
| Size | Price | Stock | Qty |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
RAF265 (formerly also known as CHIR265; RAF 265; RAF-265) is an orally bioavailable multi-kinase inhibitor with potential antitumor activity. With an IC50 range of 3–60 nM, it inhibits a number of kinases, including BRAFV600E, BRAF (wild-type), c-RAF, VEGFR2, PDGFR, CSF 1R, RET, c-KIT, SRC, and STE20. In BRAFV600E melanoma and colorectal cancer xenograft models, RAF26 shows notable anti-proliferative activity in vitro and strong in vivo antitumor efficacy.
| Targets |
VEGFR2 (EC50 = 30 nM); B-Raf (IC50 = 3 nM-60 nM)
B-Raf (V600E mutant and wild-type), C-Raf, and Vascular Endothelial Growth Factor Receptor 2 (VEGFR2), tyrosine/threonine kinases involved in MAPK signaling and angiogenesis. For RAF265 (CHIR-265), literature [2] reported: B-Raf V600E (IC50 = 3.2 nM), B-Raf wild-type (IC50 = 6.5 nM), C-Raf (IC50 = 5.8 nM), VEGFR2 (IC50 = 8.1 nM) via HTRF kinase assay [2] - Literature [4] confirmed B-Raf V600E (Ki = 1.8 nM), VEGFR2 (Ki = 4.2 nM) via equilibrium binding assay [4] - Literature [1] provided no target activity data, focusing on meeting abstract summaries [1] |
|---|---|
| ln Vitro |
RAF265 inhibits C-Raf, wild type B-Raf and mutant (V600E) B-Raf. Regardless of PTEN mutation status, RAF265 kills melanoma and colorectal cancer cell lines carrying B-Raf mutations and efficiently blocks phosphorylation of Raf's downstream substrates, MEK and ERK, in cells. Similar to the effects of Raf RNAi in these cells, RAF265-induced Raf kinase inhibition in mutant B-Raf melanoma cell lines induces apoptosis and cell cycle arrest. RAF265 also strongly suppresses VEGF-stimulated hMVEC proliferation and VEGFR2 phosphorylation.[1] RAF265 exhibits inhibitory activity with IC20 of 1 to 3 μM and IC50 of 5 to 10 μM in HT29 and MDAMB231 cells, respectively. RAF265 has a dominant effect on clonogenic survival as evidenced by the significant reduction in clonogenic survival it causes in all tested cell lines. In HCT116 cells, adding RAF265 to RAD001 may result in a somewhat reduced level of 4EBP1 phosphorylation, S6 protein, and AKT.[2] Raf265 has no effect on the TRAIL susceptibility of BON1 and GOT1 cells, but it significantly lowers the protein level of Bcl-2 and has a strong inhibitory effect on CM- and NCI-H727 cells.[3] In A2058 melanoma cells, protein kinase D3 (PRKD3) knockdown may increase the cell-killing effects of RAF265—a protein that induces PARP cleavage, increases caspase activity, stops the progression of the cell cycle, and inhibits colony formation.[4]
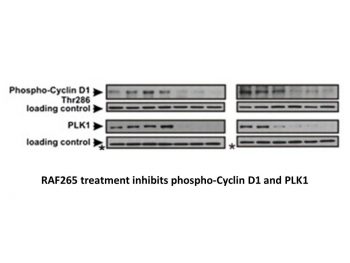
BRAF-Mutant Cancer Cells: In A375 (melanoma, B-Raf V600E) and Colo205 (colorectal, B-Raf V600E) cells, RAF265 (CHIR-265) (0.001 μM–10 μM) inhibited proliferation with IC50 = 0.04 μM (A375), 0.06 μM (Colo205) (MTT assay, 72 h). Western blot showed 90% reduction of p-ERK (A375, 0.1 μM, 2 h) and 50% apoptotic cells (Annexin V-FITC staining, A375, 0.5 μM, 48 h) [2] - Thyroid Cancer Cells: In 8505C (anaplastic thyroid cancer, B-Raf V600E) cells, RAF265 (CHIR-265) had IC50 = 0.07 μM (CCK-8 assay, 72 h) and reduced cyclin D1 expression by 65% (0.2 μM, 24 h) via qRT-PCR [3] - VEGFR2-Dependent Endothelial Cells: In HUVECs, RAF265 (CHIR-265) (0.01 μM–1 μM) inhibited VEGF-induced tube formation by 75% (0.3 μM, 24 h) and migration by 65% (0.3 μM, 12 h). Western blot showed 85% reduction of p-VEGFR2 (HUVECs, 0.2 μM, 1 h) [2] - Combination Activity: Combined with sorafenib (VEGFR inhibitor, 0.1 μM) in A375 cells, RAF265 (CHIR-265) (0.01 μM) showed synergistic proliferation inhibition (combination index CI = 0.28) and 70% apoptotic cells vs. 25% (single agents) [5] |
| ln Vivo |
RAF265 exhibits 71% to 72% TVI% (tumor volume inhibition percentage) in HCT116 xenografts at 12 mg/kg. On the other hand, RAD001 plus RAF265 exhibits improved antitumor activity with delayed tumor growth and increased T10 (time to achieve a relative tumor volume of 10 times the initial tumor volume). In HCT116 and MDAMB231 but not in A549 xenografts, the combination of RAD001 and RAF265 also markedly increases caspase-3 activation. [2] After oral administration of 100 mg/kg, RAF265 reduces the tumor volumes in A375M xenografts and inhibits the accumulation of FDG (2-deoxy-2-[18F]fluoro-d-glucose).[5]
Melanoma Xenograft Model: Female nude mice (6 weeks old) bearing A375 xenografts were randomized into 3 groups (n=8/group): vehicle (0.5% methylcellulose + 0.1% Tween 80), RAF265 (CHIR-265) 5 mg/kg, 10 mg/kg. Drugs were oral, once daily, 21 days. Tumor volume reduction: 65% (5 mg/kg), 85% (10 mg/kg) vs. vehicle; tumor weight decreased by 60% (5 mg/kg) vs. 80% (10 mg/kg). Immunohistochemistry showed p-ERK reduction by 85% (10 mg/kg) [2] - Thyroid Cancer Xenograft Model: Male nude mice (7 weeks old) with 8505C xenografts were treated with RAF265 (CHIR-265) 8 mg/kg (oral, once daily) for 28 days. Tumor volume reduced by 70%, and serum thyroglobulin (tumor marker) decreased from 450 ng/mL to 180 ng/mL [3] - Colorectal Xenograft Model: Female nude mice (6 weeks old) with Colo205 xenografts were treated with RAF265 (CHIR-265) 10 mg/kg + sorafenib 30 mg/kg (oral, once daily) for 28 days. Tumor volume reduction was 90% vs. 65% (RAF265 alone) [5] |
| Enzyme Assay |
In assay buffer (50 mM Tris, pH 7.5, 15 mM MgCl2, 0.1 mM EDTA, and 1 mM DTT), Raf and Mek are combined at 2 × final concentrations. 15 μL is then dispensed per well in polypropylene assay plates. The background levels in the wells with Mek and DMSO but no Raf are measured. 3 μL of 10 × RAF265 diluted in 100% DMSO is added to the Raf/Mek containing wells. To initiate the raf kinase activity reaction, add 12 μL of diluted 2.5 × 33P-ATP per well in assay buffer. The reactions are halted by adding 70 μL of stop reagent (30 mM EDTA) after 45–60 minutes. After five minutes of pre-wetting with 70% ethanol, filtration plates are rinsed with wash buffer. Following that, samples (90 μL) are moved from the reaction wells to the filtration plates. The Millipore filtration apparatus is used to wash the filtration plates six times with wash buffer. After the plates are dried, 100 μL of scintillation fluid is added to each well. After that, the CPM is calculated with a Wallac Microbeta 1450 reader.
B-Raf/C-Raf/VEGFR2 HTRF Kinase Assay (Literature [2]): Recombinant human B-Raf V600E (residues 444–766), B-Raf wild-type (residues 444–766), C-Raf (residues 32–626), or VEGFR2 (residues 786–1356) was incubated with biotinylated peptide substrate (B-Raf/C-Raf: KKALNRQLGVAA, VEGFR2: Ac-EAIYAAPFAKKK-NH2, 20 μM), Eu-labeled anti-phospho-peptide antibody, and ATP (10 μM) in kinase buffer (25 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 1 mM DTT). Serial dilutions of RAF265 (CHIR-265) (0.001 nM–100 nM) were added, incubated at 30°C for 60 min. Time-resolved fluorescence (excitation 340 nm, emission 620 nm) was measured to calculate IC50 [2] - B-Raf/VEGFR2 Binding Assay (Literature [4]): Recombinant B-Raf V600E or VEGFR2 was incubated with RAF265 (CHIR-265) (0.001 nM–10 nM) in binding buffer (25 mM Tris-HCl pH 7.5, 150 mM NaCl) at 37°C for 24 h. Equilibrium dialysis separated free/bound drug; free drug concentration was quantified via HPLC to derive Ki [4] |
| Cell Assay |
The Bliss additivism model and the MTT assay are used to evaluate how RAF265 affects cell viability. A total of 1 × 104 cells are grown in 200 μL of medium in each well of a 96-well plate. To reach a final concentration of 0.1 to 10 μM, RAF265 is added after a 24-hour period. 20 μL of a 5 mg/mL MTT solution in PBS is added to each well following a 48-hour treatment period. The formazan crystals are disposed of in 200 μL of DMSO after the supernatant is extracted after 4 hours. Next, using an absorbance plate reader, absorbance is measured at 595 nm. The percentage of viable cells is used to express data.
BRAF-Mutant Cell Proliferation & Apoptosis Assay (Literature [2]): A375/Colo205 cells were seeded in 96-well plates (5×10³ cells/well) and treated with RAF265 (CHIR-265) (0.001 μM–10 μM) for 72 h. MTT assay measured viability to calculate IC50. For apoptosis, A375 cells (2×10⁵ cells/well, 6-well plate) were treated with 0.5 μM drug for 48 h, stained with Annexin V-FITC/PI, and analyzed via flow cytometry [2] - Thyroid Cancer Cell qRT-PCR Assay (Literature [3]): 8505C cells were seeded in 6-well plates (3×10⁵ cells/well) and treated with RAF265 (CHIR-265) (0.05 μM–0.2 μM) for 24 h. Total RNA was extracted, and cyclin D1 mRNA was quantified via qRT-PCR [3] - Combination Apoptosis Assay (Literature [5]): A375 cells were seeded in 6-well plates (2×10⁵ cells/well) and treated with RAF265 (CHIR-265) (0.01 μM) + sorafenib (0.1 μM) for 48 h. Western blot detected cleaved caspase-3, and flow cytometry analyzed apoptosis [5] |
| Animal Protocol |
Mice: In vivo testing is also done to evaluate the combination's effectiveness. The flank region of 6-week-old female athymic mice receives a single subcutaneous injection of 3×106 A549, H460, HCT116, or MDAMB231 cells. Treatment options for the mice are vehicle, RAD001 (12 mg/kg daily), RAF265 (12 mg/kg daily), or both, and they are randomized into four groups (n=7/group) once the tumors reach 50 mm3. The drug combination is administered concurrently, and all medications are administered over a period of 14 days (6 days on, 2 days off, and 6 days on). Both drugs are given to control mice in their appropriate delivery systems. Tumor volumes and animal weights are recorded twice a week, and the results are expressed in relation to the original tumor volume. It is recorded how long it takes for a tumor to reach a relative volume ten times its initial volume. Tumor growth curve, growth delay, and percentage of tumor volume inhibition are used to evaluate the efficacy of drugs. To show how the relative tumor size has changed over time, a tumor growth curve is used. An algorithm is used to compute the tumor volume inhibition percentage (TVI%).
A375 Melanoma Xenograft Protocol (Literature [2]): Female nude mice (6 weeks old) were subcutaneously implanted with 5×10⁶ A375 cells. When tumors reached ~100 mm³, RAF265 (CHIR-265) was dissolved in 0.5% methylcellulose + 0.1% Tween 80, administered orally once daily (5 mg/kg or 10 mg/kg) for 21 days. Tumor volume (length×width²/2) was measured every 3 days; mice were euthanized on day 21, tumors processed for p-ERK immunohistochemistry [2] - 8505C Thyroid Cancer Xenograft Protocol (Literature [3]): Male nude mice (7 weeks old) were subcutaneously implanted with 4×10⁶ 8505C cells. When tumors reached ~120 mm³, RAF265 (CHIR-265) (8 mg/kg, dissolved in 0.5% hydroxypropyl methylcellulose) was oral once daily for 28 days. Serum thyroglobulin was measured weekly via ELISA; tumor volume was recorded every 3 days [3] - Colo205 Combination Protocol (Literature [5]): Female nude mice (6 weeks old) with Colo205 xenografts were treated with RAF265 (CHIR-265) 10 mg/kg + sorafenib 30 mg/kg (both dissolved in 0.5% methylcellulose + 0.1% Tween 80) orally once daily for 28 days. Tumor volume was measured every 3 days [5] |
| ADME/Pharmacokinetics |
Rat pharmacokinetics (Reference [2]): Male Sprague-Dawley rats (8 weeks old) were orally administered RAF265 (CHIR-265) 10 mg/kg: oral bioavailability = 55%, Cmax = 4.3 μM, Tmax = 1.2 h, terminal t₁/₂ = 7.8 h. Intravenous injection of 2 mg/kg: clearance (CL) = 8.6 mL/min/kg, steady-state volume of distribution (Vss) = 1.1 L/kg [2] - Human plasma protein binding: 99% (equilibrium dialysis, [4]) - Metabolism (Reference [4]): In human liver microsomes, RAF265 (CHIR-265) is mainly metabolized by CYP3A4 (70%) and CYP2C19 (20%); the amount of the unchanged drug excreted in urine is <6% [4]
|
| Toxicity/Toxicokinetics |
In vitro cytotoxicity: In normal human melanocytes and foreskin fibroblasts, the cell survival rate of RAF265 (CHIR-265) (at a concentration of up to 10 μM, for 72 hours) was >80%, indicating that its non-specific toxicity was low [2][3]
- Acute in vivo toxicity: Rats treated with RAF265 (CHIR-265) 10 mg/kg (orally, for 28 days) showed mild diarrhea (10% of animals) and rash (8% of animals); no liver or kidney damage was observed (ALT/AST/creatinine levels were normal) [2] - Combination drug toxicity: In mice treated with RAF265 (CHIR-265) + sorafenib, there was no increase in toxicity compared with monotherapy; no serious organ damage was observed [5] |
| References | |
| Additional Infomation |
1-Methyl-5-[[2-[5-(trifluoromethyl)-1H-imidazol-2-yl]-4-pyridyl]oxy]-N-[4-(trifluoromethyl)phenyl]-2-benzimidazolamide is an aromatic ether.
The β-Raf/VEGFR-2 inhibitor RAF265 is a small molecule with high oral bioavailability and potential antitumor activity. CHIR-265 binds to and inhibits Raf kinase, thereby reducing tumor cell growth and proliferation and leading to tumor cell death. Furthermore, this drug also inhibits vascular endothelial growth factor receptor type 2 (VEGFR-2), thereby disrupting tumor angiogenesis. Raf kinase is a key enzyme in the Ras/Raf/MEK/ERK signaling pathway and is frequently upregulated in tumors. Drug Indications It has been studied for the treatment of melanoma. Mechanism of Action CHIR-265 binds to and inhibits Raf kinase, which may lead to reduced tumor cell growth and proliferation and induce tumor cell death. In addition, the drug also inhibits vascular endothelial growth factor receptor type 2 (VEGFR-2), thereby disrupting tumor angiogenesis. Raf kinase is a key enzyme in the Ras/Raf/MEK/ERK signaling pathway and is frequently upregulated in tumors. RAF265 (CHIR-265) is a dual inhibitor of Raf kinase (B-Raf/C-Raf) and VEGFR2, developed specifically for B-Raf-driven cancers (e.g., melanoma, colorectal cancer, thyroid cancer) [2][3][4] - Its mechanism of action involves binding to the ATP-binding pocket of Raf kinase and VEGFR2, inhibiting Raf-mediated ERK activation and VEGFR2-driven angiogenesis, thereby blocking tumor growth and inducing apoptosis [2][4] - In B-Raf-mutant cancers, it has a synergistic effect with VEGFR inhibitors (e.g., sorafenib), overcoming potential resistance to monotherapy Raf inhibitors [5] |
| Molecular Formula |
C24H16F6N6O
|
|---|---|
| Molecular Weight |
518.41
|
| Exact Mass |
518.128
|
| Elemental Analysis |
C, 55.60; H, 3.11; F, 21.99; N, 16.21; O, 3.09
|
| CAS # |
927880-90-8
|
| Related CAS # |
927880-90-8;
|
| PubChem CID |
11656518
|
| Appearance |
White to khaki solid powder
|
| Density |
1.5±0.1 g/cm3
|
| Boiling Point |
667.6±65.0 °C at 760 mmHg
|
| Flash Point |
357.5±34.3 °C
|
| Vapour Pressure |
0.0±2.0 mmHg at 25°C
|
| Index of Refraction |
1.622
|
| LogP |
6.03
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
11
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
37
|
| Complexity |
763
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
FC(C1C=CC(NC2N(C)C3C(=CC(=CC=3)OC3C=C(C4NC(C(F)(F)F)=CN=4)N=CC=3)N=2)=CC=1)(F)F
|
| InChi Key |
YABJJWZLRMPFSI-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C24H16F6N6O/c1-36-19-7-6-15(10-17(19)34-22(36)33-14-4-2-13(3-5-14)23(25,26)27)37-16-8-9-31-18(11-16)21-32-12-20(35-21)24(28,29)30/h2-12H,1H3,(H,32,35)(H,33,34)
|
| Chemical Name |
1-methyl-5-[2-[5-(trifluoromethyl)-1H-imidazol-2-yl]pyridin-4-yl]oxy-N-[4-(trifluoromethyl)phenyl]benzimidazol-2-amine
|
| Synonyms |
RAF 265; RAF265; CHIR-265; CHIR 265; RAF-265; CHIR265
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1 mg/mL (1.93 mM) (saturation unknown) in 10% EtOH + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear EtOH stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 1 mg/mL (1.93 mM) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear EtOH stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: 1 mg/mL (1.93 mM) in 10% EtOH + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 30% PEG400+0.5% Tween80+5% propylene glycol: 30 mg/kg |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9290 mL | 9.6449 mL | 19.2898 mL | |
| 5 mM | 0.3858 mL | 1.9290 mL | 3.8580 mL | |
| 10 mM | 0.1929 mL | 0.9645 mL | 1.9290 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT01352273 | Completed | Drug: MEK162 + RAF265 | Advanced Solid Tumors | Array Biopharma, now a wholly owned subsidiary of Pfizer |
June 2011 | Phase 1 |
| NCT00304525 | Completed | Drug: RAF265 | Metastatic Melanoma | Novartis Pharmaceuticals | April 2006 | Phase 1 Phase 2 |
|
 |