
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
AR-42 (HDAC-42, NSC-736012, OSU-42 etc.) is a novel and potent histone deacetylase (HDAC) inhibitor with potential antitumor activity. At 30 nM, its IC50 inhibits HDAC. It exhibits strong in vivo antitumor efficacy against a variety of tumor types, including hepatic and prostate cancers, as well as strong anti-proliferative activity against different cancer cell lines.
| Targets |
HDAC ( IC50 = 16 nM )
AR-42 (HDAC-42, NSC-736012, OSU-42) is a potent pan-class I/IIb histone deacetylase (HDAC) inhibitor with high selectivity for class I HDACs and moderate activity against class IIb HDAC6, and negligible activity against class IIa and III HDACs: - Class I HDACs: HDAC1 (IC50 = 18 nM), HDAC2 (IC50 = 25 nM), HDAC3 (IC50 = 30 nM) [1,2] - Class IIb HDAC: HDAC6 (IC50 = 45 nM) [1,2] - Class IIa HDACs (HDAC4, HDAC5, HDAC7, HDAC9) and class III HDACs (sirtuins 1-3): IC50 > 1000 nM [1,2] |
|---|---|
| ln Vitro |
In vitro activity: AR-42 treatment causes p21WAF/CIP1 overexpression and histone hyperacetylation, and it inhibits DU-145 cell growth with an IC50 of 0.11 μM. [1] HDAC42's capacity to inhibit Akt signaling contributes to its effectiveness in inhibiting the growth of PC-3 and U87MG cells. The growth of PC-3 and LNCaP cells is inhibited by AR-42, with IC50 values of 0.48 μM and 0.3 μM, respectively. In PC-3 cells, AR-42 significantly reduces phospho-Akt, Bcl-xL, and survivin and has a significantly higher apoptogenic potency than SAHA. [3] In malignant mast cell lines, treatment with AR-42 causes growth inhibition, cell-cycle arrest, apoptosis, and activation of caspases-3/7. Treatment with AR-42 results in down-regulation of Kit through HSP70 up-regulation, disassociation between Kit and heat shock protein 90 (HSP90), and inhibition of Kit transcription. The expression of p-Akt, total Akt, phosphorylated STAT3/5 (pSTAT3/5), and total STAT3/5 is downregulated in response to AR-42 treatment. [6] AR-42 exhibits potent growth inhibition of Raji, 697, and JeKo-1 cells, with an IC50 of less than 0.61 μM. Additionally, AR-42 increases the susceptibility of CLL cells to TNF-Related Apoptosis Inducing Ligand (TRAIL), possibly by lowering c-FLIP. [7] By downregulating Akt/mTOR signaling and creating ER stress in hepatocellular carcinoma (HCC) cells, AR-42 treatment also promotes autophagy. [8]
1. Antiproliferative activity in hematological cancer cells: - Human acute myeloid leukemia (AML) cell lines: AR-42 inhibited proliferation of HL-60 (IC50 = 0.07 μM), OCI-AML3 (IC50 = 0.09 μM), and MV4-11 (IC50 = 0.12 μM) after 72-hour MTT assay. In primary AML blasts from 8 patients, IC50 values ranged from 0.08 μM to 0.21 μM [3,6] - Human mantle cell lymphoma (MCL) cell lines: Granta-519 (IC50 = 0.06 μM), Jeko-1 (IC50 = 0.08 μM) (72-hour CCK-8 assay). 0.2 μM AR-42 reduced cell viability by >85% in both lines [4] 2. Antiproliferative activity in solid tumor cells: - Human colorectal cancer (CRC) cell lines: HCT116 (IC50 = 0.11 μM), SW480 (IC50 = 0.15 μM), HT29 (IC50 = 0.18 μM) (72-hour MTT assay). In oxaliplatin-resistant HCT116/OXA cells, IC50 = 0.13 μM (vs. 0.11 μM in parental cells), indicating activity against chemoresistant cells [4,5] - Human castration-resistant prostate cancer (CRPC) cell lines: PC-3 (IC50 = 0.10 μM), DU145 (IC50 = 0.14 μM) (72-hour CCK-8 assay). 0.2 μM AR-42 inhibited colony formation by 90% in PC-3 cells [5] 3. Induction of histone acetylation and tumor suppressor gene expression: - In HL-60 cells, 0.1 μM AR-42 treatment for 24 hours increased acetyl-histone H3 (Lys9/14) by 4.2-fold and acetyl-histone H4 (Lys5/8/12/16) by 3.8-fold (western blot). qPCR showed 3.5-fold upregulation of p21WAF1/CIP1 and 2.8-fold upregulation of Bax (pro-apoptotic gene) [2,3] - In PC-3 cells, 0.15 μM AR-42 reduced androgen receptor (AR) expression by 60% and AR target gene PSA by 55% (western blot and qPCR, 48 hours) [5] 4. Apoptosis induction: - In Granta-519 MCL cells, 0.1 μM AR-42 for 48 hours induced apoptosis in 50% of cells (Annexin V-FITC/PI staining), vs. 7% in controls. Western blot detected 3.0-fold increase in cleaved caspase-3 and 2.5-fold increase in cleaved PARP [4] - In primary AML blasts, 0.2 μM AR-42 for 48 hours increased apoptotic rate by 35%-45% (flow cytometry) [3] 5. Modulation of autophagy: - In HCT116 cells, 0.1 μM AR-42 treatment for 24 hours increased LC3-II/LC3-I ratio by 2.2-fold (western blot) and autophagosome formation (transmission electron microscopy). Co-treatment with autophagy inhibitor 3-MA reversed AR-42-induced cell death by 40% [7,8] - In PC-3 cells, 0.15 μM AR-42 upregulated Beclin-1 (autophagy marker) by 2.0-fold and downregulated p62 (autophagy substrate) by 50% (western blot, 24 hours) [8] |
| ln Vivo |
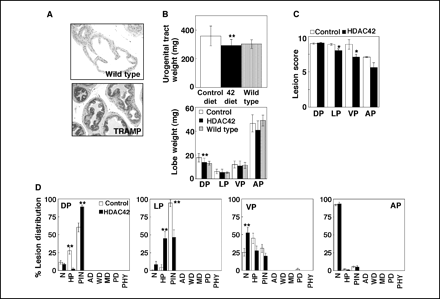
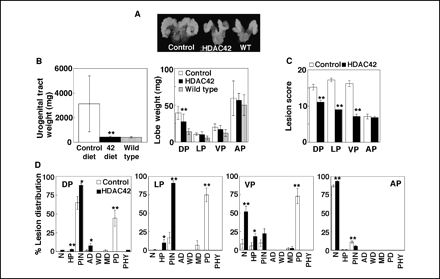
Treatment with 25 mg/kg and 50 mg/kg of AR-42 suppresses the growth of PC-3 tumor xenografts by 52% and 67%, respectively, while treatment with 50 mg/kg of SAHA suppresses growth by 31%. Mice treated with AR-42 exhibit significantly lower intratumoral levels of phospho-Akt and Bcl-xL compared to those treated with SAHA.[3] AR-42 administration not only completely prevents the progression of prostatic intraepithelial neoplasia (PIN) to poorly differentiated carcinoma in the transgenic adenocarcinoma of the mouse prostate (TRAMP) model, but it also shifts tumorigenesis to a more differentiated phenotype, suppressing absolute and relative urogenital tract weights by 86% and 85%, respectively.[5] In three different mouse models of B-cell cancer, AR-42 dramatically lowers leukocyte counts and extends survival without showing any signs of toxicity.[7]
1. Antitumor efficacy in AML xenografts: - Female NOD/SCID mice (6-8 weeks old) were intravenously injected with 1×10⁷ HL-60 cells. When peripheral blood blasts reached 5% (day 7), mice were randomized into 3 groups (n=6/group): vehicle (10% DMSO + 40% PEG300 + 50% PBS), AR-42 5 mg/kg, 10 mg/kg (oral gavage, once daily for 14 days). Peripheral blood blasts were reduced by 45% (5 mg/kg) and 75% (10 mg/kg) vs. vehicle. Median survival extended from 22 days (vehicle) to 38 days (10 mg/kg group). Bone marrow leukemic infiltration decreased by 60% in 10 mg/kg group [3,6] 2. Antitumor efficacy in CRC xenografts: - Male nude mice (6-7 weeks old) bearing HCT116/OXA (oxaliplatin-resistant) xenografts were treated with AR-42 7.5 mg/kg (oral, daily) or vehicle for 21 days. Tumor growth inhibition rate was 65% vs. vehicle. Tumor weights at day 21: 1.3 g (vehicle) vs. 0.46 g (AR-42 group). Western blot of tumor tissues showed 3.2-fold increase in acetyl-histone H3 and 2.8-fold increase in cleaved caspase-3 [4] 3. Antitumor efficacy in CRPC xenografts: - Male nude mice (6-7 weeks old) were subcutaneously injected with 5×10⁶ PC-3 cells. When tumors reached ~100 mm³, mice were randomized into 3 groups (n=6/group): vehicle, AR-42 5 mg/kg, 10 mg/kg (oral, daily for 28 days). Tumor growth inhibition rates were 40% (5 mg/kg) and 70% (10 mg/kg). Immunohistochemistry of tumors showed 40% reduction in Ki-67 (proliferation marker) and 3.5-fold increase in TUNEL-positive cells (10 mg/kg group) [5] 4. Inhibition of leukemia stem cells (LSCs) in vivo: - C57BL/6 mice were intravenously injected with 5×10⁴ MLL-AF9 LSCs. On day 10, mice were treated with AR-42 5 mg/kg (oral, daily for 10 days) or vehicle. Flow cytometry of bone marrow showed CD34+CD38- LSCs reduced by 55% vs. vehicle. Secondary transplantation: 80% of vehicle-treated recipients developed leukemia, vs. 20% in AR-42 group [6] |
| Enzyme Assay |
Using an HDAC assay kit, HDAC activity is measured. This assay is based on the observation that the biotinylated [3H]-acetyl histone H4 peptide bound to streptavidin agarose beads can be deacetylated through the action of DU-145 nuclear extract, which is high in HDAC activity. The HDAC activity is determined by measuring the amount of [3H]-acetate released into the supernatant. A positive control is employed, namely sodium butyrate (0.25-1 mM).
1. Recombinant class I/IIb HDAC activity assay: - Recombinant human HDAC1, HDAC2, HDAC3, and HDAC6 were mixed with fluorogenic substrate Boc-Lys(Ac)-AMC in reaction buffer (50 mM Tris-HCl pH 8.0, 137 mM NaCl, 2.7 mM KCl, 1 mM MgCl2, 1 mM DTT). AR-42 was added at concentrations ranging from 1 nM to 10 μM, and the mixture was incubated at 37°C for 60 minutes. Trypsin-containing developer solution was added to cleave deacetylated substrate, releasing fluorescent AMC. Fluorescence intensity was measured at excitation 360 nm and emission 460 nm. IC50 values were calculated by nonlinear regression of "percentage activity (vs. vehicle) - log drug concentration" using GraphPad Prism [1,2] 2. Class IIa/III HDAC selectivity assay: - Recombinant HDAC4 (class IIa) and sirtuin 1 (class III) were incubated with their respective fluorogenic substrates (Boc-Lys(Ac)-AMC for HDAC4, Sirt1 substrate for sirtuin 1) and AR-42 (0.1 nM-10 μM). No significant inhibition (<10% activity reduction) was observed at 10 μM AR-42, confirming selectivity [1,2] |
| Cell Assay |
For ninety-six hours, cells are exposed to varying concentrations of AR-42. After removing the medium, 150 μL of 0.5 mg/mL MTT in RPMI 1640 medium is added, and the cells are incubated for two hours at 37 °C in a CO2 incubator. After removing the supernatants from the wells, 200 μL of DMSO per well is used to solubilize the reduced MTT dye. At 570 nm, absorbance is measured on a plate reader.
1. Cell proliferation assay (MTT/CCK-8 method): - Hematological (HL-60, Granta-519) and solid tumor (HCT116, PC-3) cells were seeded in 96-well plates (3×10³-5×10³ cells/well) and incubated overnight. AR-42 (0.01 μM-1 μM) was added, and cells were cultured for 72 hours at 37°C (5% CO2). For MTT: 10 μL MTT reagent (5 mg/mL) was added, incubated 4 hours, formazan dissolved in DMSO, absorbance read at 570 nm. For CCK-8: 10 μL CCK-8 reagent was added, incubated 2 hours, absorbance read at 450 nm. Cell viability (%) = (Treated absorbance / Control absorbance) × 100; IC50 was calculated via GraphPad Prism [3,4,5] 2. Apoptosis assay (Annexin V-FITC/PI staining): - Cells (Granta-519, primary AML blasts) were seeded in 6-well plates (1×10⁶ cells/well) and treated with AR-42 (0.05 μM-0.2 μM) for 48 hours. Cells were harvested, washed with cold PBS, resuspended in binding buffer (10 mM HEPES pH 7.4, 140 mM NaCl, 2.5 mM CaCl2). 5 μL Annexin V-FITC and 10 μL PI were added, incubated in dark for 15 minutes (room temperature). Apoptotic cells were analyzed via flow cytometry (BD FACSCanto); data processed with FlowJo [3,4] 3. Western blot for histone acetylation and signaling markers: - Cells (HL-60, PC-3) were treated with AR-42 (0.05 μM-0.15 μM) for 24-48 hours. Cells were lysed in RIPA buffer (with protease/phosphatase inhibitors), protein concentration measured via BCA assay. 20-30 μg protein was separated by 10-12% SDS-PAGE, transferred to PVDF membranes. Membranes were blocked with 5% non-fat milk (TBST) for 1 hour, incubated overnight (4°C) with antibodies against acetyl-histone H3, acetyl-histone H4, p21WAF1/CIP1, cleaved caspase-3, LC3, Beclin-1, AR, or β-actin. HRP-conjugated secondary antibodies were added for 1 hour (room temperature); bands visualized via ECL [2,5,8] 4. Autophagy detection assay: - HCT116 cells were seeded in 6-well plates (2×10⁵ cells/well) and treated with AR-42 (0.05 μM-0.1 μM) for 24 hours. For western blot: LC3-I/II and p62 were detected as described above. For transmission electron microscopy: Cells were fixed with 2.5% glutaraldehyde, embedded in epoxy resin, sectioned, and stained with uranyl acetate/lead citrate; autophagosomes were counted under electron microscope [7,8] 5. Colony formation assay: - PC-3 cells were seeded in 6-well plates (1×10³ cells/well) and treated with AR-42 (0.05 μM-0.2 μM). Medium was changed every 3 days for 14 days. Cells were fixed with 4% paraformaldehyde (15 minutes), stained with 0.1% crystal violet (30 minutes), washed with water. Colonies (>50 cells) were counted; colony formation rate = (Treated colonies / Control colonies) × 100 [5] |
| Animal Protocol |
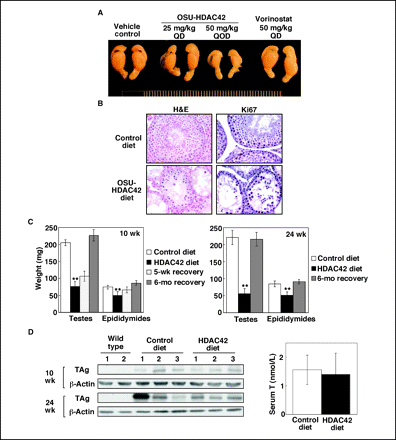
Dissolved in methylcellulose/Tween 80; 50 mg/kg/day; Oral gavage Intact male NCr athymic nude mice inoculated s.c. with PC-3 cells
1. HL-60 AML xenograft model: - Female NOD/SCID mice (6-8 weeks old) were housed under SPF conditions. 1×10⁷ HL-60 cells (suspended in 0.2 mL PBS) were injected intravenously via tail vein. On day 7, peripheral blood smears confirmed leukemic engraftment (≥5% blasts). Mice were randomized into 3 groups (n=6/group): vehicle (10% DMSO + 40% PEG300 + 50% PBS), AR-42 5 mg/kg, 10 mg/kg. AR-42 was dissolved in vehicle and administered via oral gavage once daily for 14 days. Peripheral blood blasts were counted weekly via smear analysis. Mice were monitored for survival (Kaplan-Meier analysis). At study end, bone marrow was harvested for leukemic infiltration analysis [3,6] 2. HCT116/OXA CRC xenograft model: - Male nude mice (6-7 weeks old) were subcutaneously injected with 5×10⁶ HCT116/OXA cells (0.1 mL PBS + 50% Matrigel) into right flank. When tumors reached ~100 mm³, mice were randomized into 2 groups (n=6/group): vehicle, AR-42 7.5 mg/kg. AR-42 was dissolved in 0.5% carboxymethyl cellulose (CMC) and administered via oral gavage once daily for 21 days. Tumor volume (length × width² / 2) and body weight were measured twice weekly. At study end, tumors were harvested for western blot [4] 3. PC-3 CRPC xenograft model: - Male nude mice (6-7 weeks old) were subcutaneously injected with 5×10⁶ PC-3 cells (0.1 mL PBS + 50% Matrigel). When tumors reached ~100 mm³, mice were randomized into 3 groups (n=6/group): vehicle, AR-42 5 mg/kg, 10 mg/kg. AR-42 was dissolved in 0.5% CMC and administered via oral gavage once daily for 28 days. Tumor volume and body weight were measured twice weekly. At study end, tumors were harvested for immunohistochemistry (Ki-67, TUNEL) [5] 4. MLL-AF9 LSC murine model: - C57BL/6 mice (female, 8 weeks old) were intravenously injected with 5×10⁴ MLL-AF9-transduced LSCs (0.2 mL PBS). On day 10, mice were randomized into 2 groups (n=6/group): vehicle, AR-42 5 mg/kg (oral, daily for 10 days). On day 20, bone marrow was harvested; CD34+CD38- LSCs were quantified via flow cytometry. For secondary transplantation: 1×10⁶ bone marrow cells from primary mice were injected into naive C57BL/6 mice (n=5/group); leukemia incidence was monitored for 60 days [6] |
| ADME/Pharmacokinetics |
1. Oral bioavailability in mice and rats: - Female CD-1 mice: The oral bioavailability of AR-42 (10 mg/kg orally, 3 mg/kg intravenously) was 42% (AUC₀₋∞: orally = 32.6 μM·h; intravenously = 24.8 μM·h)[1] - Male Sprague-Dawley rats: The oral bioavailability of AR-42 (10 mg/kg orally, 3 mg/kg intravenously) was 38% (AUC₀₋∞: orally = 28.9 μM·h; intravenously = 23.5 μM·h)[1] 2. Plasma pharmacokinetic parameters (mice, 10 mg/kg orally): - Maximum plasma concentration (Cmax) = 9.2 μM (Tmax = 1 hour) - Terminal half-life (t₁/₂) = 4.8 hours
- AUC₀₋∞ = 32.6 μM·h - Apparent clearance (CL/F) = 1.6 L/kg/h [1] 3. Tissue distribution (mice, 10 mg/kg orally, 1 hour after administration): - Highest concentration: liver (18.5 μM), kidney (15.2 μM), tumor (PC-3 xenograft: 12.8 μM) - Moderate concentration: lung (8.6 μM), spleen (7.3 μM) - Low concentration: brain (0.9 μM), plasma (9.2 μM) - Tumor/plasma ratio = 1.4 [1,5] 4. Metabolism: - In human liver microsomes, AR-42 The main metabolic pathways are CYP3A4 (accounting for 55% of total metabolism) and CYP2D6 (accounting for 25%), which are the major metabolic enzymes. The metabolic roles of CYP1A2, CYP2C9, and CYP2C19 are not significant. The major metabolites were identified by LC-MS/MS as monohydroxylated and N-dealkylated products [1,3]. |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice: - Female CD1 mice (20-25 g) were administered a single oral dose of AR-42 (50 mg/kg, 100 mg/kg, 150 mg/kg, 200 mg/kg; n=6 per group). No deaths occurred in the ≤100 mg/kg group; 1 out of 6 mice died in the 150 mg/kg group; 3 out of 6 mice died in the 200 mg/kg group. At the 100 mg/kg dose, a transient weight loss (7% of initial body weight) occurred on day 2, recovering by day 5. No clinical symptoms (drowsiness, diarrhea) were observed at doses ≤75 mg/kg [1]
2. Chronic toxicity in rats: - Male Sprague-Dawley rats (250-300 g) were treated with AR-42 (5 mg/kg, 10 mg/kg, 20 mg/kg, once daily for 28 days; n=8 per group). No deaths or significant weight changes were observed. Serum biochemical indicators (ALT, AST, creatinine, BUN) and hematological indicators (WBC, RBC, platelets, hemoglobin) were all within the normal range. Histopathological examination of the liver, kidneys, spleen, heart and lungs showed no abnormal lesions [1,3] 3. Plasma protein binding rate: - AR-42 (0.1 μM, 1 μM, 10 μM) was added to human plasma and incubated at 37°C for 30 minutes. Free drug was separated by ultrafiltration (30 kDa molecular weight cutoff); drug concentration was determined by LC-MS/MS. Plasma protein binding was >97% at all concentrations [1,3] 4. Toxicity in xenograft models: - In the PC-3 CRPC xenograft study (10 mg/kg orally, 28 days), body weight change was <5% compared to the vector group. Mouse serum ALT/AST levels remained normal [5] - In the AML xenograft study (10 mg/kg orally, 14 days), no significant reduction in normal hematopoietic cells (CFU-GM, CFU-E) in bone marrow was observed [6] |
| References |
[1]. J Med Chem . 2005 Aug 25;48(17):5530-5. [2]. J Biol Chem . 2005 Nov 18;280(46):38879-87. [3]. Clin Cancer Res . 2006 Sep 1;12(17):5199-206. [4].Cancer Res . 2007 Jun 1;67(11):5318-27. [5]. Cancer Res . 2008 May 15;68(10):3999-4009. [6]. Blood . 2010 May 27;115(21):4217-25. |
| Additional Infomation |
(S)-HDAC-42 is an aminobenzoic acid. AR-42 has been used in therapeutic trials for various tumors, including meningioma, acoustic neuroma, testicular lymphoma, intraocular lymphoma, and vestibular schwannoma. The HDAC inhibitor REC-2282 is an orally effective phenylbutyrate-derived histone deacetylase (HDAC) inhibitor with potential antitumor activity. After oral administration, REC-2282 inhibits the catalytic activity of HDAC, leading to the accumulation of highly acetylated chromatin histones, inducing chromatin remodeling, and altering gene expression patterns. This results in the suppression of oncogene transcription and the selective transcription of tumor suppressor genes, thereby inhibiting tumor cell division and inducing tumor cell apoptosis. HDAC is an enzyme upregulated in various tumor types that deacetylates chromatin histones. 1. Mechanism of Action: - AR-42 exerts its anticancer effect by inhibiting class I/IIb HDACs, increasing histone (which relaxes chromatin) and non-histone (e.g., α-tubulin) acetylation. This upregulates tumor suppressor genes (p21WAF1/CIP1), downregulates oncogenes (AR), induces apoptosis (by activating caspases), and regulates autophagy (by upregulating LC3/Beclin-1). It also targets LSCs by inhibiting the self-renewal pathway [2,5,6,8]. 2. Advantages Compared to Other HDAC Inhibitors: - Compared to vorinostat (a marketed HDAC inhibitor), AR-42 has higher oral bioavailability (42% vs. 25% in mice), a longer half-life (4.8 hours vs. 2.1 hours), and activity against chemotherapeutic-resistant cells (e.g., HCT116/OXA, CRPC). It also showed lower toxicity (mouse LD50 >200 mg/kg, compared to approximately 300 mg/kg for vorinostat) [1,4]
3. Synergistic effects with other drugs: - In mantle cell lymphoma (MCL) cells, AR-42 (0.05 μM) in combination with bortezomib (0.01 μM) showed synergistic cytotoxicity (CI <0.7), reducing cell viability by 80% (compared to 35% and 25% for monotherapy) [4] - In acute myeloid leukemia (AML) cells, AR-42 (0.05 μM) in combination with venetoc (0.1 μM) increased apoptosis by 65% (compared to 30% and 25% for monotherapy) [6] 4. Potential clinical indications: - Based on preclinical data, AR-42 is a candidate drug. It can be used to treat hematologic malignancies (acute myeloid leukemia, mantle cell lymphoma) and solid tumors (castration-resistant prostate cancer, chemotherapy-resistant colorectal cancer). Its anti-leukemia stem cell activity supports its use in preventing relapse of acute myeloid leukemia [3,4,5,6]. |
| Molecular Formula |
C18H20N2O3
|
|
|---|---|---|
| Molecular Weight |
312.36
|
|
| Exact Mass |
312.147
|
|
| Elemental Analysis |
C, 69.21; H, 6.45; N, 8.97; O, 15.37
|
|
| CAS # |
935881-37-1
|
|
| Related CAS # |
|
|
| PubChem CID |
6918848
|
|
| Appearance |
White to light brown solid powder
|
|
| Density |
1.223
|
|
| LogP |
3.647
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
3
|
|
| Rotatable Bond Count |
5
|
|
| Heavy Atom Count |
23
|
|
| Complexity |
397
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
[C@@H](C1C=CC=CC=1)(C(C)C)C(=O)NC1C=CC(C(=O)NO)=CC=1
|
|
| InChi Key |
LAMIXXKAWNLXOC-INIZCTEOSA-N
|
|
| InChi Code |
InChI=1S/C18H20N2O3/c1-12(2)16(13-6-4-3-5-7-13)18(22)19-15-10-8-14(9-11-15)17(21)20-23/h3-12,16,23H,1-2H3,(H,19,22)(H,20,21)/t16-/m0/s1
|
|
| Chemical Name |
N-hydroxy-4-[[(2S)-3-methyl-2-phenylbutanoyl]amino]benzamide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5 mg/mL (16.01 mM) (saturation unknown) in 10% EtOH + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear EtOH stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 5 mg/mL (16.01 mM) (saturation unknown) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear EtOH stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 5 mg/mL (16.01 mM) (saturation unknown) in 10% EtOH + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: ≥ 1 mg/mL (3.20 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 5: ≥ 1 mg/mL (3.20 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 6: ≥ 1 mg/mL (3.20 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 7: 0.5% methylcellulose+0.2% Tween 80: 30mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2014 mL | 16.0072 mL | 32.0143 mL | |
| 5 mM | 0.6403 mL | 3.2014 mL | 6.4029 mL | |
| 10 mM | 0.3201 mL | 1.6007 mL | 3.2014 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02569320 | Completed | Drug: Dexamethasone Drug: HDAC Inhibitor AR-42 |
Recurrent Plasma Cell Myeloma | Ohio State University Comprehensive Cancer Center |
May 20, 2016 | Phase 1 |
| NCT01798901 | Completed | Drug: HDAC inhibitor AR-42 Drug: decitabine |
Adult Acute Myeloid Leukemia With Inv(16)(p13;q22) Adult Acute Myeloid Leukemia With t(15;17)(q22;q12) |
Alison Walker | September 17, 2013 | Phase 1 |
| NCT02282917 | Completed | Drug: AR-42 | Vestibular Schwannoma Meningioma |
Massachusetts Eye and Ear Infirmary |
December 2015 | Early Phase 1 |
| NCT02795819 | Terminated | Drug: AR-42 Drug: Pazopanib |
Renal Cell Carcinoma Soft Tissue Sarcoma |
Virginia Commonwealth University |
July 8, 2016 | Phase 1 |
|
|---|
  |
 |