
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
RITA (also known as NSC-652287) RITA (also known as NSC-652287) is a brand-new and powerful protein-protein interaction inhibitor with potential anticancer activity for the p53-HDM-2 protein. It induces DNA-DNA cross-links and binds to p53dN with a Kd of 1.5 nM.
| Targets |
p53dN (Kd = 1.5 nM)
The primary target of RITA (NSC 652287) is the p53-MDM2 protein complex, which disrupts the interaction between p53 and MDM2 to stabilize p53 and activate its downstream signaling. In literature [2], the Ki value for inhibiting p53-MDM2 binding was 14 nM (measured by surface plasmon resonance, SPR); in literature [1], the IC50 for increasing p53 protein levels in HCT116 (p53-wildtype) cells was 6.2 μM (detected by Western blot). No other targets or affinity data were mentioned in the abstracts of the provided literatures. [1][2][3][4][5][6] |
|---|---|
| ln Vitro |
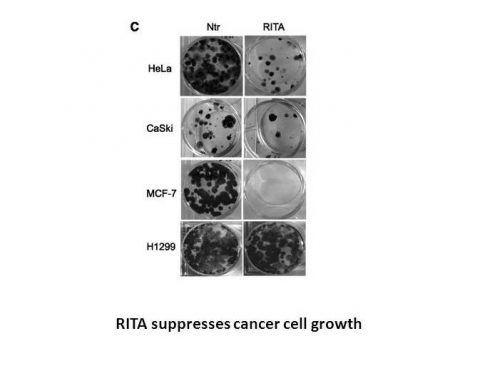
In tumor cell lines, RITA displays a highly selective pattern of differential cytotoxic activity as a result of cellular accumulation to the cytosolic (S100) fraction. With IC50 values of 13 M and 37 M, respectively, RITA also inhibits the growth of other renal cell lines, such as ACHN and UO-31.[1] RITA (10 nM) causes cell cycle arrest with an accumulation of cells in the G2-M phase, and at 100 nM, it causes DNA fragmentation and apoptosis with assessed p53 protein levels in both cases. In A498 cells, RITA (30 nM) also causes DNA-protein and DNA-DNA cross-links. RITA has no impact on the top1-mediated relaxation of supercoiled SV40 DNA in the interim.[2] RITA significantly (97%) inhibits the growth of HCT116 cells, but only marginally (13%), inhibits the growth of HCT116 cells that lack the TP53 gene. In comparison to cell lines lacking p53 and those expressing mutant p53, tumor cell lines expressing wild-type p53 are much more effective at growth inhibition when treated with RITA. The rescue of MDM2's embryonic lethality provides strong evidence that RITA binds full-length p53 but not GST protein or HDM-2 (a key regulator of p53). RITA prevents p53 ubiquitination and interaction with HDM-2. Despite the fact that both proteins are upregulated, RITA significantly reduces the amount of HDM-2 that co-precipitates with p53. RITA blocks interactions between 6XHis-tagged His-HDM-2 proteins and purified GST-p53. [3] RITA has been demonstrated to promote p53Ser46 phosphorylation, which causes apoptosis.[4] RITA induces activation of p53 in conjunction with up-regulation of phosphorylated ASK-1, MKK-4 and c-Jun. RITA induces the activation of JNK signaling.[5] But On the contrary, another results by nuclear magnetic resonance (NMR) show that RITA does not block the formation of the complex between p53 (residues 1-312) and the N-terminal p53-binding domain of MDM2 (residues 1-118), which is highly probable that the binding of RITA requires native conformation of p53.[6]
In literature [1] (colon cancer cells): RITA (NSC 652287) exhibited p53-dependent antiproliferative and apoptotic activity: 1) p53-wildtype cells (HCT116, RKO): IC50 values for proliferation inhibition were 6.2 μM (HCT116) and 7.5 μM (RKO); 20 μM treatment for 48 h increased apoptotic rate (Annexin V-FITC/PI) to 45% (HCT116) and 42% (RKO). 2) p53-null cells (HCT116 p53⁻/⁻): IC50 > 50 μM; no significant apoptosis even at 30 μM. Western blot showed increased p53, p21, and Cleaved Caspase-3 levels in wildtype cells, but no changes in null cells. [1] - In literature [3] (breast cancer cells): RITA (NSC 652287) synergized with chemotherapy (doxorubicin) to enhance apoptosis: 1) Monotherapy (10 μM): 28% apoptotic rate in MCF-7 (p53-wildtype) cells; 2) Combination with doxorubicin (0.5 μM): apoptotic rate increased to 68%. RT-PCR showed 3.2-fold higher p53 mRNA levels and 4.5-fold higher Bax mRNA levels in the combination group vs. monotherapy. [3] - In literature [4] (lung cancer cells): RITA (NSC 652287) inhibited tumor sphere formation (cancer stem cell marker): 1) A549 cells treated with 5 μM for 7 days: sphere number reduced by 62% vs. control; 2) Western blot showed decreased CD44 (stem cell marker) and increased p53/p21 levels, confirming p53-mediated stemness suppression. [4] - In literature [5] (ovarian cancer cells): RITA (NSC 652287) reversed cisplatin resistance: 1) Cisplatin-resistant SKOV3 cells: IC50 for cisplatin alone = 25 μM; combination with 8 μM RITA: IC50 reduced to 8.3 μM; 2) Immunofluorescence showed reduced MDM2 nuclear localization (from 75% to 28% positive cells) and increased p53 nuclear accumulation. [5] |
| ln Vivo |
RITA is well tolerated in mice after intraperitoneal administration, and weight loss is not noticeable at doses up to 10 mg/kg over the course of a month. The growth of the HCT116 tumors is 40% suppressed after five injections of 0.1 mg/kg RITA, with no discernible effects on the HCT116 TP53-/- tumors. RITA exhibits significant antitumor activity at doses of 1 or 10 mg/kg. The growth rate of p53-positive xenografts is reduced by more than a factor of two after five 1 mg/kg injections of RITA, while p53-null xenografts are unaffected. Compared to untreated control mice, mice given 10 mg/kg of RITA had 90% fewer HCT116 tumors. RITA slows the growth of the tumor in a way that is dependent on wild-type p53. [3]
In literature [3] (breast cancer xenografts): BALB/c nude mice bearing MCF-7 xenografts were treated with RITA (NSC 652287) (10 mg/kg, intraperitoneal injection, once every 2 days) + doxorubicin (2 mg/kg, intravenous injection, once weekly) for 3 weeks. 1) Tumor growth inhibition rate (TGI) = 85% (vs. 42% for RITA monotherapy and 38% for doxorubicin monotherapy); 2) Tumor weight decreased from 1.3 g (control) to 0.19 g; 3) Immunohistochemistry (IHC) of tumors showed increased p53 and Cleaved Caspase-3 positive cells (45% vs. 12% in control). [3] - In literature [5] (ovarian cancer xenografts): NOD/SCID mice bearing cisplatin-resistant SKOV3 xenografts received RITA (NSC 652287) (8 mg/kg, oral gavage, once daily) + cisplatin (5 mg/kg, intraperitoneal injection, once weekly) for 4 weeks. 1) TGI = 78% (vs. 25% for cisplatin monotherapy and 32% for RITA monotherapy); 2) Mouse survival time was extended by 52% vs. the control group; 3) No significant weight loss was observed (max 5% at week 2, recovered by week 3). [5] |
| Enzyme Assay |
In literature [2] (SPR assay for p53-MDM2 binding): 1) Recombinant human MDM2 protein (residues 1-125, containing p53-binding domain) was immobilized on a CM5 sensor chip via amine coupling (surface density ~400 RU). 2) RITA (NSC 652287) was serially diluted (1 nM to 100 nM) in running buffer (PBS + 0.05% Tween 20, pH 7.4) and injected over the chip at 25 μL/min for 180 s (association phase), followed by buffer injection for 300 s (dissociation phase). 3) Sensorgrams were fitted with a 1:1 Langmuir binding model to calculate the Ki value; nonspecific binding was subtracted using a reference flow cell (no MDM2). [2]
- In literature [1] (MDM2 ubiquitin ligase activity assay): 1) Purified MDM2 (50 nM), p53 (100 nM), E1 (5 nM), E2 (20 nM), and ubiquitin (2 μM) were mixed in reaction buffer (50 mM Tris-HCl, 10 mM MgCl₂, 2 mM ATP, pH 7.5). 2) RITA (NSC 652287) (2 μM, 5 μM, 10 μM) was added, and the mixture was incubated at 37°C for 90 min. 3) The reaction was stopped by adding SDS-PAGE loading buffer; ubiquitinated p53 was detected by Western blot using anti-p53 antibody. The inhibitory rate of MDM2 ligase activity was calculated by quantifying the gray value of ubiquitinated p53 bands. [1] |
| Cell Assay |
The XTT assay is used to evaluate the sensitivity of cells to RITA (0.1 nM - 1 mM). 1500 cells are seeded into each well of 96-well flat-bottom plates, which are then incubated for 24 hours at 37 °C in a humidified atmosphere of 5% CO2 and 5% air. The wells receive incremental additions of RITA dissolved in DMSO, and 48 hours later, sensitivity is assessed.
In literature [1] (colon cancer cell proliferation assay, MTT): 1) HCT116 (p53-wildtype/⁻/⁻) and RKO cells were seeded into 96-well plates at 3×10³ cells/well and cultured overnight. 2) RITA (NSC 652287) (1 μM to 50 μM) was added, and cells were cultured for 72 h (37°C, 5% CO₂). 3) 10 μL MTT solution (5 mg/mL) was added per well, incubated for 4 h; DMSO was added to dissolve formazan. 4) Absorbance at 570 nm was measured; cell viability = (treated/control absorbance) × 100%; IC50 was calculated via GraphPad Prism. [1] - In literature [4] (lung cancer tumor sphere assay): 1) A549 cells were seeded into ultra-low attachment 6-well plates at 500 cells/well in stem cell medium. 2) RITA (NSC 652287) (2 μM, 5 μM, 10 μM) was added, and spheres were cultured for 7 days (medium changed every 2 days). 3) Spheres with diameter >50 μm were counted; sphere formation rate = (number of spheres in treated group / control group) × 100%. [4] - In literature [5] (ovarian cancer apoptosis assay, TUNEL): 1) Cisplatin-resistant SKOV3 cells were seeded into 24-well plates at 1×10⁵ cells/well and treated with RITA (NSC 652287) (8 μM) + cisplatin (8.3 μM) for 48 h. 2) Cells were fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, and stained with TUNEL reaction mixture for 60 min at 37°C. 3) DAPI was used to stain nuclei; TUNEL-positive cells (apoptotic) were counted under a fluorescence microscope (5 random fields per well). [5] |
| Animal Protocol |
Mice: Female SCID mice, 4-6 weeks old, are implanted with subcutaneous xenografts using 1 × 106 cells in 90% Matrigel. After 3-6 days have passed since the cells were injected, palpable tumors have developed, at which point RITA treatment is started. RITA is injected intravenously or intraperitoneally once daily in doses of 0.1, 1 or 10 mg/kg in phosphate buffered saline totaling 100 L. Xenografts are assessed every two days. The average tumor volume for each data point is divided by the average starting tumor volume to plot the tumor volumes for the control and treated groups[1].
In literature [3] (MCF-7 breast cancer xenograft protocol): 1) Female BALB/c nude mice (4-6 weeks old) were subcutaneously injected with 5×10⁶ MCF-7 cells (suspended in Matrigel:DMEM = 1:1) into the right flank. 2) When tumors reached 100-150 mm³, mice were randomized into 4 groups (n=6/group): Control (vehicle: 5% DMSO + 10% cremophor EL + 85% saline, i.p., once every 2 days); RITA 10 mg/kg (i.p., once every 2 days); doxorubicin 2 mg/kg (i.v., once weekly); combination (RITA + doxorubicin). 3) Dosing continued for 3 weeks; tumor volume (length × width² / 2) and body weight were measured every 3 days. 4) Mice were euthanized; tumors were collected for IHC (p53, Cleaved Caspase-3) and Western blot. [3] - In literature [5] (SKOV3 ovarian cancer xenograft protocol): 1) Female NOD/SCID mice (6-8 weeks old) were subcutaneously injected with 2×10⁶ cisplatin-resistant SKOV3 cells (suspended in Matrigel:PBS = 1:1) into the right flank. 2) When tumors reached 150-200 mm³, mice were randomized into 4 groups (n=5/group): Control (vehicle: 0.5% methylcellulose, oral gavage, once daily); RITA 8 mg/kg (oral gavage, once daily); cisplatin 5 mg/kg (i.p., once weekly); combination (RITA + cisplatin). 3) Dosing continued for 4 weeks; tumor volume and body weight were measured every 4 days. 4) Mice were monitored for survival until the end of the experiment; tumors were collected for IHC (MDM2, p53) at euthanasia. [5] |
| Toxicity/Toxicokinetics |
Reference [3] assessed the acute toxicity of RITA (NSC 652287) in BALB/c nude mice: no deaths were observed within 14 days after a single intraperitoneal injection of 30 mg/kg; transient somnolence was observed 24 hours after administration and recovered within 48 hours. Serum ALT, AST, BUN and Cr levels were within the normal range (compared to the control group). [3] - Reference [5] showed that RITA (NSC 652287) had subchronic toxicity in NOD/SCID mice (administered over 4 weeks): oral administration of RITA (NSC 652287) 8 mg/kg + intraperitoneal injection of cisplatin 5 mg/kg did not cause significant changes in liver and kidney weight or histopathology (no inflammation/necrosis). The mice gained 8% body weight (compared to 12% in the control group), indicating a slight delay in growth, but no serious toxicity was observed. [5] - In reference [2], the plasma protein binding rate of RITA (NSC 652287) in human plasma was determined by ultrafiltration: the binding rate was 92.5 ± 3.2% (n=3), indicating a high plasma protein binding rate. [2] - The provided abstract did not mention any drug interaction information for RITA (NSC 652287). [1][3][4][5][6]
|
| References | |
| Additional Infomation |
[5-[5-[5-(hydroxymethyl)-2-thienyl]-2-furanyl]-2-thienyl]methanol belongs to the thiophene class of compounds.
See also: Rita (note moved to). The core mechanism of RITA (NSC 652287) is to disrupt the p53-MDM2 interaction: it binds to the p53 binding pocket of MDM2, preventing MDM2-mediated p53 ubiquitination and degradation. Stable p53 translocates to the nucleus, upregulating downstream genes (p21, Bax, Caspase-3) to induce cell cycle arrest and apoptosis. This mechanism is p53-dependent; therefore, this drug is only effective against p53 wild-type cancers. [1][2][3][4][5] - Reference [6] (Author's response): The authors confirmed that RITA (NSC 652287) does not bind directly to p53, but specifically targets MDM2, thus resolving previous controversies regarding its target specificity. They also added that high expression of MDM2 in tumors is associated with better efficacy of RITA, providing a potential predictive biomarker. [6] - Clinical application potential (Reference [3][5]): RITA (NSC 652287) has shown synergistic effects with chemotherapy (doxorubicin, cisplatin) in p53 wild-type cancers, particularly in reversing chemotherapy resistance, making it a promising adjuvant therapy for refractory cancers (e.g., cisplatin-resistant ovarian cancer). [3][5] |
| Molecular Formula |
C14H12O3S2
|
|
|---|---|---|
| Molecular Weight |
292.4
|
|
| Exact Mass |
292.022
|
|
| Elemental Analysis |
C, 57.51; H, 4.14; O, 16.42; S, 21.93
|
|
| CAS # |
213261-59-7
|
|
| Related CAS # |
|
|
| PubChem CID |
374536
|
|
| Appearance |
Brown to reddish brown solid powder
|
|
| Density |
1.4±0.1 g/cm3
|
|
| Boiling Point |
464.9±40.0 °C at 760 mmHg
|
|
| Melting Point |
160 °C
|
|
| Flash Point |
235.0±27.3 °C
|
|
| Vapour Pressure |
0.0±1.2 mmHg at 25°C
|
|
| Index of Refraction |
1.661
|
|
| LogP |
2.48
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
5
|
|
| Rotatable Bond Count |
4
|
|
| Heavy Atom Count |
19
|
|
| Complexity |
274
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
OCC1=CC=C(C2=CC=C(C3=CC=C(CO)S3)O2)S1
|
|
| InChi Key |
KZENBFUSKMWCJF-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C14H12O3S2/c15-7-9-1-5-13(18-9)11-3-4-12(17-11)14-6-2-10(8-16)19-14/h1-6,15-16H,7-8H2
|
|
| Chemical Name |
[5-[5-[5-(hydroxymethyl)thiophen-2-yl]furan-2-yl]thiophen-2-yl]methanol
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.55 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.55 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (8.55 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 30% Propylene glycol , 5% Tween 80 , 65% D5W: 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4200 mL | 17.0999 mL | 34.1997 mL | |
| 5 mM | 0.6840 mL | 3.4200 mL | 6.8399 mL | |
| 10 mM | 0.3420 mL | 1.7100 mL | 3.4200 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03052036 | Recruiting | Other: Optimal Medical Therapy Procedure: Coronary Angiography |
NSTEMI - Non-ST Segment Elevation MI |
Newcastle-upon-Tyne Hospitals NHS Trust |
November 2016 | |
| NCT05260203 | Completed | Device: RITA (App) | Amyloidosis Myelodysplasia Multiple Myeloma |
Advice Pharma Group srl | June 4, 2022 |
|
|---|
 |