| Size | Price | Stock | Qty |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 100mg | |||
| 250mg | |||
| Other Sizes |
| Targets |
TSPO/mitochondrial translocator protein
|
|---|---|
| ln Vitro |
In SHSY-5Y neuroblastoma cells, 4'-chlorodiazepam has neuroprotective properties against amyloid-β (Aβ). In organotypic hippocampal preparations, Aβ decreases cell viability but 4'-chlordiazepam, at doses of 100nM and 1000nM, is neuroprotective. The upregulation of superoxide dismutase (SOD) expression is associated with the neuroprotective effects of 4'-chlordiazepam on Aβ [1]. In glucose-deprived cells, 4'-clozazepam decreases nuclear fragmentation and maintains cell viability. In cells treated with 4'-chlordiazepam, these effects are accompanied by a decrease in the formation of free radicals and the preservation of mitochondrial function [2].
|
| Cell Assay |
The translocator protein (TSPO) is an outer mitochondrial membrane protein involved in the transport of cholesterol into the mitochondria, which is the first step for the synthesis of steroid hormones, as well as in the regulation of mitochondrial permeability transition pore opening and apoptosis. Studies have shown that the activation of TSPO may promote neuroprotective actions in experimental models of neurodegeneration and brain injury. In a previous study, our group showed that 4'-chlorodiazepam (4'-CD), a TSPO ligand, was neuroprotective against amyloid-beta (Aβ) in SHSY-5Y neuroblastoma cells. The aim of this study was to evaluate if 4'-CD was also neuroprotective against Aβ in organotypic hippocampal cultures and to identify its mechanisms of action. Aβ decreased the cell viability of organotypic hippocampal cultures, while 4'-CD had a neuroprotective effect when administered at 100nM and 1000nM. The neuroprotective effects of 4'-CD against Aβ were associated with an increased expression of superoxide dismutase (SOD). No differences were found in the expression of catalase, glial fibrillary acidic protein, Akt and procaspase-3. In summary, our results show that 4'-CD is neuroprotective against Aβ by a mechanism involving the modulation of SOD protein expression[1].
The translocator protein (TSPO), formerly known as the peripheral-type benzodiazepine receptor (PBR), is considered an important regulator of steroidogenesis and a potential therapeutic target in neurological disorders. Previous evidence suggests that TSPO ligands can protect cells during injury and prevent apoptosis in central nervous system (CNS) cells. However, its actions on astrocytic cells under metabolic injury are not well understood. In this study, we explored whether 4'-chlorodiazepam (Ro5-4864), a TSPO ligand, might protect astrocyte mitochondria under glucose deprivation. Our results showed that 4'-chlorodiazepam preserved cell viability and reduced nuclear fragmentation in glucose-deprived cells. These effects were accompanied by a reduced production of free radicals and maintenance of mitochondrial functions in cells treated with 4'-chlorodiazepam. Finally, our findings suggest that TSPO might be involved in reducing oxidative stress by preserving mitochondrial functions in astrocytic cells exposed to glucose withdrawal[2]. |
| References |
[1]. B D Arbo, et al. 4'-Chlorodiazepam is neuroprotective against amyloid-beta in organotypic hippocampal cultures. J Steroid Biochem Mol Biol. 2017 Jul;171:281-287.
[2]. Eliana Baez, et al. 4'-Chlorodiazepam Protects Mitochondria in T98G Astrocyte Cell Line from Glucose Deprivation. Neurotox Res. 2017 Aug;32(2):163-171. |
| Additional Infomation |
Mechanism of Action
Mitochondria isolated from rat brains were found to cleave cholesterol to produce pregnenolone (a precursor to steroid hormones) at an average rate of 21.0 pmol pregnenolone·mg protein·min⁻¹. This rate-limiting step in steroid synthesis can be significantly stimulated by PK 11195 (1-(2-chlorophenyl)-N-methyl-(1-methylpropyl)-3-isoquinoline carboxamide) and Ro5 4864 (4'-chlorodiazepine), both of which bind to peripheral benzodiazepine receptors with high affinity. Low-affinity peripheral benzodiazepine receptor ligands, such as Ro15 1788 (ethyl-8-fluoro-5,6-dihydro-5-methyl-6-oxo-4H-imidazo[1,5α][1,4]benzo-3-carboxylate) and clonazepam, have no significant effect on the rate of pregnenolone synthesis. Furthermore, the potency ranking of these compounds as [3H]Ro5 4864 binding inhibitors was consistent with their potency ranking in steroidogenesis. Since the 86-amino acid peptide diazepam binding inhibitor is also thought to bind to peripheral benzodiazepine receptors, the ability of four fragments of this peptide, a random sequence, and a steroidogenesis-activating peptide to interact with peripheral benzodiazepine receptors and stimulate steroidogenesis in rat brain mitochondria was also evaluated. The steroidogenesis-activating peptide and two fragments of the diazepam binding inhibitor significantly stimulated pregnenolone biosynthesis. Unlike peripheral benzodiazepine receptor ligands, no correlation was observed between the potency of peptide-substituted [3H]Ro5 4864 binding and steroidogenesis. Peripheral benzodiazepine receptors mediate cholesterol transport between the outer and inner mitochondrial membranes in steroid-producing tissues. They are also present in many other tissues, including the liver. We investigated the effects of peripheral benzodiazepine receptor ligands PK11195 [1-(2-chlorophenyl)-N-methyl-N-(1-methylpropyl)isoquinoline-3-carboxamide], Ro 5-4864 (4-chlorodiazepine), heme, protoporphyrin IX, and N-methylprotoporphyrin IX on intramural cholesterol transport in rat liver mitochondria. PK11195 and N-methylprotoporphyrin IX significantly increased the transport of endogenous cholesterol from the outer mitochondrial membrane to the inner membrane (140% and 150%, respectively, at 1 μM, P < 0.01). 5 μM protoporphyrin IX, 1 μM Ro 5-4864, and 5 μM heme had no such effect. When mitochondria were labeled with exogenous [4-14C] cholesterol, PK11195 and N-methylprotoporphyrin IX were most effective in increasing total cholesterol incorporation and cholesterol transport to the inner membrane, and their effects were dose-dependent. These data suggest that in the liver, binding to peripheral benzodiazepine receptors is associated with cholesterol transport, and the interaction between ligands and these receptors may play a role in the complex regulatory mechanisms of cholesterol transport between the liver mitochondrial membranes. The effects of various benzodiazepines were investigated in isolated uteruses from ovariectomized rats. These uteruses had been pre-treated with different estrogen groups (estrogen, progesterone, and estrogen + progesterone) for extended periods. All groups of uteruses exhibited spontaneous rhythmic activity. Spontaneous activity observed in the control uteruses was inhibited in a concentration-dependent manner by diazepam, 4'-chlorodiazepam, clonazepam, or 1-(2-chlorophenyl)-N-methyl-N-(1-methylpropyl)-3-isoquinoline carboxamide (PK 11195), or completely disappeared in [Ca2+]O-free solution. Diazepam, 4'-chlorodiazepam, clonazepam, and PK 11195 all dilated the uterus in a concentration-dependent manner after pre-contraction with [K+]o, with the potency order being: PK 11195 > 4'-chlorodiazepam > diazepam > clonazepam. Administration of [Ca2+]o (1 μM to 10 mM) induced concentration-dependent contractions of the uterus in saline solutions without [Ca2+]o prepared in different pretreatment groups. Incubation with different concentrations (μM) of diazepam, 4'-chlorodiazepam, clonazepam, and PK 11195 reduced the contractile response of the rat uterus to extracellular calcium ion concentration ([Ca2+]o) in all groups. These results indicate the existence of micromolar-level benzodiazepine binding sites in the rat uterus. Diazepam, 4'-chlorodiazepam, clonazepam, and PK 11195 all induce uterine relaxation in pre-contracted rats, an effect likely related to inhibition of extracellular calcium ion influx ([Ca2+]o). The vasodilatory effects of different benzodiazepines observed in this study can be modulated by pretreatment with different estrogens. This study also determined the interaction between the atypical benzodiazepine 4'-chlorodiazepam (Ro 5-4864) and functionally expressed human GABAA receptor cDNA. Co-transfection of human α2, β1, and γ2 subunits reconstructed the 4'-chlorodiazepam recognition site, manifested as a dose-dependent enhancement of the binding of t-[35S]butylbicyclic thiophosphate ([35S]-TBPS) to GABA-activated chloride channels. This site is located on the GABAA receptor complex, which contains binding sites for GABA agonist-like benzodiazepines and neuroactive steroids. The importance of the α subunit is further confirmed because, under the same experimental conditions, replacing the α2 subunit with the α1 or α3 subunits did not reconstruct the 4'-chlorodiazepine recognition site that regulates [35S]TBPS binding. The regulatory site of 4'-chlorodiazepine differs from that of benzodiazepines, but the effects of phenylquinoline compounds PK 8165 and PK 9084 are similar to those of 4'-chlorodiazepine, consistent with previous analyses of the 4'-chlorodiazepine site in brain homogenates. Further analysis of subunit requirements showed that co-expression of only the α2 and β1 subunits was sufficient to reconstruct the 4'-chlorodiazepine recognition site. Interestingly, however, the study found that the 4'-chlorodiazepine site can inhibit the binding of [35S]TBPS to GABA-activated chloride channels. Therefore, the 4'-chlorodiazepine site can likely be reconstructed from only the α and β subunits. For more complete data on the mechanisms of action of the six 4'-chlorodiazepines, please visit the HSDB record page. |
| Molecular Formula |
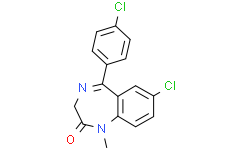
C16H12CL2N2O
|
|---|---|
| Molecular Weight |
319.185
|
| Exact Mass |
318.033
|
| Elemental Analysis |
C, 60.21; H, 3.79; Cl, 22.21; N, 8.78; O, 5.01
|
| CAS # |
14439-61-3
|
| PubChem CID |
1688
|
| Appearance |
Typically exists as white to light yellow solids at room temperature
|
| Density |
1.35g/cm3
|
| Boiling Point |
517.1ºC at 760mmHg
|
| Melting Point |
160-163ºC
|
| Flash Point |
266.6ºC
|
| Vapour Pressure |
8.43E-11mmHg at 25°C
|
| Index of Refraction |
1.647
|
| LogP |
3.307
|
| Hydrogen Bond Donor Count |
0
|
| Hydrogen Bond Acceptor Count |
2
|
| Rotatable Bond Count |
1
|
| Heavy Atom Count |
21
|
| Complexity |
432
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
ClC1C([H])=C([H])C2=C(C=1[H])C(C1C([H])=C([H])C(=C([H])C=1[H])Cl)=NC([H])([H])C(N2C([H])([H])[H])=O
|
| InChi Key |
PUMYFTJOWAJIKF-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C16H12Cl2N2O/c1-20-14-7-6-12(18)8-13(14)16(19-9-15(20)21)10-2-4-11(17)5-3-10/h2-8H,9H2,1H3
|
| Chemical Name |
7-chloro-5-(4-chlorophenyl)-1-methyl-3H-1,4-benzodiazepin-2-one
|
| Synonyms |
RO-5-4864; RO 5-4864; Chlordiazepam; RO5-4864; 4-Chlorodiazepam; Ro 5-4864; 2H-1,4-Benzodiazepin-2-one, 7-chloro-5-(4-chlorophenyl)-1,3-dihydro-1-methyl-; Ro-05-4864; RO5-4864
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~313.29 mM)
|
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.83 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1329 mL | 15.6647 mL | 31.3293 mL | |
| 5 mM | 0.6266 mL | 3.1329 mL | 6.2659 mL | |
| 10 mM | 0.3133 mL | 1.5665 mL | 3.1329 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.