
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
Verteporfin (also known as DB00460, CL318952, BPD-MA, BpdMA, Benzoporphyrin derivative monoacid ring A or BPD-MA) is a novel and potent second-generation photosensitizing agent derived from porphyrin in endothelial cells. It can be used for angiographic visualization of choroidal vessels and CNV.
| Targets |
YAP-TEAD interaction
Ph-positive acute lymphoblastic leukemia (ALL): No explicit IC₅₀/Ki values; Verteporfin (DB-00460; CL-318952; BPD-MA; BpdMA) exerts light-independent anti-leukemic activity, potentially associated with suppression of BCR-ABL signaling (no direct kinase inhibition demonstrated) [1] - YAP protein (in NSC 125973-resistant HCT-8/T cells): No explicit IC₅₀/Ki values; Verteporfin inhibits YAP expression and activity, reversing drug resistance [2] - Autophagic pathway (in pancreatic ductal adenocarcinoma, PDAC): No explicit IC₅₀/Ki values; Verteporfin acts as an autophagy inhibitor by reducing autophagy-related protein expression (e.g., LC3-II) [3] |
|---|---|
| ln Vitro |
PDX cell screening was used to specifically select verteporfin. PhLO, PhLH, and PhLK had GI50 values of 228 nM, 395 nM, and 538 nM, respectively, causing 50% growth inhibition. In contrast, ALL-1, TCC-Y/sr, and NPhA1 had GI50 values of 3.93 μM, 2.11 μM, and 5.61 μM, respectively. Two of the three PDX cells' sensitivity to verteporfin was significantly decreased by GSH. In PDX cells, verteporfin lowers the potential of the mitochondrial membrane [1]. By suppressing YAP expression, verteporfin lowers the PTX resistance of HCT-8/T cells. When Verteporfin and NSC 125973 are treated together, they have a synergistic effect that inhibits YAP and decreases HCT-8/T cytotoxicity [2].
Ph-positive ALL cells: 1. Proliferation inhibition: Verteporfin (0.5 μM–10 μM, light-independent) concentration-dependently inhibited growth of SUP-B15 and BV173 cells. IC₅₀ values (MTT assay, 72-hour treatment): ~2.5 μM (SUP-B15), ~3.0 μM (BV173). At 5 μM, cell viability reduced by ~60% (SUP-B15) and ~55% (BV173) vs. control [1] 2. Synergy with BMS-354825: Combining Verteporfin (2 μM) with BMS-354825 (1 μM) reduced SUP-B15 cell viability by ~80% (vs. ~30% with single agents), with a combination index (CI) <1 (synergism). Western blot showed p-BCR-ABL (Tyr412), p-STAT5, and p-AKT levels reduced by ~70%–80% [1] 3. Apoptosis induction: 5 μM Verteporfin (48-hour treatment) increased apoptotic rate of SUP-B15 cells from ~5% (control) to ~45% (Annexin V-FITC/PI staining, flow cytometry). Cleaved caspase-3 levels upregulated by ~3.5-fold (Western blot) [1] - YAP-overexpressing HCT-8/T cells: 1. Proliferation inhibition: Verteporfin (1 μM–20 μM, no photoactivation) inhibited NSC 125973-resistant HCT-8/T cell growth, IC₅₀ ~8.5 μM (MTT, 72 hours). At 10 μM, cell viability reduced by ~50% vs. control [2] 2. Resistance reversal: Verteporfin (10 μM) restored NSC 125973 sensitivity in HCT-8/T cells: IC₅₀ of NSC 125973 decreased from ~20 μM (resistant) to ~5 μM (combined). Western blot showed YAP protein levels reduced by ~65% [2] 3. Apoptosis induction: 10 μM Verteporfin + 5 μM NSC 125973 increased apoptotic rate by ~60% (vs. ~15% with NSC 125973 alone) [2] - Pancreatic cancer cells: 1. Proliferation inhibition: Verteporfin (2 μM–25 μM) inhibited PANC-1 and MIA PaCa-2 cell growth, IC₅₀ ~12 μM (PANC-1) and ~15 μM (MIA PaCa-2) (MTT, 72 hours) [3] 2. Autophagy inhibition: 10 μM Verteporfin (24-hour treatment) reduced LC3-II/LC3-I ratio by ~50% (Western blot) and autophagosome number by ~45% (immunofluorescence staining for LC3) in PANC-1 cells [3] 3. Synergy with gemcitabine: Verteporfin (5 μM) + gemcitabine (100 nM) reduced PANC-1 cell viability by ~75% (vs. ~30% with gemcitabine alone), with CI <1. Apoptotic rate increased by ~55% (vs. ~20% with gemcitabine alone) [3] |
| ln Vivo |
BMS-354825 and verteporfin (10 mg/kg, csc) both dramatically decreased the percentage of leukemia cells, and their combination therapy also decreased the total amount of leukemia cells in the spleen [1].
Using image-based and molecular analyses, we show that verteporfin inhibits autophagy stimulated by gemcitabine, the current standard treatment for PDAC. Pharmacokinetic and efficacy studies in a BxPC-3 xenograft mouse model demonstrated that verteporfin accumulated in tumors at autophagy-inhibiting levels and inhibited autophagy in vivo, but did not reduce tumor volume or increase survival as a single agent. In combination with gemcitabine verteporfin moderately reduced tumor growth and enhanced survival compared to gemcitabine alone. While our results do not uphold the premise that autophagy inhibition might be widely effective against PDAC as a single-modality treatment, they do support autophagy inhibition as an approach to sensitize PDAC to gemcitabine.[3] Nude mouse (nu/nu, 6–8 weeks old) PDAC xenograft model: 1. Grouping: Mice randomized into 4 groups (n=6/group): (1) Control (intraperitoneal injection of 5% DMSO + 95% normal saline); (2) Verteporfin 50 mg/kg; (3) Gemcitabine 25 mg/kg; (4) Verteporfin 50 mg/kg + gemcitabine 25 mg/kg [3] 2. Treatment: Tumors induced by subcutaneous injection of PANC-1 cells (5×10⁶ cells/mouse). When tumors reached ~100 mm³ (day 0), drugs administered intraperitoneally: Verteporfin twice weekly, gemcitabine once weekly, for 3 weeks [3] 3. Efficacy outcomes: - Tumor volume: Reduced by ~35% (Verteporfin alone), ~40% (gemcitabine alone), and ~70% (combination) vs. control at day 21; - Tumor weight: Decreased by ~30% (Verteporfin alone), ~35% (gemcitabine alone), and ~65% (combination) at sacrifice; - Tumor lysates: LC3-II/LC3-I ratio reduced by ~45% (Verteporfin alone) and ~60% (combination); cleaved caspase-3 upregulated by ~2.5-fold (combination) [3] |
| Enzyme Assay |
To study the relationship between YAP and PTX resistance, a stable YAP-over-expression or YAP silencing cell line was generated by transfected with YAP-plasmids or siYAP-RNA. WST-1 assay was performed to detect the cytotoxicity of PTX on HCT-8 and HCT-8/T cells. Clone formation assay and Transwell assay was preformed to determine the cell proliferation and invasion ability respectively. Immunofluorescence and Western blot analysis was performed for protein detection.[2]
Results: YAP was stronger expressed in HCT-8/T than in HCT-8, and PTX resistance was positively correlated with the level of YAP expression. VP, a strongly YAP inhibitor, could reduce the PTX resistance on HCT-8/T cells without light activation by inhibiting YAP. Beside, VP and PTX combination therapy showed synergism on inhibition of YAP and cytotoxicity to HCT-8/T. Moreover, verteporfin and PTX combination therapy affect the invasion and colony formation ability and induce apoptosis of HCT-8/T cells.[2] Conclusions: VP can reverse the PTX resistance induced by YAP over-expression in HCT-8/T cells without photoactivation through inhibiting YAP expression.[2] |
| Cell Assay |
In vitro effects of verteporfin on PDAC cell lines[3]
A panel of 8 human PDAC cell lines was exposed to 0-10µM verteporfin for up to 7 days and live cells were quantified using automated fluorescence microscopy to count nuclei or by measuring absorbance in an MTT assay. Four lines (Capan 1, Capan 2, HS766T, CFPAC-1) were insensitive to verteporfin at all concentrations. Panc-1 and MIA PaCa-2 cells grew normally in ≤ 5µM verteporfin, but cell proliferation was significantly inhibited in 10µM verteporfin. Notably, proliferation of BxPC-3 and SU86.86 cells was completely inhibited in 10µM verteporfin, and reduced by >50% in 5µM verteporfin.[3] Estimation of drug interactions[1] PDX cells co-cultured with S17 cells were treated with 16 combinations of verteporfin (60 nM, 120 nM, 180 nM, and 240 nM) and dasatinib (12 nM, 24 nM, 36 nM, and 48 nM). The viabilities of cells treated with each combination were measured after 48 h using FACS Aria flow cytometer. In order to estimate drug interaction between verteporfin and dasatinib, a normalized isobologram and fraction affected-combination index (CI) plot were made using CompuSyn software. CI values greater than 1.0 indicated antagonistic effects, equal to 1.0 additive effects, and below 1.0 synergistic effects.[1] Ph-positive ALL cell experiments: 1. MTT assay: SUP-B15/BV173 cells seeded in 96-well plates (5×10³ cells/well), treated with Verteporfin (0.5–10 μM) alone or + BMS-354825 (1 μM). Incubated 72 hours (37°C, 5% CO₂), MTT (5 mg/mL) added for 4 hours, DMSO dissolved formazan, absorbance measured at 570 nm. IC₅₀ and CI calculated [1] 2. Apoptosis assay: SUP-B15 cells (2×10⁵ cells/well, 6-well plates) treated with 5 μM Verteporfin for 48 hours. Cells harvested, washed with PBS, stained with Annexin V-FITC/PI for 15 minutes (dark), analyzed via flow cytometry [1] 3. Western blot: Cells treated as above, lysed with RIPA buffer (含 protease/phosphatase inhibitors). 30 μg protein separated by SDS-PAGE, transferred to PVDF membranes, probed with anti-p-BCR-ABL (Tyr412), p-STAT5, cleaved caspase-3, and β-actin antibodies. ECL detected signals [1] - HCT-8/T cell experiments: 1. Resistance reversal assay: HCT-8/T cells seeded in 96-well plates, treated with Verteporfin (1–20 μM) + NSC 125973 (0–20 μM). MTT assay performed as above to determine IC₅₀ changes [2] 2. Western blot: Cells treated with 10 μM Verteporfin for 24 hours, lysed, probed with anti-YAP, cleaved caspase-3, and β-actin antibodies [2] - Pancreatic cancer cell experiments: 1. Autophagy detection: PANC-1 cells seeded on coverslips, treated with 10 μM Verteporfin for 24 hours. Fixed with 4% paraformaldehyde, stained with anti-LC3 antibody (autophagosome marker) and DAPI. Autophagosomes counted via confocal microscopy [3] 2. MTT and Western blot: Performed as described in [1], with drugs replaced by Verteporfin (2–25 μM) and gemcitabine (100 nM); blotted antibodies included anti-LC3, cleaved caspase-3 [3] |
| Animal Protocol |
Mice: PhLO cells (1.0×107/mouse) are injected intravenously into 6-week-old male NOG mice, which are then treated with vehicle, verteporfin (140 mg/kg/day), dasatinib (20 mg/kg/day), and a combination of these drugs from days 22 to 28. Verteporfin is administered by continuous subcutaneous infusion (c.s.c.) using Alzet osmotic pumps. An intraperitoneal injection (i.p.) is performed for dasatinib. All mice are sacrificed on day 28 and the chimerism of leukemia cells is investigated by flow cytometer using an anti-human CD19 antibody and antimouse CD45 antibody. Blood concentrations of verteporfin are calculated by LCMS-2020.[1]
\n Estimation of in vivo drug effects[1] \nPhLO cells (1.0 × 10~7 /mouse) were injected intravenously into 6-week-old male NOG mice, which were then treated with vehicle, verteporfin (140 milligram (mg)/kilogram (kg)/day), dasatinib (20 mg/kg/day), and a combination of these drugs from days 22 to 28. Verteporfin was administered by continuous subcutaneous infusion (c.s.c.) using Alzet osmotic pumps. An intraperitoneal injection (i.p.) was performed for dasatinib. All mice were sacrificed on day 28 and the chimerism of leukemia cells was investigated by flow cytometer using an anti-human CD19 antibody and anti-mouse CD45 antibody. Blood concentrations of verteporfin were calculated by LCMS-2020.[1] \n Verteporfin efficacy studies[3] \nFemale Rag2M mice (20-25g) were inoculated subcutaneously in the centre of the lower back with 5 x 106 BxPC-3 or SU86.86 cells (1:1 RPMI:matrigel; 100µL volume; expressed as day 0). Tumors appeared within three weeks of implantation. Once the tumors were palpable, tumor growth was monitored by measuring tumor dimensions with digital calipers. When tumors reached 100-150mg (calculated according to the equation (length X width2)/2 converted to tumor weight in mg for each 1mm3), mice were randomized in groups of eight animals and treatment was initiated. \nVerteporfin was administered i.p. Monday, Wednesday, Friday for 4 weeks at a dose of 45mg/kg (injection volume 400µl/20g mouse). Gemcitabine was administered i.p. once weekly (Monday) for 4 weeks at 120mg/kg or 240mg/kg (injection volume 200µl/20g mouse). Groups treated with both verteporfin and gemcitabine received gemcitabine 6h after verteporfin administration. This time was selected because maximum verteporfin tumor levels were achieved 8h post-administration and maximum gemcitabine tumor levels were observed 2h post-administration. Animals in the control group were treated with the delivery vehicle DSPE-mPEG2000 at the same concentration and schedule as verteporfin. Care was taken to house animals treated with verteporfin in dark conditions until the morning after treatment because verteporfin is a photosensitizer and exposure to bright light could be harmful. A One-Way ANOVA with Tukey's multiple comparison test was used to compare differences in tumor growth. \n Pharmacokinetic studies of verteporfin DSPE-mPEG2000 micelles in BxPC-3 tumor-bearing mice[3] \nRag2M mice (20-25g; n=3) were inoculated subcutaneously with 5 x 106 BxPC-3 cells. Mice were injected i.p. with verteporfin at 45mg/kg when tumors were approximately 200-250mg. Mice were euthanized by CO2 inhalation, and blood and tumors were collected at 2, 8, 16 and 24h post administration of verteporfin. Plasma was prepared by centrifuging samples at 1000 x g for 15min at 4°C. Tumors were excised, rinsed in PBS, and snap-frozen in cryovials in liquid nitrogen and stored at -80°C. Tumors were sectioned while frozen and one half was used for determining verteporfin concentration by UPLC-MS/MS and the other for immunoblot analysis of p62 and LC3. \nPlasma samples were thawed, homogenized and extracted with acetonitrile containing 0.1% formic acid. Protein precipitation and filtration was carried out using ISOLUTE® PPT+ protein precipitation plates (Biotage). Samples were analyzed using a Waters® ACQUITY® UPLC system with mass spectrometry detection. Separations were performed using an isocratic method where mobile phase A was 0.1% formic acid in water and B was 0.1% formic acid in acetonitrile (70% B for 2.5min followed by 95% B for the wash). Verteporfin regioisomer A (monoacid A form) was eluted at 1.77min and regioisomer B (monoacid B form) was eluted at 2.16min with a total run time of 4.5min per sample. The MS/MS system was operated with an ESI interface in a positive ionization mode. Quantification was performed using multiple reactions monitoring (MRM) mode with a precursor mass m/z of 719.27 and product mass m/z of 645.36. The levels of verteporfin were measured against external calibration standards prepared using the same process. \n Formulation of verteporfin for animal studies[3] \nVerteporfin was formulated in DSPE-mPEG2000 micelles. Briefly, 200mg verteporfin dissolved in 2ml DMSO was slowly added, with stirring, to 1500mg DSPE-mPEG2000 dissolved in 50ml PBS at pH 7.4. Stirring was continued for 1h at 23°C followed by dialysis overnight against PBS using Spectra/Por dialysis membranes (15,000 MWCO). The concentration of verteporfin was measured in triplicate and quantified against an external standard curve using a Waters® ACQUITY® UPLC equipped with a PDA detector. Separations were done using a C18 column (Waters® BEH; column size 50 x 2.1mm, particle size 1.7µm) and a mobile phase of 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile:methanol (1:1; B) [10-80% B over 2min at 0.5ml/min flow rate]. The concentration of verteporfin was adjusted to 2.25mg/ml with PBS followed by filter-sterilization and sterile vialing of the formulation. The concentration was reconfirmed before proceeding with the animal studies. The formulation was chemically and physically stable for an observation period of 4 weeks, which covered the duration of the animals studies as verified by UPLC and polarized light microscopy. \nNude mouse PDAC xenograft protocol: \n 1. Animal housing: Female nude mice (6–8 weeks old, 18–22 g) housed in SPF facilities (22–25°C, 12-hour light/dark cycle) with free access to food/water [3] \n 2. Tumor implantation: PANC-1 cells (5×10⁶ cells/mouse) resuspended in 100 μL PBS/matrigel (1:1), subcutaneously injected into right flank of mice [3] \n 3. Grouping and treatment: When tumors reached ~100 mm³ (day 0), mice randomized into 4 groups: (1) Control: intraperitoneal injection of solvent (10 μL/g body weight); (2) Verteporfin: 50 mg/kg (dissolved in solvent), intraperitoneal, twice weekly; (3) Gemcitabine: 25 mg/kg, intraperitoneal, once weekly; (4) Combination: Verteporfin + gemcitabine (same doses/frequency). Treatment lasted 3 weeks [3] \n 4. Monitoring and analysis: Tumor volume measured every 3 days (volume = length × width² / 2). Mice euthanized via CO₂ inhalation, tumors excised/weighed. Tumor lysates prepared for Western blot (anti-LC3, cleaved caspase-3) [3] |
| Toxicity/Toxicokinetics |
In vitro toxicity: Human peripheral blood mononuclear cells (PBMCs) treated with 5 μM verteporfen for 72 hours: cell viability decreased by <15%, indicating low toxicity to normal cells [1]
- In vivo toxicity: Nude mice treated with verteporfen (50 mg/kg, intraperitoneal injection, twice a week for 3 weeks): 1. No significant weight loss (<5% vs. baseline) or death; 2. Serum ALT, AST, creatinine and BUN levels were within the normal range; 3. No pathological damage (histological examination) was observed in the liver, kidneys or spleen [3] |
| References |
|
| Additional Infomation |
benzo[b]porphyrin derivative for photochemotherapy in the treatment of wet macular degeneration. A synthetic photoactivator with photodynamic activity. After systemic administration, verteporfen accumulates in ocular neovascularization and, upon stimulation with non-thermal red light in the presence of oxygen, generates highly reactive short-lived singlet oxygen and other reactive oxygen free radicals, leading to localized damage to the neovascular endothelium and vascular occlusion. An equimolar mixture of the 9-methyl and 13-methyl esters of trans-(+-)-18-vinyl-4,4a-dihydro-3,4-bis(methoxycarbonyl)-4a,8,14,19-tetramethyl-23H,25H-benzo[b]porphyrin-9,13-dipropionic acid. It is used as a photosensitizer in photodynamic therapy to eliminate ocular abnormalities associated with neovascular (wet) age-related macular degeneration. Vertepofen accumulates in these abnormal blood vessels and, upon activation by red light (693 nm) laser in the presence of oxygen, generates highly reactive, short-lived singlet oxygen and other reactive oxygen species, leading to localized endothelial damage and vascular occlusion. Cell lines have been used as cancer models for drug discovery; however, they do not faithfully reproduce cancer, especially in terms of microenvironment independence. Patient-derived xenografts (PDXs), established by directly transplanting primary tumor cells from a patient into immunodeficient mice, can provide the required number of similar primary tumor cells at the desired time. We developed a high-throughput drug screening system using PDX cells and performed drug screening using PDX cells from Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL). We constructed four Ph+ ALL PDX mouse models and used leukemia cells from these PDX mice to perform high-throughput screening of 3440 compounds (PDX cell screening). The drug spectrum obtained from PDX cell screening differed significantly from that obtained from Ph+ ALL cell line screening. We found that the FDA-approved drug verteporfen exhibited strong PDX cell-specific cytotoxicity. In validation experiments, the GI50 values of verteporfen in the three PDX cell lines were 228 nM, 395 nM and 538 nM, respectively, and the GI50 values in the three cell lines were 3.93 µM, 2.11 µM and 5.61 µM, respectively. Although verteporfen is a photosensitizer that requires light activation, its cytotoxic effect is mediated by the generation of light-independent reactive oxygen species; therefore, its anti-leukemic effect can be exerted in vivo even without light. In addition, it showed synergistic effects with the ABL kinase inhibitor dasatinib. These results suggest that verteporfen has the potential to be a novel anti-leukemic drug. [1]
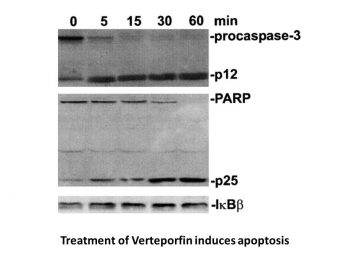
Paclitaxel (PTX) is one of the most effective anticancer drugs. However, multidrug resistance remains a major factor hindering the effective treatment of cancer with PTX. A variety of factors, including YAP overexpression, can lead to PTX resistance. This study aims to verify the role of YAP in PTX resistance, explore the method of reversing PTX resistance with verteporfen (VP), and investigate the effect of combined PTX and VP treatment on PTX-resistant colon cancer cells (HCT-8/T). [2] Pancreatic ductal adenocarcinoma (PDAC) is highly resistant to chemotherapy. Studies have shown that the growth of PDAC requires an increase in autophagy levels, so inhibiting autophagy is considered a treatment strategy. To date, all preclinical reports and clinical trials on the pharmacological inhibition of autophagy have used chloroquine or hydroxychloroquine, which interfere with lysosomal function and block autophagy in the later stages. Verteporfen is a newly discovered autophagy inhibitor that blocks autophagy in the early stages by inhibiting the formation of autophagosomes. This article reports that in vitro experiments showed that pancreatic ductal adenocarcinoma (PDAC) cell lines had different sensitivities to verteporfen, suggesting that cell lines have different dependence on autophagy. Through image analysis and molecular analysis, we found that verteporfen can inhibit gemcitabine (currently the standard treatment for PDAC)-induced autophagy. Pharmacokinetic and efficacy studies in the BxPC-3 xenograft mouse model showed that verteporfen accumulated in tumors to levels sufficient to inhibit autophagy and did so in vivo, but as monotherapy, it did not reduce tumor volume or prolong survival. When used in combination with gemcitabine, verteporfen moderately inhibited tumor growth and prolonged survival, superior to gemcitabine monotherapy. While our results do not support the premise that autophagy inhibition is a broadly effective monotherapy for pancreatic ductal adenocarcinoma (PDAC), they do support it as a method to enhance the sensitivity of PDAC to gemcitabine. [3] Verteporfin Traditionally, it is a photosensitizer for photodynamic therapy (PDT), but the literature mainly focuses on its light-independent biological activities: anti-leukemia, reversal of drug resistance and autophagy inhibition[1][2][3] - Mechanism of action: (1) In Ph-positive acute lymphoblastic leukemia (ALL): it inhibits the downstream signaling pathways of BCR-ABL (p-STAT5, p-AKT) to induce apoptosis and works synergistically with BMS-354825 (Src inhibitor)[1]; (2) In YAP-overexpressing cells: it reduces YAP protein expression to reverse NSC 125973 resistance[2]; (3) In pancreatic ductal adenocarcinoma: it inhibits autophagy (reduces LC3-II) to enhance the antitumor effect of gemcitabine[3] |
| Molecular Formula |
C41H42N4O8
|
|
|---|---|---|
| Molecular Weight |
718.79
|
|
| Exact Mass |
718.30
|
|
| Elemental Analysis |
C, 68.51; H, 5.89; N, 7.79; O, 17.81
|
|
| CAS # |
129497-78-5
|
|
| Related CAS # |
|
|
| Appearance |
Typically exists as Brown to black solid at room temperature
|
|
| LogP |
10.246
|
|
| SMILES |
[C@]12(C)[C@@H](C(=O)OC)C(C(=O)OC)=CC=C1C1=CC3=NC(C(C=C)=C3C)=CC3NC(=C(C=3C)CCC(=O)O)C=C3C(CCC(=O)OC)=C(C)C(=N3)C=C2N1.[C@]12(C)[C@@H](C(=O)OC)C(=CC=C1C1NC2=CC2C(C)=C(CCC(=O)O)C(=CC3NC(=C(C)C=3CCC(=O)OC)C=C3C(C=C)=C(C)C(=N3)C=1)N=2)C(=O)OC |c:26,t:16,40,54,73,84,99,109,&1:0,2,53,55|
|
|
| InChi Key |
YTZALCGQUPRCGW-MXVXOLGGSA-N
|
|
| InChi Code |
InChI=1S/C41H42N4O8/c1-9-23-20(2)29-17-34-27-13-10-26(39(49)52-7)38(40(50)53-8)41(27,5)35(45-34)19-30-22(4)25(12-15-37(48)51-6)33(44-30)18-32-24(11-14-36(46)47)21(3)28(43-32)16-31(23)42-29/h9-10,13,16-19,38,42,44H,1,11-12,14-15H2,2-8H3,(H,46,47)/b28-16-,29-17-,30-19-,31-16-,32-18-,33-18-,34-17-,35-19-/t38-,41+/m0/s1
|
|
| Chemical Name |
(1): 3-[(23S,24R)-14-ethenyl-5-(3-methoxy-3-oxopropyl)-22,23-bis(methoxycarbonyl)-4,10,15,24-tetramethyl-25,26,27,28-tetraazahexacyclo[16.6.1.13,6.18,11.113,16.019,24]octacosa-1,3,5,7,9,11(27),12,14,16,18(25),19,21-dodecaen-9-yl]propanoic acid.
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 5 mg/mL (6.96 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 5 mg/mL (6.96 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 50.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3912 mL | 6.9561 mL | 13.9123 mL | |
| 5 mM | 0.2782 mL | 1.3912 mL | 2.7825 mL | |
| 10 mM | 0.1391 mL | 0.6956 mL | 1.3912 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04590664 | Recruiting | Drug: Verteporfin | Glioblastoma Recurrent Glioblastoma |
Emory University | January 15, 2021 | Phase 1 Phase 2 |
| NCT03067051 | Recruiting | Drug: Verteporfin Device: SpectraCure P18 System |
Recurrent Prostate Cancer | SpectraCure AB | March 21, 2017 | Phase 1 Phase 2 |
| NCT00674323 | Completed Has Results | Drug: Verteporfin Photodynamic Therapy |
Polypoidal Choroidal Vasculopathy | Novartis | April 2008 | Phase 4 |
| NCT00433017 | Terminated Has Results | Drug: Verteporfin Photodynamic Therapy |
Macular Degeneration | Novartis | May 2007 | Phase 2 Phase 3 |
|
 |