
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
Aclidinium Bromide (formerly LAS34273, LAS-W 330; brand names Tudorza Genuair; Eklira Genuair; Bretaris Genuair) is a potent, long-acting and inhalable muscarinic antagonist used as a maintenance treatment for chronic obstructive pulmonary disease (COPD). It acts by potently inhibiting human muscarinic acetylcholine receptors-AChR M1, M2, M3, M4 and M5 with Ki values of 0.1 nM, 0.14 nM, 0.14 nM, 0.21 nM and 0.16 nM, respectively.
| Targets |
Muscarinic acetylcholine receptors (M1-M5), Ki values: M1 (1.6 nM), M2 (3.7 nM), M3 (0.8 nM), M4 (2.3 nM), M5 (4.1 nM) [1][3][4]
- Airway smooth muscle M3 muscarinic receptors (primary target) [1][2] |
|---|---|
| ln Vitro |
Aclidinium shows strong antagonistic activity against human muscarinic receptors in a preclinical setting. It has a longer half-life at M3 receptors than it does at M2 receptors, indicating that it may have a long-lasting bronchodilator action. Acclidinium hydrolyzes quickly in human plasma, in contrast to tiotropium and other antimuscarinics that are already on the market. Single- and multiple-dose aclidinium has a good safety profile and a low systemic bioavailability, according to early clinical trials conducted in healthy participants. Aclidinium exhibited prolonged bronchodilatory action and was well tolerated in 464 individuals with moderate to severe COPD who participated in a subsequent phase IIb research.
Aclidinium Bromide (LAS 34273; LAS-W 330) (0.01 μM-100 μM) exhibited potent and selective competitive antagonism of M3 muscarinic receptors, with the highest affinity for M3 subtype (Ki=0.8 nM). It concentration-dependently inhibited acetylcholine-induced contraction of isolated human and guinea-pig airway smooth muscle, with an IC50 of 1.2 nM [1][4] - In human bronchial epithelial cells, Aclidinium Bromide (0.1 μM-10 μM) reduced acetylcholine-induced mucus secretion by 35% (1 μM) and 58% (10 μM) compared to control, via inhibiting M3 receptor-mediated Ca²⁺ influx [2][4] - It was rapidly inactivated in human plasma by esterases, producing two main metabolites: N-desalkyl aclidinium (Ki for M3=12 nM) and a carboxylic acid derivative (Ki for M3=25 nM), both with significantly lower muscarinic receptor affinity than the parent drug [3] |
| ln Vivo |
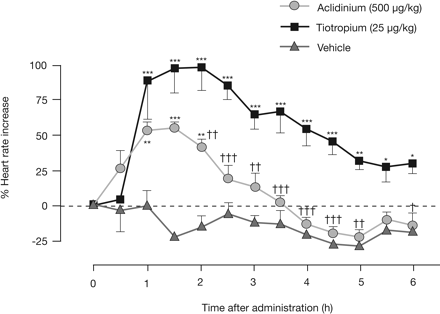
Aclidinium shows an onset of action with IC50 (95% CI) of 140 μg/mL and tmax of 30 min in the acetylcholine-induced bronchoconstriction model in anesthetized guinea pigs. Aclidinium (500 μg/kg) induces a maximal increase in heart rate of 55% after 1 hour in conscious beagle dogs. Aclidinium (1 mg/ml) produces a potent and sustained bronchoprotection (72%–88.4%) over the 120-min study period in anaesthetised guinea pigs.
In guinea-pigs with acetylcholine-induced bronchoconstriction, inhaled Aclidinium Bromide (0.1 mg/kg, 0.3 mg/kg, 1 mg/kg) exerted dose-dependent bronchoprotective effects: the maximum inhibition of bronchial contraction was 52% (0.3 mg/kg) and 75% (1 mg/kg), with a duration of action exceeding 12 hours [1][4] - In rats with ovalbumin-induced allergic airway inflammation, repeated inhalation of Aclidinium Bromide (0.5 mg/kg, once daily for 7 days) reduced airway hyperresponsiveness by 48% and decreased eosinophil infiltration in lung tissues by 55% compared to control [2] - Intravenous administration of Aclidinium Bromide (0.3 mg/kg) in dogs inhibited methacholine-induced increases in airway resistance by 68%, with no significant effect on heart rate or blood pressure [1] |
| Enzyme Assay |
Muscarinic receptor binding assay: Membrane fractions enriched with human recombinant M1-M5 receptors or rat brain tissues were incubated with serial concentrations of Aclidinium Bromide (0.001 μM-100 μM) in the presence of a tritiated muscarinic agonist. Incubation was performed at 25°C for 90 minutes, unbound ligands were removed by vacuum filtration, and bound radioactivity was measured. Ki values for each receptor subtype were calculated via nonlinear regression of displacement curves [1][3]
- Plasma esterase metabolism assay: Human plasma was incubated with Aclidinium Bromide (1 μM) at 37°C for 0-120 minutes. The reaction was terminated by adding acetonitrile, and parent drug and metabolites were separated and quantified by HPLC-MS. The metabolic rate and metabolite formation kinetics were analyzed [3] |
| Cell Assay |
Airway smooth muscle contraction assay: Human airway smooth muscle cells were isolated and cultured on collagen-coated plates. Cells were preincubated with Aclidinium Bromide (0.01 μM-100 μM) for 30 minutes, then stimulated with acetylcholine (10 μM). Cell contraction was assessed by measuring changes in cell length using phase-contrast microscopy. Concentration-response curves were constructed to determine IC50 values [1][4]
- Mucus secretion assay: Human bronchial epithelial cells were seeded in air-liquid interface cultures. Aclidinium Bromide (0.1 μM-10 μM) was added to the basolateral side 1 hour before acetylcholine (10 μM) stimulation. Mucus secretion was quantified by enzyme-linked lectin assay, and intracellular Ca²⁺ concentration was measured using a calcium-sensitive fluorescent probe [2][4] |
| Animal Protocol |
Prepared as Aerosol solutions; 500 μg/kg; administered through a nebulizer
Beagle dogs Guinea-pig bronchoconstriction model: Male guinea-pigs were anesthetized, tracheotomized, and connected to a ventilation system. Aclidinium Bromide was administered via nebulization at doses of 0.1 mg/kg, 0.3 mg/kg, and 1 mg/kg. Thirty minutes later, acetylcholine was inhaled to induce bronchoconstriction. Bronchial resistance and dynamic compliance were measured for 12 hours to evaluate the bronchoprotective effect [1][4] - Rat allergic airway inflammation model: Rats were sensitized with ovalbumin (OVA) on day 0 and day 14, then challenged with OVA aerosol from day 21 to day 27. Aclidinium Bromide (0.5 mg/kg) was administered via nebulization once daily during the challenge period. On day 28, rats were sacrificed, and lung tissues were collected for histological analysis and eosinophil counting [2] - Dog cardiovascular safety model: Beagle dogs were anesthetized and instrumented to monitor heart rate, blood pressure, and airway resistance. Aclidinium Bromide was administered via intravenous injection at 0.3 mg/kg, followed by methacholine inhalation. Cardiovascular parameters and airway resistance were recorded for 6 hours [1] |
| ADME/Pharmacokinetics |
The bioavailability of acrylamide in the human body is about 15-20%, and the peak plasma concentration is reached 15-30 minutes after nebulization [4]. The steady-state volume of distribution (Vdss) is about 0.3 L/kg. Due to its quaternary ammonium structure, very little of the drug penetrates into the central nervous system [3][4]. The drug is rapidly metabolized in plasma by non-specific esterases, and the elimination half-life is 5-8 hours (inhalation route) [3]. About 60% of the drug is excreted in urine and 30% in feces, mainly in the form of inactive metabolites; less than 5% of the drug is excreted unchanged [3][4].
|
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Overview of use during lactation Although there is currently no publicly available data on the use of aprazole during lactation, maternal serum concentrations are low due to its rapid hydrolysis to inactive metabolites. Breastfed infants face minimal risk from aprazole inhalation. Breastfeeding can continue during aprazole treatment. ◉ Effects on breastfed infants As of the revision date, no relevant published information was found. ◉ Effects on lactation and breast milk As of the revision date, no relevant published information was found. Clinical adverse reactions: Mild to moderate anticholinergic symptoms, including dry mouth (10-15%), constipation (8-12%), and urinary retention (≤2%) [4] - The plasma protein binding of aprazole bromide is approximately 40-45% [3] |
| References |
|
| Additional Infomation |
Aclidinium bromide is a quaternary ammonium salt, the bromide of akleclidin. It is a muscarinic acetylcholine M3 receptor antagonist used for long-term maintenance therapy of bronchospasm associated with chronic obstructive pulmonary disease (COPD). It has a dual role as a muscarinic receptor antagonist and a bronchodilator. It is a quaternary ammonium salt and an organic bromide salt containing the akleclidin group.
See also: Akleclidin (containing the active moiety); Akleclidin bromide; Formoterol fumarate (component). Drug IndicationsEklira Genuair is indicated for maintenance bronchodilator therapy in adult patients with chronic obstructive pulmonary disease (COPD) to relieve symptoms. Bretaris Genuair is indicated for maintenance bronchodilator therapy in adult patients with chronic obstructive pulmonary disease (COPD) to relieve symptoms. Ackleidine bromide (LAS 34273; LAS-W 330) is a novel long-acting inhaled muscarinic acetylcholine receptor antagonist (LAMA) [1][2][3][4] - Its main mechanism of action is competitive antagonism of M3 muscarinic receptors in airway smooth muscle and epithelial glands, thereby inhibiting bronchoconstriction and excessive mucus secretion [1][2][4] - Its clinical indication is for long-term maintenance treatment of chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema [1][2][4] - Compared with other long-acting anticholinergic drugs (LAMA), it has higher selectivity for M3 receptors than M2 receptors, thereby reducing the risk of cardiovascular side effects [1][4] - It has been approved by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for the treatment of adult COPD, administered by inhalation twice daily [4] |
| Molecular Formula |
C26H30NO4S2.BR
|
|---|---|
| Molecular Weight |
564.55
|
| Exact Mass |
563.079
|
| CAS # |
320345-99-1
|
| Related CAS # |
320345-99-1 (bromide);727649-81-2 (cation);
|
| PubChem CID |
11519741
|
| Appearance |
Off-white to pink solid powder
|
| LogP |
1.629
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
7
|
| Rotatable Bond Count |
10
|
| Heavy Atom Count |
34
|
| Complexity |
647
|
| Defined Atom Stereocenter Count |
1
|
| SMILES |
C1C[N+]2(CCC1[C@H](C2)OC(=O)C(C3=CC=CS3)(C4=CC=CS4)O)CCCOC5=CC=CC=C5.[Br-]
|
| InChi Key |
XLAKJQPTOJHYDR-QTQXQZBYSA-M
|
| InChi Code |
InChI=1S/C26H30NO4S2.BrH/c28-25(26(29,23-9-4-17-32-23)24-10-5-18-33-24)31-22-19-27(14-11-20(22)12-15-27)13-6-16-30-21-7-2-1-3-8-21;/h1-5,7-10,17-18,20,22,29H,6,11-16,19H2;1H/q+1;/p-1/t20?,22-,27?;/m0./s1
|
| Chemical Name |
[(3R)-1-(3-phenoxypropyl)-1-azoniabicyclo[2.2.2]octan-3-yl] 2-hydroxy-2,2-dithiophen-2-ylacetate;bromide
|
| Synonyms |
LAS 34273, LAS W 330; LAS-34273, LAS-W 330; LAS34273, LASW 330; trade name: Eklira Genuair; Tudorza Genuair; Bretaris Genuair
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.43 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.43 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (4.43 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 30% PEG400+0.5% Tween80+5% Propylene glycol : 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7713 mL | 8.8566 mL | 17.7132 mL | |
| 5 mM | 0.3543 mL | 1.7713 mL | 3.5426 mL | |
| 10 mM | 0.1771 mL | 0.8857 mL | 1.7713 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
|
|---|
|
 |