
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
Tideglusib (formerly known as NP031112, NP12) is a novel, potent, irreversible, non-ATP-competitive and chemical inhibitor of GSK-3β (glycogen synthase kinase-3β) with potential neuroprotective effects and may be used as an anti-AD (Alzheimer disease) agent. Tideglusib is presently undergoing phase II clinical trials for Alzheimer's disease and progressive supranuclear palsy; it inhibits GSK-3 with an IC50 of 60 nM in cell-free assay. The lack of recovery in enzyme activity after unbound Tideglusib was removed from the reaction medium and the fact that its dissociation rate constant is not significantly different from zero are evidence that Tideglusib inhibits GSK-3 irreversibly. Additionally, tideglusib was unable to inhibit a number of kinases with an active site Cys homologous to Cys-199, indicating that its inhibition of GSK-3 is the result of a specific mechanism and not nonspecific reactivity.
| Targets |
GSK-3β (IC50 = 5 nM); GSK-3β (IC50 = 60 nM)
Glycogen Synthase Kinase 3β (GSK3β): The IC₅₀ value for irreversible inhibition of recombinant human GSK3β was 60 nM; no inhibitory activity against GSK3α (even at 10 μM) or other kinases (e.g., CDK2, ERK1, JNK1) was detected [1] |
|---|---|
| ln Vitro |
Tideglusib (NP031112) treatment completely eliminates the induction of TNF- and COX-2 expression after glutamate treatment in both astrocyte and microglial cultures. Because the 24-hour exposure of astrocyte and microglial cells to this TDZD has no effect on cell viability, these effects of NP031112 are not brought on by a reduction in cell viability[2].
1. For GSK3β inhibition: Incubation of recombinant human GSK3β with Tideglusib (NP-031112, NP-12) (100 nM for 1 hour) resulted in >90% inhibition of kinase activity, measured via [γ-³²P]-ATP incorporation into a peptide substrate. The inhibition was irreversible: after extensive dialysis (to remove unbound drug) for 24 hours, GSK3β activity remained suppressed by >80%, whereas reversible GSK3 inhibitors (e.g., CHIR99021) showed complete activity recovery post-dialysis. Mass spectrometry analysis confirmed covalent binding of Tideglusib (NP-031112, NP-12) to Cys¹⁹⁹ of GSK3β, a residue unique to GSK3β (absent in GSK3α) [1] 2. In primary rat cortical neurons (cultured for 14 days), pretreatment with Tideglusib (NP-031112, NP-12) (1 μM, 10 μM, 1 hour before excitotoxic insult) dose-dependently reduced glutamate-induced neuronal death. At 10 μM, neuron viability (MTT assay) increased from 42% (glutamate alone) to 78% (~86% protection). The drug also inhibited glutamate-induced activation of caspase-3 (Western blot: ~60% reduction at 10 μM) and DNA fragmentation (TUNEL staining: ~55% fewer positive cells at 10 μM) [2] 3. In primary rat microglia, Tideglusib (NP-031112, NP-12) (0.1 μM-10 μM, 24 hours) suppressed lipopolysaccharide (LPS)-induced release of pro-inflammatory cytokines: TNF-α secretion was reduced by ~70% at 10 μM, and IL-1β by ~65% at 10 μM (ELISA). It also inhibited LPS-induced upregulation of iNOS (Western blot: ~60% reduction at 10 μM) and NO production (Griess assay: ~58% reduction at 10 μM) [2] 4. In SH-SY5Y neuroblastoma cells, Tideglusib (NP-031112, NP-12) (1 μM-10 μM, 24 hours) protected against kainate-induced cell death: viability (MTT) increased from 38% (kainate alone) to 72% at 10 μM, accompanied by reduced intracellular reactive oxygen species (ROS) production (~45% reduction at 10 μM, DCFH-DA staining) [2] |
| ln Vivo |
Tideglusib (NP031112) (50 mg/kg) injection into the rat hippocampus significantly reduces kainic acid-induced inflammation as measured by edema formation using T2-weighted magnetic resonance imaging and glial activation and has a neuroprotective effect in the damaged areas of the hippocampus[2].
Coinjection of Tideglusib (NP031112), a more potent thiadiazolidinone derivative, into the rat hippocampus dramatically reduces kainic acid-induced inflammation, as measured by edema formation using T2-weighted magnetic resonance imaging and glial activation and has a neuroprotective effect in the damaged areas of the hippocampus. Last, NP031112-induced neuroprotection, both in vitro and in vivo, was substantially attenuated by cotreatment with GW9662 (2-chloro-5-nitrobenzanilide), a known antagonist of the nuclear receptor peroxisome proliferator-activated receptor gamma, suggesting that the effects of NP031112 can be mediated through activation of this receptor. As such, these findings identify NP031112 as a potential therapeutic agent for the treatment of neurodegenerative disorders.[2] 1. In male Wistar rats (250-300 g) with kainate-induced excitotoxicity (10 mg/kg, i.p. kainate injection), pretreatment with Tideglusib (NP-031112, NP-12) (10 mg/kg, i.p., 1 hour before kainate) reduced hippocampal neurodegeneration: Nissl staining showed ~60% fewer degenerating neurons in the CA3 region vs. vehicle. The drug also improved behavioral outcomes: kainate-induced seizures (scored via Racine scale) were reduced from grade 4.2 (vehicle) to grade 2.1, and latency to first seizure increased from 18 min to 45 min [2] 2. In the same rat model, Tideglusib (NP-031112, NP-12) (10 mg/kg, i.p., 1 hour pre-kainate) decreased hippocampal pro-inflammatory cytokine levels at 24 hours post-kainate: TNF-α was reduced by ~55%, IL-1β by ~50% (ELISA), and iNOS protein expression by ~45% (Western blot). It also inhibited microglial activation (Iba1 immunostaining: ~40% fewer activated microglia in CA3) [2] |
| Enzyme Assay |
[35S]Tideglusib (207 Bq/nmol) at 55 μM is incubated with 5 μM GSK-3β for 1 h at 25 °C in 315 μL of 50 mM Tris-HCl, pH 7.5, containing 150 mM NaCl and 0.1 mM EGTA. The incubation is extended for another 30 min after having added 35 μL of the same buffer with or without 100 mM DTE. Finally, a third 40-μL aliquot of each original sample is mixed with 10 μL of denaturing electrophoresis sample buffer without reducing agents, and 35 μL of this mixture is loaded onto a 10% polyacrylamide gel and subjected to SDS-PAGE, followed by fluorography of the dried gel. Finally, a third 40-L aliquot of each original sample is combined with 10 L of denaturing electrophoresis sample buffer without reducing agents, and 35 L of this mixture is loaded onto a 10% polyacrylamide gel and subjected to SDS-PAGE followed by fluorography of the dried gel.[1]
Evaluation of Inhibition on Kinase Panel[1] The inhibitory activities of Tideglusib (NP031112) and hypothemycin on a panel of selected kinases were evaluated in the Invitrogen European Screening Center. Compounds were tested in duplicate at a single concentration of 10 μm on a group of selected kinases, the enzymatic activity of which was measured using the Z′-LYTETM technology at ATP and peptide concentrations around their Km values, except for MEK1, MEK2, p38α, and JNK1, for which ATP was at 100 μm. In a few cases where it was not possible to monitor the activity of the kinases (MEK3, NIK, TAK1-TAB1, MNK2, NLK, and ZAK) the ability of the compounds to displace the binding of fluorescent analogues of known ATP-competitive inhibitors was measured using the time-resolved FRET-based LanthaScreenTM technology. Details on the nature of the kinases tested and the average of the results obtained in each case are presented in supplemental Table 1. 1. GSK3β kinase activity assay: Recombinant human GSK3β (10 ng) was incubated with a synthetic peptide substrate (YRRAAVPPSPSLSRHSSPHQpSEDEEE, 50 μM) in reaction buffer containing 20 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 1 mM DTT, and 10 μM [γ-³²P]-ATP. Tideglusib (NP-031112, NP-12) (0.1 nM-1 μM) was added, and the mixture was incubated at 30°C for 60 minutes. The reaction was terminated by spotting 20 μL onto phosphocellulose paper, which was washed 3 times with 1% phosphoric acid to remove unincorporated radioactivity. Radioactivity was measured via liquid scintillation counting, and IC₅₀ was calculated from dose-response curves [1] 2. Irreversible inhibition verification assay: After incubating GSK3β (10 ng) with Tideglusib (NP-031112, NP-12) (100 nM) for 1 hour, the mixture was dialyzed against reaction buffer (4°C, 24 hours, buffer changed every 6 hours) to remove unbound drug. GSK3β activity was then measured via the kinase assay above, with a reversible GSK3 inhibitor (CHIR99021, 100 nM) as control [1] 3. Covalent binding mass spectrometry assay: GSK3β (5 μg) was incubated with Tideglusib (NP-031112, NP-12) (1 μM) for 2 hours at 30°C, then digested with trypsin. Tryptic peptides were analyzed via liquid chromatography-tandem mass spectrometry (LC-MS/MS) to identify modified residues; a peptide containing Cys¹⁹⁹ with a mass shift corresponding to Tideglusib (NP-031112, NP-12) adduction was detected [1] |
| Cell Assay |
Rat primary astrocytes, microglia, and neurons were harvested and cultured as described previously (Luna-Medina et al., 2005). The purity of the cultures was >95%, as determined by immunofluorescence analysis using anti-cd11b (OX-42) to detect microglial cells, glial fibrillary acidic protein (GFAP) to identify astrocytes, and anti-microtubule-associated protein 2 (MAP2) to identify neurons. Tideglusib (NP031112) (2.5 μm) was added to the culture medium of astrocytes and microglia 1 h before exposure to glutamate (500 μm), cells were incubated for 24 h before tissue culture medium was collected, and the cells were evaluated for tumor necrosis factor-α (TNF-α) and cyclooxygenase type 2 (COX-2) expression. For transient transfection experiments, primary cultures of astrocytes were transfected with the reporter plasmid pPPRE-tk-luc, containing three PPARγ consensus binding sites upstream of a minimal promoter using Transfast according to the manufacturer's guidelines. Typically, cells received 0.2 μg of luciferase reporter plasmid and were harvested 24 h after treatment with different concentrations of Tideglusib (NP031112) for determination of luciferase and β-galactosidase (to determine transfection efficiency) activities. Each transient transfection experiment was repeated at least three times in triplicate.[1]
Measurement of apoptosis. To calculate the extent of apoptotic cell death, cortical neuronal cultures were treated or not with Tideglusib (NP031112) and incubated with glutamate (100 μm), and phosphatydilserine exposure on the surface of apoptotic cells was detected by confocal microscopy after staining with Annexin V-FITC. Neuronal cell death was assessed by counting the percentage of Annexin-V-positive cells in four independent high-magnification (200×) fields per culture, as described above.[1] 1. Primary cortical neuron excitotoxicity assay: Cortices from E18 rat embryos were dissected, dissociated, and plated at 5×10⁴ cells/well in 96-well plates (poly-L-lysine-coated). Cells were cultured in Neurobasal medium supplemented with B27 and glutamine for 14 days. On day 14, medium was replaced with serum-free medium, and Tideglusib (NP-031112, NP-12) (0.1 μM-10 μM) was added 1 hour before glutamate (100 μM, excitotoxic insult). After 24 hours, MTT reagent was added (0.5 mg/mL), incubated at 37°C for 4 hours, dissolved in DMSO, and absorbance was measured at 570 nm to calculate viability [2] 2. Microglial inflammation assay: Primary microglia were isolated from P1-P3 rat brains, plated at 1×10⁵ cells/well in 24-well plates, and cultured in DMEM + 10% FBS for 7 days. Cells were serum-starved for 2 hours, then treated with Tideglusib (NP-031112, NP-12) (0.1 μM-10 μM) 1 hour before LPS (100 ng/mL). After 24 hours, supernatant was collected for TNF-α/IL-1β ELISA and NO Griess assay; cells were lysed for iNOS Western blot (primary antibody against iNOS, β-actin as loading control) [2] 3. Neuronal apoptosis assay: Primary cortical neurons (14 days in vitro) were treated with Tideglusib (NP-031112, NP-12) (10 μM) 1 hour before glutamate (100 μM). After 24 hours, cells were fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, and stained with TUNEL reagent. TUNEL-positive cells were counted under a fluorescence microscope (10 random fields/well) [2] |
| Animal Protocol |
Rats; In this study, adult male Wistar rats (8–12 weeks old) are used. Rats (n≥5) are put into a stereotaxic machine. The hippocampus is given an injection of KA (1 μg in 2.5 μL l PBS) alone or in conjunction with Tideglusib (2 ng in 2.5 μL PBS). Animals in the control group are given vehicle injections and are the same age.
KA administration.[2] Adult male Wistar rats (8–12 weeks old) were used in this study. Adequate measures were taken to minimize pain or discomfort of animals. Experiments were performed in accordance with the European Communities Council, directive 86/609/EEC. Rats (n ≥ 5 per group) were anesthetized by intraperitoneal injection of ketamine (60 mg/kg) and Domtor (5 μg/kg) and placed into a stereotaxic apparatus. KA (1 μg in 2.5 μl PBS) alone or in combination with Tideglusib (NP031112) (2 ng in 2.5 μl PBS) was injected into the hippocampus [coordinates from bregma: posterior, −3.0 mm; lateral, −2.0 mm; depth, 3.5 mm; according to the atlas of Paxinos and Watson (1998)]. Control animals of the same age were injected with vehicle. Two groups of animals also received 0.7 μg of the PPARγ antagonist GW9662 (2-chloro-5-nitrobenzanilide), either alone or in combination with KA. Each injection was performed for >2.5 min using a micropump. The amounts of NP031112 and GW9662 used were calculated based on the in vitro results to reach active concentrations within the hippocampus. Lithium chloride (LiCl), a potent inhibitor of GSK-3β activity, was administered (40 mg/kg/d) by intraperitoneal injection to a further two groups of animals, either alone or in combination with KA. The rats were then housed individually to recover.[2] Seizures were induced by intraperitoneal administration of rats with KA (10 mg/kg) in PBS. Control animals received saline only. Behavioral analysis was monitored for a period of 3 h by trained observers blind to the treatment of the rats. The convulsive behavior was classified according to Racine (1972) and Sperk et al. (1985) as follows: stage 0, no changes; stage 0.5, wet dog shakes (WDS); stage 1, mouth and facial movements; stage 2, head nodding; stage 3, forelimbs clonus; stage 4, rearing; stage 5, rearing and falling; stage 6, death. Status epilepticus (SE) was defined as continuous behavioral seizure activity (stage 5) for ≥5 min. The number of WDS before SE was also examined. In trials using Tideglusib (NP031112), the TDZD was administered intragastrically (50 mg/kg) 1 h before KA injection. 1. Rat kainate excitotoxicity model: Male Wistar rats (250-300 g) were randomly divided into 3 groups (n=8/group): vehicle (0.9% saline + 5% DMSO, i.p.), Tideglusib (NP-031112, NP-12) (10 mg/kg, i.p., dissolved in 0.9% saline + 5% DMSO), and kainate alone (10 mg/kg, i.p.). The drug or vehicle was administered 1 hour before kainate injection. Seizure activity was monitored for 4 hours post-kainate, scored using the Racine scale (0-5). At 24 hours post-kainate, rats were euthanized; brains were harvested, fixed in 4% paraformaldehyde, paraffin-embedded, and sectioned (5 μm) for Nissl staining (hippocampal CA3 region) and Iba1 immunostaining. Hippocampal tissue was also dissected for cytokine ELISA and Western blot [2] |
| Toxicity/Toxicokinetics |
1. In vitro experiments showed that Tideglusib (NP-031112, NP-12) at concentrations up to 10 μM had no cytotoxicity to primary cortical neurons, microglia, or SH-SY5Y cells (cell viability >90% as detected by MTT assay, compared with the control group)[2]
2. In vivo experiments showed that 24 hours after intraperitoneal injection of 10 mg/kg Tideglusib (NP-031112, NP-12) in rats, there were no significant changes in body weight, serum ALT/AST (liver function), or creatinine (kidney function) compared with the solvent group. During the 24-hour monitoring period, no obvious toxic symptoms (such as drowsiness, ataxia, abnormal hair combing) were observed[2] |
| References |
|
| Additional Infomation |
Tideglusib, a thiadiazolidine compound, is chemically named 1,2,4-thiadiazolidine-3,5-dione, with a naphth-1-yl substitution at position 2 and a benzyl substitution at position 4. It is a non-ATP-competitive glycogen synthase kinase 3β (GSK3β) inhibitor with neuroprotective effects. Clinical trials are currently underway for Alzheimer's disease and progressive supranuclear palsy. It possesses multiple functions, including acting as an EC 2.7.11.26 (tau protein kinase) inhibitor, neuroprotective agent, anti-inflammatory agent, and apoptosis inducer. It belongs to the naphthalene, benzene, and thiadiazolidine classes of compounds. Tideglusib is being investigated for development into a drug for the treatment of Alzheimer's disease and progressive supranuclear palsy. Tideglusib has been reported as a potent anti-inflammatory and neuroprotective agent, belonging to the non-ATP-competitive inhibitors of glycogen synthase kinase 3 (GSK-3). Tideglusib was developed by the Spanish pharmaceutical company Zeltia Group, but its approval for the treatment of Alzheimer's disease was revoked in 2012.
Drug Indications Tideglusib was initially developed for the treatment of Alzheimer's disease and progressive supranuclear palsy. Growing interest in Tideglusib stems from the significant upregulation of GSK-3 expression in the brains of Alzheimer's patients. Tideglusib also functions importantly as a β-catenin degrader, as it inhibits the transcription of cell survival genes. All these factors have prompted current research to consider this kinase as a potential target. Alzheimer's disease is the most common type of dementia. The most widely accepted hypothesis explaining the disease is related to the presence of β-amyloid protein, which triggers a cascade of reactions that alter Tau protein, leading to synaptic dysfunction and neuronal death. The importance of GSK-3 in tissue repair pathways also provides a new potential application for Tideglusib. Therefore, it is also being investigated for its potential to treat the natural repair of deep caries lesions. Mechanism of Action GSK-3 is a proline/serine protein kinase widely expressed in various cells and involved in multiple cell signaling pathways. Among its many functions, GSK-3 plays a crucial role in Alzheimer's disease. This role is related to its association with β-amyloid and tau protein pathology. Studies have shown that abnormal Wnt or insulin signaling pathways lead to enhanced GSK-3 function. This kinase acts on γ-secretase, leading to hyperphosphorylation of tau protein, which in turn forms neurofibrillary tangles and senile plaques. Tideglusib irreversibly inhibits GSK-3 through a non-competitive inhibition mode of ATP. The binding of Tideglusib appears to be directly related to motifs containing Cys199. Pharmacodynamics Tideglusib administration has been reported to inhibit the activation of astrocytes and microglia, thereby exerting a neuroprotective effect. Furthermore, inactivation of GSK-3 is known to prevent excitotoxicity. Preclinical trials have reported reduced Tau protein hyperphosphorylation, decreased amyloid plaque burden in the brain, enhanced learning and memory, prevention of neuronal loss, and a significant increase in insulin-like growth factor 1 (a potent neurotrophic peptide with therapeutic value). Clinical trial reports show that Alzheimer's patients receiving 24 weeks of treatment exhibited an upward trend in cognitive function. 1. Tideglusib (NP-031112, NP-12) is an irreversible GSK3β inhibitor that works by covalently binding to the Cys¹⁹⁹ residue of GSK3β—a residue absent in GSK3α, which explains its strict selectivity for GSK3β relative to GSK3α[1] 2. The neuroprotective effect of Tideglusib (NP-031112, NP-12) in excitotoxicity is mediated by two mechanisms: (1) inhibition of GSK3β-dependent neuronal apoptosis (reducing caspase-3 activation and ROS generation); (2) inhibition of microglia activation and release of pro-inflammatory cytokines (inhibition of the NF-κB pathway, downregulation of iNOS)[2] 3. Tideglusib (NP-031112, NP-12) It has shown potential in treating brain diseases associated with excitotoxicity and neuroinflammation, such as epilepsy, Alzheimer's disease and stroke [2] 4. Unlike reversible GSK3 inhibitors, the irreversibility of Tideglusib (NP-031112, NP-12) may result in a lower frequency of administration in vivo, as it continues to target GSK3β even after the free drug is cleared [1] |
| Molecular Formula |
C19H14N2O2S
|
|---|---|
| Molecular Weight |
334.3917
|
| Exact Mass |
334.077
|
| Elemental Analysis |
C, 68.25; H, 4.22; N, 8.38; O, 9.57; S, 9.59
|
| CAS # |
865854-05-3
|
| Related CAS # |
865854-05-3
|
| PubChem CID |
11313622
|
| Appearance |
White to off-white solid powder
|
| Density |
1.4±0.1 g/cm3
|
| Boiling Point |
511.3±43.0 °C at 760 mmHg
|
| Melting Point |
148-150ºC
|
| Flash Point |
263.0±28.2 °C
|
| Vapour Pressure |
0.0±1.3 mmHg at 25°C
|
| Index of Refraction |
1.735
|
| LogP |
3.28
|
| Hydrogen Bond Donor Count |
0
|
| Hydrogen Bond Acceptor Count |
3
|
| Rotatable Bond Count |
3
|
| Heavy Atom Count |
24
|
| Complexity |
492
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
O=C1N(CC2C=CC=CC=2)C(=O)N(C2C3C(=CC=CC=3)C=CC=2)S1
|
| InChi Key |
PMJIHLSCWIDGMD-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C19H14N2O2S/c22-18-20(13-14-7-2-1-3-8-14)19(23)24-21(18)17-12-6-10-15-9-4-5-11-16(15)17/h1-12H,13H2
|
| Chemical Name |
4-benzyl-2-naphthalen-1-yl-1,2,4-thiadiazolidine-3,5-dione
|
| Synonyms |
Tideglusib; NP031112, NP-12; NP-12; NP031112; Tideglusib [INN]; 4-Benzyl-2-(naphthalen-1-yl)-1,2,4-thiadiazolidine-3,5-dione; NP-031112; tideglusibum; NP031112; NP 031112; NP-031112
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO: ~66 mg/mL (~197.4 mM)
Water: <1 mg/mL Ethanol: <1 mg/mL |
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.48 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.48 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. View More
Solubility in Formulation 3: 4% DMSO+corn oil: 2.5mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9905 mL | 14.9526 mL | 29.9052 mL | |
| 5 mM | 0.5981 mL | 2.9905 mL | 5.9810 mL | |
| 10 mM | 0.2991 mL | 1.4953 mL | 2.9905 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Status | Interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT05004129 | Recruiting | Drug: Tideglusib | Congenital Myotonic Dystrophy | AMO Pharma Limited | August 23, 2021 | Phase 2 Phase 3 |
| NCT05105958 | Not yet recruiting | Drug: Tideglusib | Amyotrophic Lateral Sclerosis | University Hospital, Geneva | December 1, 2025 | Phase 2 |
| NCT02858908 | Completed | Drug: Tideglusib | Myotonic Dystrophy 1 | AMO Pharma Limited | July 20, 2016 | Phase 2 |
| NCT01350362 | Completed | Drug: tideglusib Drug: Placebo |
Alzheimer's Disease | Noscira SA | April 2011 | Phase 2 |
| NCT00948259 | Completed | Drug: NP031112 Drug: Placebo |
Alzheimer´s Disease | Noscira SA | December 2008 | Phase 1 Phase 2 |
Inhibition of PI3K pathway signaling in cells. KPL-4 cells were treated with the indicated concentrations of CH5132799 for 2 hours. Clin Cancer Res, 2011, 17(10), 3272-3281. |
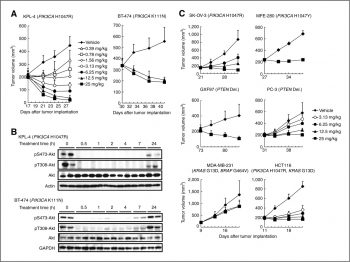 Antitumor activity in mouse xenograft models of cell lines harboring genetic alterations, including PIK3CA mutations |
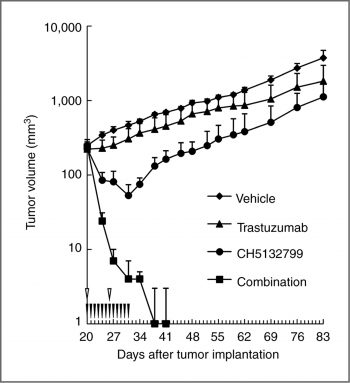 Antitumor activity in combination with trastuzumab in the trastuzumab-insensitive model. |