg/day, 3 mg/kg/day, 10 mg/kg/day) for 4 weeks. Scratching behavior was recorded for 1 hour daily; skin lesions were scored based on erythema, edema, scaling, and excoriation [3] - PCA guinea pig model: Male Hartley guinea pigs (300-350 g) were intradermally injected with anti-ovalbumin IgE (0.1 mL) on the back. After 48 hours, Bepotastine Besilate (Bepreve) (0.3 mg/kg, 1 mg/kg) was administered via intraperitoneal injection, followed by intravenous injection of ovalbumin (1 mg/kg) + Evans blue (5 mg/kg) 1 hour later. Guinea pigs were euthanized after 30 minutes, and skin wheal area was measured [4] - Pharmacokinetic animal experiment: Male rats (200-250 g) and beagle dogs (10-15 kg) were administered Bepotastine Besilate (Bepreve) via oral gavage (1-10 mg/kg) or intravenous injection (0.5-2 mg/kg). Blood samples were collected at predetermined time points, and plasma drug concentrations were determined by HPLC-MS/MS [4] |
| ADME/Pharmacokinetics |
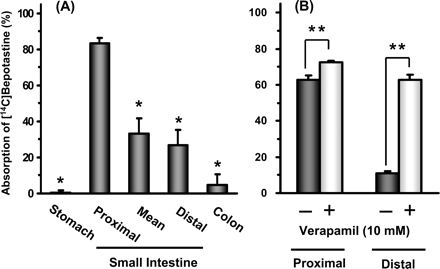
Absorption: The oral bioavailability in rats is 85% and in dogs is 90%; the peak plasma concentration (Cmax) is reached 1-2 hours after oral administration [4]
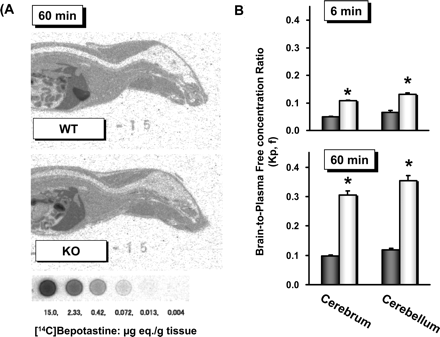
- Distribution: The volume of distribution (Vd) in rats is 1.2 L/kg and in dogs is 1.5 L/kg; the drug is widely distributed in tissues and has very low blood-brain barrier penetration [4] - Metabolism: Very little is metabolized in the liver (≤10% of the dose), and most (90%) is excreted unchanged [4] - Excretion: 75% of the dose is excreted in urine (68% as unchanged drug and 7% as metabolites), and 20% is excreted in feces. The elimination half-life (t1/2) in rats is 4.2 hours and in dogs is 6.8 hours [4] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Overview of Use During Lactation There are currently no reports of mothers breastfeeding their infants while their mothers are using bebombatine. Due to limited absorption of bebombatine through the eyes, no adverse reactions are expected in breastfed infants. After using the eye drops, to significantly reduce the amount of medication entering breast milk, press the tear duct at the corner of the eye for at least 1 minute, then blot away any excess medication with absorbent tissue. ◉ Effects on Breastfed Infants As of the revision date, no published information on bebombatine was found. In a telephone follow-up study, mothers reported that 10% of infants exposed to various antihistamines experienced irritability and colic, and 1.6% experienced drowsiness. All adverse reactions did not require medical attention, and none of the patients were using betahistine. ◉ Effects on Lactation and Breast Milk Higher doses of antihistamines can lower baseline serum prolactin levels in non-lactating women and early postpartum women. However, antihistamine pretreatment in postpartum mothers does not affect lactation-induced prolactin secretion. Prolactin levels in established lactating mothers are unlikely to affect their lactation capacity. Low-dose ophthalmic bebostatin is unlikely to have the same effect on serum prolactin. Plasma protein binding rate: Bepreve has a plasma protein binding rate of 55-60% in human plasma, 50-55% in rat plasma, and 58-63% in canine plasma[4] -Acute toxicity: LD50 in rats and mice >2000 mg/kg (oral); LD50 in rats via intraperitoneal injection >1000 mg/kg [4] -Chronic toxicity: After 6 months of oral administration of Bepreve (10-100 mg/kg/day) to rats and dogs, no significant hepatotoxicity, hematological abnormalities, or organ weight changes were observed[4] -Clinical side effects: Topical ophthalmic use may cause mild eye irritation (burning, itching) in 5-7% of patients; oral administration may cause headache, fatigue, and dry mouth in 3-5% of patients. No sedation or cognitive impairment was observed at therapeutic doses [4] - Genetic toxicity: No genotoxicity was observed in either the in vitro Ames test or the in vivo micronucleus test [4] |
| References |
|
| Additional Infomation |
Bepotastine besylate is an organic sulfonate prepared by reacting equimolar amounts of bebostatin and benzyl sulfonic acid. It is a topical, selective, non-sedating histamine (H1) receptor antagonist used to treat pruritus caused by allergic conjunctivitis. It has a dual role as an H1 receptor antagonist and an anti-allergic agent. It contains bebostatin (1+).
See also: Bebostatin (note moved to). Bebostatin besylate (Bepreve) is a highly selective, non-sedating histamine H1 receptor antagonist with anti-allergic, anti-inflammatory, and antipruritic effects [1,3,4]. Its mechanism of action includes competitive H1R antagonism, inhibition of vascular hyperpermeability, inhibition of eosinophil recruitment, and downregulation of pro-inflammatory cytokines/neural elongation factors [1,2,4]. Indications include allergic conjunctivitis (topical ophthalmic preparation) and atopic dermatitis (oral preparation), which can relieve symptoms such as itching, redness, swelling and scratching [1,3,4]. Its high selectivity for H1R and extremely low blood-brain barrier penetration mean it does not have a sedative effect. Compared with first-generation antihistamines, it has the characteristics of rapid onset of action (within 1 hour after administration) and long duration of action (12-24 hours), so it can be taken once or twice daily. [4] |
| Molecular Formula |
C27H31CLN2O6S
|
|
|---|---|---|
| Molecular Weight |
547.06
|
|
| Exact Mass |
546.159
|
|
| Elemental Analysis |
C, 59.28; H, 5.71; Cl, 6.48; N, 5.12; O, 17.55; S, 5.86
|
|
| CAS # |
190786-44-8
|
|
| Related CAS # |
(Rac)-Bepotastine besilate; 1415692-17-9; Bepotastine; 125602-71-3; Bepotastine tosylate; 1160415-45-1
|
|
| PubChem CID |
164521
|
|
| Appearance |
Off-white to light yellow solid powder
|
|
| Boiling Point |
546.8ºC at 760 mmHg
|
|
| Melting Point |
161-163°
|
|
| Flash Point |
284.5ºC
|
|
| Vapour Pressure |
8.63E-13mmHg at 25°C
|
|
| LogP |
6.122
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
8
|
|
| Rotatable Bond Count |
9
|
|
| Heavy Atom Count |
37
|
|
| Complexity |
632
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
ClC1C([H])=C([H])C(=C([H])C=1[H])[C@@]([H])(C1=C([H])C([H])=C([H])C([H])=N1)OC1([H])C([H])([H])C([H])([H])N(C([H])([H])C([H])([H])C([H])([H])C(=O)O[H])C([H])([H])C1([H])[H].S(C1C([H])=C([H])C([H])=C([H])C=1[H])(=O)(=O)O[H]
|
|
| InChi Key |
UDGHXQPQKQPSBB-BOXHHOBZSA-N
|
|
| InChi Code |
InChI=1S/C21H25ClN2O3.C6H6O3S/c22-17-8-6-16(7-9-17)21(19-4-1-2-12-23-19)27-18-10-14-24(15-11-18)13-3-5-20(25)26;7-10(8,9)6-4-2-1-3-5-6/h1-2,4,6-9,12,18,21H,3,5,10-11,13-15H2,(H,25,26);1-5H,(H,7,8,9)/t21-;/m0./s1
|
|
| Chemical Name |
benzenesulfonic acid;4-[4-[(S)-(4-chlorophenyl)-pyridin-2-ylmethoxy]piperidin-1-yl]butanoic acid
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.57 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.57 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (4.57 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8280 mL | 9.1398 mL | 18.2795 mL | |
| 5 mM | 0.3656 mL | 1.8280 mL | 3.6559 mL | |
| 10 mM | 0.1828 mL | 0.9140 mL | 1.8280 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03932435 | Completed | Drug: Fisrt period TWLO_C Other: Washout period |
Healthy | Dong-A ST Co., Ltd. | April 23, 2019 | Not Applicable |
| NCT01840605 | Completed | Drug: Bepotastine besilate Drug: ketotifen fumarate |
Dermatitis Atopic |
Mitsubishi Tanabe Pharma Corporation | March 2013 | Phase 3 |
| NCT01753739 | Completed | Drug: Bepotastine besilate Drug: Placebo |
Seasonal Allergic Rhinitis | Bausch & Lomb Incorporated | January 2013 | Phase 2 |
| NCT01861522 | Completed | Drug: Bepotastine besilate Drug: Placebo |
Perennial Allergic Rhinitis | Mitsubishi Tanabe Pharma Corporation |
April 2013 | Phase 3 |
| NCT01900054 | Completed | Drug: Bepotastine besilate | Perennial Allergic Rhinitis | Mitsubishi Tanabe Pharma Corporation |
June 2013 | Phase 3 |
 |
|---|
 |
 |