
| Size | Price | Stock | Qty |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| Other Sizes |
Purity: ≥98%
Nitazoxanide (also known as NTZ; NSC 697855) is a synthetic nitrothiazolyl-salicylamide derivative and a broad spectrum antiprotozoal agent with IC50 for canine influenza virus ranges from 0.17 to 0.21 μM. It was approved for treating human protozoan infections. Nitazoxanide reduces parasite growth in cell culture by more than 90% with little evidence of drug-associated cytotoxicity. Nitazoxanide is a new thiazolide antiparasitic agent that shows excellent in vitro activity against a wide variety of protozoa and helminths. Nitazoxanide and its metabolite tizoxanide are more active in vitro than metronidazole against G. intestinalis, E. histolytica and T. vaginalis.
Nitazoxanide is a new nitrothiazole compound with broad-spectrum activity against numerous intestinal protozoa, helminths, and anaerobic bacteria. It is presently approved to treat infections due to G. intestinalis in children and adults and infections due to Cryptosporidium species in children. Approval for use in adults with Cryptosporidium infection and the immunocompromised population is on the horizon. Nitazoxanide is an important new addition to the antiparasitic pharmacopeia. The drug has few side effects and requires a short course of treatment. Nevertheless, a need remains for further studies of its molecular mechanisms of action, bioavailability, and drug interactions to learn whether it can be safely used in a variety of patient groups. Because the majority of parasitic infections occur in the developing world, further data about drug potency and stability are also needed to support its widespread use in this context. Similarly, clinical and pharmacological data on absorption, dosage, and duration of therapy in patients with AIDS and chronic cryptosporidiosis are necessary. In view of its unique mechanism of action, nitazoxanide should be considered for further clinical evaluation in the treatment of parasitic infections (e.g., in combination with paromomycin or azithromycin for treatment of cryptosporidiosis and in combination with albendazole for treatment of intestinal helminth infections) and in reducing the emergence of metronidazole resistance, particularly with Giardia and H. pylori. Additional clinical trial data that would expand our knowledge of nitazoxanide's utility in these contexts is important. With these questions answered, nitazoxanide may represent a significant advance in the treatment of intestinal parasitic infections worldwide.| Targets |
Broad spectrum anthelmintic
|
|---|---|
| ln Vitro |
Globally, the most frequent cause of persistent diarrhea is the flagellated protozoan Giardia lamblia[1].
With an IC50 of 2.4 μM, nitazoxanide has an impact on the growth of G. lamblia trophozoite in axenic culture[1]. NTZ/Nitazoxanide significantly inhibited the replication of JEV in cultured cells in a dose dependent manner with 50% effective concentration value of 0.12 ± 0.04 μg/ml, a non-toxic concentration in cultured cells (50% cytotoxic concentration = 18.59 ± 0.31 μg/ml). The chemotherapeutic index calculated was 154.92. The viral yields of the NTZ-treated cells were significantly reduced at 12, 24, 36 and 48 h post-infection compared with the mock-treated cells. NTZ was found to exert its anti-JEV effect at the early-mid stage of viral infection. [2] NTZ/Nitazoxanide derivatives containing a bromo instead of a nitro group (Table 1) were all inactive against Giardia trophozoites (IC50s of >50 μM), with the exception of RM4820, which showed a moderate inhibitory activity (IC50 of 18.8 μM) that was lower than the ones for RM4802 and RM4805. The efficacy of MTZ was lower than those of NTZ and TIZ but still higher than that of any other drug tested in this study. In order to visualize the morphological alterations of G. lamblia trophozoites induced by Nitazoxanide/NTZ and compare them with those induced by MTZ, confluent axenic trophozoite cultures were treated with 50 μM NTZ or MTZ or with DMSO as a control. Observations with the light microscope showed that after 3 h of treatment with NTZ, approximately 50% of trophozoites were immobile; at 5 h of treatment, over 95% of trophozoites were immobile and formed large multicellular aggregates; and at 24 h of NTZ treatment, no motile trophozoites were found (data not shown). They all exhibited aberrant vacuolar cytoplasmic structures. Inspection by TEM (Fig. 1) largely confirmed these findings and showed that at 1 h of NTZ treatment, a considerable number of trophozoites already exhibited relatively small, aberrant cytoplasmic inclusions within their cytoplasm (Fig. 1A and B). At 3 h of treatment, larger vacuoles containing membranous inclusions or membrane stacks could be observed in a large number of parasites (Fig. 1C), and at 24 h of treatment, these parasites were seriously damaged, as exhibited either by a dissociation of their cytoplasmic organization (Fig. 1D) or, in many instances, by large vacuoles containing membrane residues (Fig. 1E). Besides this, however, the cytoskeletal elements of trophozoites, such as filaments associated with the ventral disk or the flagella and basal bodies, were not notably altered. In control preparations incubated in DMSO alone, no obvious changes in parasite ultrastructure could be detected (Fig. 1F).[1] In a first series of experiments, Caco2 cells were incubated with increasing numbers of trophozoites (103 to 106 parasites per well) in the presence of DMSO as a solvent control or in the presence of 30 μM NTZ/Nitazoxanide. The parasites attaching to Caco2 cells were then quantified by real-time PCR (Fig. 4). In the absence of any drug and at an initial inoculum density of 105 parasites per well, 70 to 90% of the trophozoites remained attached to the Caco2 cells for a period of 24 to 48 h. At an inoculum density 10 times higher (106 parasites/well), this value was decreased to nearly 50%, indicating that binding to the Caco2 cell surface is saturable and dependent on the presence of suitable binding sites and/or host cell surface receptors. In the presence of 30 μM NTZ, with an inoculum density of 105 trophozoites, the number of parasites still attached to Caco2 cells after 24 h decreased to less than 20% of the control value. Based upon these findings, the effects of different thiazolides compared to those of MTZ were investigated. Confluent Caco2 cells were supplemented with fresh Caco2 growth medium. Trophozoites (105) were added to each well, and a number of thiazolides, MTZ, and DMSO were added (Fig. 5). Cells were harvested after 24 h, and the attached trophozoites were quantified by real-time PCR. Only those compounds that had exhibited strong inhibitory effects in axenic cultures (NTZ, TIZ, RM4802, RM4805, and MTZ) interfered with trophozoite attachment in the Caco2 coculture system. In contrast to what was seen with axenic culture, the efficacies of RM4802, RM4805, and MTZ in Caco2 coculture were comparable to those of NTZ and TIZ (Fig. 5). Concentrations lower than 15 μM did not show any significant inhibitory effects for any of the drugs tested.[1] Studies of protozoa and anaerobic bacteria have shown that Nitazoxanide inhibits pyruvate-ferredoxin oxidoreductase (PFOR), an enzyme essential to anaerobic energy metabolism. However, interference with the PFOR enzyme–dependent electron transfer reaction may not be the only pathway by which nitazoxanide exhibits antiprotozoal activity, and the mechanism of nitazoxanide's activity against helminths is unknown. Nitazoxanide has demonstrated in vitro activity against C. parvum and G. intestinalis. It has been shown to inhibit the growth of sporozoites of C. parvum on its own, and has also demonstrated combined in vitro activity with both azithromycin and rifampin, suppressing growth of C. parvum by 83.9% and 79.8%, respectively, compared with 56.1% when used alone. Similarly, in vitro studies of nitazoxanide and its derivative, tizoxanide, have shown greater efficacy than metronidazole against G. intestinalis. Specifically, tizoxanide was demonstrated to be 8 times more active than metronidazole against metronidazole-susceptible isolates of G. intestinalis and twice as active against resistant isolates. Nitazoxanide has also shown broad in vitro activity against numerous other parasitic and microbial pathogens, including E. intestinalis, V. corneae, E. histolytica, T. vaginalis, B. hominis, Echinococcus multilocularis, Echinococcus granulosus,, and F. hepatica. The antimicrobial properties of nitazoxanide and tizoxanide have been tested against 241 anaerobes, the majority of which were inhibited in vitro, with an MIC90 between 0.06 mg/L and 4 mg/L. Nitazoxanide has also shown in vitro and in vivo antimicrobial activity against Clostridium difficile and both metronidazole-susceptible and metronidazole-resistant strains of H. pylori [4]. |
| ln Vivo |
A variety of intestinal parasites, including Giardia lamblia, Entamoeba histolytica, Trichomonas vaginalis, the apicomplexan Cryptosporidium parvum, and enteric bacteria that infect both humans and animals, are resistant to nitazoxanide's broad range of in vivo activity[1].
Mice infected with the Japanese encephalitis virus (JEV) strain have a lower mortality rate when given nitazoxanide (50, 75, or 100 mg/kg/day; intragastric administration for up to 25 days) and are protected against a lethal dose challenge of JEV[2]. NTZ/Nitazoxanide reduces the mortality of mice challenged with a lethal dose of JEV [2] To evaluate the protective effect of NTZ on mice challenged with a lethal dose of JEV, NTZ was administered intragastrically at the indicated doses from 1 day post-infection, daily, for up to 25 days. The mice that were infected with JEV and received a placebo (DMSO) treatment (group JEV + DMSO) started to show the clinical signs of JE including limb paralysis, restriction of movements, piloerection, body stiffening and whole body tremors, from 5 days post-infection, and all mice (10/10 mice) died within 9 days post-infection. In contrast, the mice that were infected with JEV and received NTZ treatment (group JEV + NTZ, 100 mg/kg/day) showed the clinical signs of JE from 11 days post-infection, among these 10 mice, 1 died within 12 days post-infection and 9 survived the experimental period (25 days) (Figure 6A). The NTZ-mediated protection appeared to be dose dependent, as the infected mice receiving 50 mg/kg/day, 75 mg/kg/day and 100 mg/kg/day NTZ led to 30%, 70% and 90% mice survival, respectively (Figure 6A). These data suggested that NTZ treatment reduced the mortality of JEV-infected mice and protected mice from a lethal dose challenge of JEV. The mice that were mock-infected with JEV and received NTZ treatment (group Mock + NTZ) showed no detectable signs of abnormal behavior, similarly to the mice that were mock-infected and received DMSO treatment (group Mock + DMSO) (Figure 6A). Analysis of JEV titers in the brain samples from the experimental mice indicated that NTZ treatment significantly reduced the virus load in the brain from the group JEV + NTZ compared with that from group JEV + DMSO (Figure 6B). The brain samples from the experimental mice were examined for the presence of viral NS3 protein by immunohistochemistry. The viral NS3 protein was stained as brown deposits in the cytoplasm of neuronal cells (Additional file 1, arrowed cells). The brain sections from the JEV + DMSO group of mice showed remarkably higher numbers of NS3-stained positive cells than the sections from the JEV + NTZ group. No NS3-stained positive cells were detected in the sections from the Mock + NTZ or Mock + DMSO groups of mice. Studies with mice. [3] The efficacy of NTZ/Nitazoxanide, either alone or in combination with PRM, was tested in the anti-IFN-γ-conditioned SCID mouse model of acute cryptosporidiosis. While an initial study showed that a partial reduction in oocyst shedding (data not shown) was produced by NTZ, we were unable to replicate this result in several subsequent experiments. The following are the combined results of two independent trials that failed to show efficacy in this model. No difference in the log oocyst shedding between any of the groups treated with NTZ alone and the placebo control group was observed (Fig. 1). In contrast, all mice treated with PRM shed oocysts at significantly lower levels than mice treated with either NTZ alone or the placebo (P < 0.001). Coadministration of NTZ and PRM was no more effective at reducing the level of oocyst shedding than treatment with PRM alone. In general, no significant differences in mean body weight were observed between any of the groups of mice (data not shown). Student-Newman-Keuls analysis of variance revealed that the extent of mucosal infection was significantly greater in mice treated with either NTZ alone or the placebo than in animals treated with PRM (Fig. 2; P < 0.001). Coadministration of NTZ and PRM did not significantly alter the extent of mucosal infection compared to that in mice that received PRM alone. Studies with piglets. [3] The piglet model offers an added advantage in that the piglets develop diarrhea as a consequence of infection. In addition to the piglet euthanized before treatment began, 4 of the 25 animals challenged with C. parvum were euthanized due to poor health associated with diarrhea, including 1 piglet from the placebo group (on day 5 after challenge), 2 from the group receiving 250 mg of Nitazoxanide/NTZ/kg (on days 8 and 9 after challenge), and 1 from the PRM-treated group (on day 5 after challenge). Three additional piglets, 1 placebo-treated, 1 PRM-treated, and 1 uninfected control piglet, were euthanized on day 8 after challenge for a comparative analysis of the extent of mucosal infection. The analysis of both the level of oocyst shedding and extent of mucosal infection revealed that NTZ at 250 mg/kg/day significantly reduced the extent of mucosal infection in these piglets, but it was not as effective as PRM at 500 mg/kg/day (Fig. 3 and 4). With the exception of those in the PRM-treated group, all infected piglets manifested diarrhea of various degrees within 56 h after challenge. Diarrhea persisted until the end of the experiment, 13 days later (11 days after the start of treatment). Table 2 provides a cumulative analysis of the number of days of diarrhea observed for each of the treatment groups. A chi-square analysis of these data revealed very significant differences among the treatment groups (overall Pearson’s chi-square value of 88.096 with 5 df; P < 0.001). Uninfected piglets given Nitazoxanide/NTZ at a dose of 125 mg/kg/day did not have diarrhea (0 of 24 days of observation). In contrast, on 22 of 36 observation days, uninfected piglets given NTZ at a dose of 250 mg/kg/day had significant drug-induced diarrhea (P ≤ 0.001 for both Pearson’s chi-square and Fisher’s exact test [two-tailed]). Among the infected groups, only the PRM-treated group had less-frequent diarrhea than the infected placebo control group. The percentage of days of observation that the piglets in the PRM-treated group showed diarrhea was significantly lower than that in any of the other infected groups (chi-square P < 0.001, and Fisher’s exact two-tailed test, P < 0.001, for comparison with the infected placebo control group, the group receiving NTZ at 125 mg/kg/day, and the group receiving NTZ at 250 mg/kg/day). |
| Cell Assay |
Cell Line: Giardia lamblia trophozoites were cultured in increasing numbers (103–106 parasites per well) in human cancer colon Caco2 cells.
Concentration: 30 μM Incubation Time: 24 hours Result: When Nitazoxanide was not present and the initial inoculum density was 105 parasites per well, 70–90% of the trophozoites stayed adhered to the Caco2 cells for a duration of 24–48 hours.In the presence of 30 μM Nitazoxanide with an inoculum density of 105 trophozoites, the number of parasites still attached to Caco2 cells after 24 hours dropped to less than 20% of the control value. Virus, cells and Nitazoxanide/NTZ administration [2] JEV strain (SH-JEV01) was grown in 3-day-old BALB/c mice and titrated by a plaque assay using BHK-21 cells as described below. BHK-21 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS) at 37°C in an atmosphere containing 5% CO2. NTZ/Nitazoxanide (purity ≥ 98%) was dissolved in culture-grade DMSO at a concentration of 50 μg/μl. NTZ solution was added immediately after a 1 h adsorption period and was kept in the culture medium for the duration of the experiment, unless specified otherwise. Controls received equal amounts of DMSO (final concentration ≤ 0.06%), which did not affect cell viability or virus replication. Cytotoxicity test[2] BHK-21 cells were seeded in a 96-well plate at a density of 5 × 103 cells per well. Following 24 h of incubation, the cells were treated with NTZ at various concentrations ranging from 0.1 to 32 μg/ml at 37°C for 48 h. Cells treated with DMSO alone were used as a control. The cellular toxicity of NTZ was assessed using MTT assay. Cell viability was calculated as a percentage of the total number of viable DMSO-treated control cells. The CC50, which is defined as the concentration that inhibits the proliferation of exponentially growing cells by 50%, was calculated as described. Analysis of the antiviral effect of NTZ/Nitazoxanide in BHK-21 cells [2] BHK-21 cells in six-well plates were infected with JEV at a MOI of 0.001. After a 1 h adsorption period, the cells were treated with NTZ at concentrations ranging from 0.01 to 10 μg/ml and incubated at 37°C for 48 h or the indicated times. The virus yield was determined by a plaque assay and qRT-PCR. The reduction in the virus titer was calculated as follows:% virus titer reduction = [1-(PFUJEV+NTZ/PFUJEV+DMSO)] × 100. The EC50 that is defined as the concentration offering 50% inhibition of viral yield in cells was calculated as described. NTZ/Nitazoxanide activity in cell culture. [3] MDBK cells selectively cloned for susceptibility to C. parvum infection were plated in 96-well microliter plates. To determine the dose response to NTZ, 3.0 × 104 C. parvum oocysts were added with or without drugs to each well 72 h later, when the cells were confluent. Paromomycin sulfate (PRM) was used as a positive control drug. All drug dilutions were made in Dulbecco’s minimum essential medium supplemented with 5% fetal bovine serum, 500 U of penicillin, 500 μg of streptomycin/ml, 1 mM sodium pyruvate, 2 mM l-glutamine, and 0.2% dimethyl sulfoxide (DMSO) (culture medium). Culture medium was added to wells containing MDBK cells infected with C. parvum oocysts as a negative control. All drug concentrations and controls were tested in quadruplicate. Following an incubation for 48 h (37°C, 8% CO2) the monolayers were methanol fixed and reacted in an indirect immunofluorescence assay to determine the intensity of infection. Fixed wells were rehydrated for 15 min with phosphate-buffered saline (PBS) containing 1% normal goat serum (NGS. Following rehydration, the parasite-reactive rabbit antiserum was diluted 1:1,000 in PBS containing 1% NGS, added to the wells, and incubated for 1 h at room temperature. All wells were washed three times with PBS, and bound antibody was detected with a fluorescein isothiocyanate-conjugated goat anti-rabbit immunoglobulin G antibody diluted 1:100 in PBS containing 1% NGS. Following a 1-h incubation at room temperature, the wells were washed three times with PBS and dried. The extent of C. parvum infection was quantitated, under UV light microscopy, with a microcomputer video imaging device specifically designed for this purpose. The percent inhibition of infection was calculated as follows: 1 − (mean number of parasites in wells with drug/mean number of parasites in control wells) × 100. All results were assigned inhibition scores of 0, 1, 2, 3, and 4 for inhibition of 0 to 30%, 31 to 55%, 56 to 70%, 71 to 90%, and 91 to 100%, respectively. Cytotoxicity assay. [3] The cytotoxicities of NTZ/Nitazoxanide and PRM to MDBK cells were determined by the CellTiter 96 AQueous non-radioactive cell proliferation assay. Controls for each cytotoxicity assay included (i) uninfected cells incubated in culture medium, (ii) infected cells incubated in culture medium, and (iii) cells exposed to a freeze-thaw lysate containing 3.0 × 104 oocyst equivalents in culture medium. The percent cytotoxicity was calculated from the optical density (OD) as follows: [(mean OD of uninfected cells − mean OD of infected cells)/mean OD of uninfected cells] × 100. All results were assigned cytotoxicity scores of 0, 1, 2, 3, and 4 for percent cytotoxicity of 0 to 5%, 6 to 25%, 26 to 50%, 51 to 75%, and 76 to 100%, respectively. Cytotoxicity scores of 0, 1, and 2 are considered to indicate nontoxicity, mild toxicity, and moderate toxicity, respectively, and scores of 3 and 4 are regarded as indicating severe toxicity for MDBK cells. A negative percent toxicity results when the OD of the infected cells is greater than the OD of the uninfected cells. |
| Animal Protocol |
Animal Model: JEV was injected intraperitoneally into female Chinese Kunming mice that were three weeks old and weighed between 12 and 14 grams.[2]
\nDosage: 50, 75 or 100 mg/kg/day \nAdministration: Administered intragastrically by gavage \nResult: 50 mg/kg/day, 75 mg/kg/day and 100 mg/kg/day led to 30%, 70% and 90% mice survival, respectively.\n\n \nNTZ/Nitazoxanide activity in SCID mice. [3] \nThe anti-gamma-interferon (IFN-γ)-conditioned SCID mouse model has been described previously. Briefly, newly weaned (3- to 4-week-old) male inbred C.B-17 SCID mice were housed in microisolator cages in IACUC-approved facilities. Prior to the initiation of a drug trial, the animals were randomized into seven groups of seven mice each. Each mouse was primed with an intraperitoneal injection of 1 mg of XMG1.2, an IFN-γ-neutralizing monoclonal antibody. Two hours later, each mouse in six of the seven groups received an oral inoculation of 107 oocysts. Drug treatment was initiated on day 6 of infection, coinciding with the onset of oocyst excretion in the feces. Treatment schedules were as follows: group 1, 200 mg of NTZ/kg of body weight/day; group 2, 100 mg of NTZ/kg/day; group 3, 200 mg of NTZ/kg/day combined with 2,500 mg of PRM/kg/day; group 4, 100 mg of NTZ/kg/day combined with 2,500 mg of PRM/kg/day, and group 5, 2,500 mg of PRM/kg/day. NTZ was dissolved in 100% DMSO and administered orally in two divided doses of 30 μl each per day. PRM was dissolved in the drinking water to a concentration of 10 mg/ml (16.2 mM), resulting in a dose of 2,500 mg/kg/day based on the daily water consumption. Group 6 consisted of uninfected mice treated with 200 mg of NTZ/kg/day combined with 2,500 mg of PRM/kg/day (drug toxicity control group). Group 7 consisted of seven mice treated orally with 30 μl of DMSO twice per day (the placebo control group). All mice were treated for 10 days and maintained for an additional 5 days after the end of treatment. The level of oocyst shedding was determined three times per week throughout the study by microscopic observation of 30 high-power fields of a modified acid-fast stained fecal smear from each infected animal. Results are presented as the mean log oocysts shed per group ± the 95% confidence intervals. Body weights were determined one time per week throughout the study. Results are presented as the mean body weight per group ± the 95% confidence intervals. At necropsy, sections were taken from the pyloric region of the stomach, mid-small intestine, ileum, cecum, and proximal colon for histologic analysis to determine the extent of mucosal infection. Each site was assigned a score depending on the extent of infection, as follows: 0, no infection; 1, very difficult-to-find parasite forms; 2, sparse but easily found parasite forms; 3, abundant parasite forms but focally distributed; 4, extensive presence of parasite forms covering most mucosal surfaces; and 5, extensive presence of parasite forms covering the entire mucosal surface. The data are presented as the mean total score of the five sites ± 95% confidence intervals.\n \nNTZ/Nitazoxanide activity in the piglet diarrhea model. [3] \nThirty-one gnotobiotic piglets derived by cesarean section from four litters were maintained inside sterile isolators for the duration of the experiment as described previously. Twenty-six of the 31 piglets were challenged with oocysts 24 h after derivation. Because these experiments were not performed simultaneously, the infecting dose of 5 × 106 excysting oocysts was calculated based on the percent oocyst excystation in vitro (rate of excystation). The rate of in vitro excystation was determined following incubation of the oocysts in 0.75% taurocholic acid for 45 min at 37°C. Once the in vitro excystation rate was determined, the inocula were adjusted accordingly so that 13 piglets received 2 × 107 oocysts and 13 piglets received 7 × 106 oocysts.\n\nPiglets were observed two or three times daily for signs of diarrhea, depression, and anorexia and for overall appearance. Diarrhea was defined as a twofold increase in the frequency, volume, and water content of the fecal discharge of a piglet compared to those in uninfected control piglets. Body weights and fecal samples were obtained daily. Within 3 days after challenge, piglets were assigned to groups based on a combination of body weight, onset of oocyst shedding, and diarrhea status. The piglets were then started on a daily treatment schedule of either 250 mg of Nitazoxanide/NTZ/kg (five piglets), 125 mg of NTZ/kg (six piglets), 500 mg of PRM/kg (five piglets), or a placebo (milk; nine piglets). Five uninfected control animals served as drug toxicity controls; three piglets received NTZ at 250 mg/kg/day, and two piglets received 125 mg/kg/day. One of the 26 infected piglets was euthanized because of severe illness and was excluded from the study. All drugs were administered via the milk diet in two divided doses daily for 11 days. The number of oocysts present in an entire modified-acid-fast-stained fecal smear was determined daily for each piglet. Since piglets develop diarrhea as a consequence of C. parvum infection, determination of the number of oocysts shed must account for any variability in fecal consistency that we observe. In particular, the presence of watery diarrhea will influence the number of oocysts detected in a fecal smear due to the effective dilution of the fecal material by the increased fluid content. Because of this, we have devised a scoring system that accounts for both the qualitative nature of the fecal material and the number of oocysts detected. Scores are assigned in this system as follows: 0, no oocysts detected; 1, ≤10 oocysts; 2, ≤25 oocysts; 3, ≤50 oocysts; 4, ≤100 oocysts; and 5, >100 oocysts. Results are presented as the mean oocyst shedding score for each treatment group ± standard error of the mean. Surviving piglets were euthanized 11 days after the onset of treatment, and six gut sections (the pyloric region of the stomach, three equally spaced small intestinal sites, the cecum, and the colon) were removed for histologic analysis of the extent of mucosal infection. Each site was assigned a score depending on the extent of infection by the system described above for the anti-IFN-γ-conditioned SCID mouse. Results are presented as the total score of the six sites for individual piglets.\n \n\nAnalysis of the antiviral effect of NTZ/Nitazoxanide in a mouse model[2] \nThree-week old female Chinese Kunming mice (12–14 g body weight) were randomly divided into six groups (10 mice/group). Group JEV + NTZ was infected with JEV and received NTZ treatment (50, 75 or 100 mg/kg/day). Group JEV + DMSO was infected with JEV and received a placebo (DMSO) treatment. Group Mock + NTZ was mock-infected with JEV and received NTZ treatment. Group Mock + DMSO was mock-infected with JEV and received a placebo (DMSO) treatment. For infection, mice were infected intraperitoneally with 6×104 PFU of JEV (containing 50 × LD50 of JEV). For NTZ treatment, NTZ was dissolved in DMSO and administered intragastrically by gavage, in which a feeding needle was introduced into the esophagus and NTZ was delivered directly into the stomach. NTZ was tested at a total dose of 50, 75 or 100 mg/kg/day, and was consecutively administered from 1 day post-infection, daily, for up to 25 days. The mice were monitored daily for morbidity and mortality. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
The relative bioavailability of the suspension is 70% compared to tablets. When taken with food, the AUC and Cmax of tablets increase by 2-fold and 50%, respectively, while the AUC and Cmax of the oral suspension increase by 45% to 50% and ≤10%, respectively. Tinazonide is excreted in urine, bile, and feces. Tinazonide glucuronide is excreted in urine and bile. Approximately two-thirds of the oral dose of tinazonide is excreted in feces, and one-third in urine. Tinazonide is cleared in urine and feces. Its metabolite, tinazonide, is also present in urine, plasma, and breast milk. The unchanged form of this drug was not detected in urine. Metabolism/Metabolites The active metabolite of this drug is tinazonide (desacetylnidazonide). The initial reaction in the nizonilide metabolic pathway is hydrolysis to tinzonilide, followed by a conjugation reaction, primarily glucuronidation, to tinzonilide glucuronide. The bioavailability of the oral suspension differs from that of the oral tablets. Compared to tablets, the bioavailability of the suspension is 70%. Compared to administration on an empty stomach, co-administration with food nearly doubles the AUCt of tinzonilide and tinzonilide glucuronide in plasma, increasing the maximum concentration by nearly 50%. When the oral suspension is taken with food, the AUC of tinzonilide and its glucuronide increases by approximately 50%, while the Cmax increases by less than 10%. Biological half-life 7.3 hours |
| Toxicity/Toxicokinetics |
Hepatotoxicity
Nizorazoxane treatment is not associated with elevated serum transaminase levels or clinically significant acute liver injury. However, studies on long-term nizorazoxane treatment are limited, and most controlled trials of this drug involved short-term treatment without serum transaminase monitoring. Nizorazoxane has been used as adjunctive therapy for chronic hepatitis C, usually in combination with pegylated interferon, with or without ribavirin; in these studies, most patients experienced improvement in serum transaminase levels, and no cases of acute hepatitis exacerbation or jaundice were reported. Probability Score: E (Unlikely a cause of clinically significant liver injury). Pregnancy and Lactation Effects ◉ Overview of Use During Lactation Limited information suggests that after a mother takes 500 mg of nizorazoxane, the concentration of the active metabolite tinizonide in breast milk is low and is not expected to have any adverse effects on breastfed infants, especially those older than 2 months. However, until more data is available, especially during breastfeeding newborns or preterm infants, other medications may be preferred. ◉ Effects on breastfed infants No relevant published information was found as of the revision date. ◉ Effects on lactation and breast milk No relevant published information was found as of the revision date. Protein binding Extremely high protein binding rate (greater than 99%), binding to plasma proteins. Drug interactions[4] Currently, no in vivo drug interaction studies of nitazoxabine have been conducted. Because nitazoxabine binds to plasma proteins at a rate greater than 99%, caution should be exercised when taking nitazoxabine concurrently with other medications that have high plasma protein binding rates and narrow therapeutic indices. Patients taking warfarin and nitazoxabine concurrently are advised to monitor prothrombin time. Side effects[4] Nitazoxabine is generally well tolerated, and no significant adverse events have been observed in human trials. Adverse events are usually mild and transient, primarily gastrointestinal, such as abdominal pain, diarrhea, and nausea. In over 2,000 HIV-free patients participating in clinical trials, adverse events occurring in less than 1% of cases included anorexia, flatulence, increased appetite, salivary gland enlargement, fever, infection, malaise, elevated creatinine levels, elevated serum alanine aminotransferase levels, pruritus, sweating, scleral pallor/jaundice, rhinitis, dizziness, and abnormal urine color. Furthermore, no significant changes were observed in electrocardiogram, vital signs, hematological, clinical chemistry, or urinalysis parameters in patients receiving nitazoxanib. Nitazoxanib is well tolerated at a maximum dose of 4 grams, whether taken on an empty stomach or with food; however, the frequency of gastrointestinal side effects increases significantly with increasing dose levels. |
| References | |
| Additional Infomation |
Pharmacodynamics
The overall action of this drug is to inhibit the activity of microorganisms by interfering with important energy pathways required for their survival and proliferation. Nizoralite exerts its antiprotozoal activity by interfering with the pyruvate feroxin/flavoprotein oxidoreductase-dependent electron transport reaction, which is essential for the anaerobic energy metabolism of many microorganisms. Therefore, the sporozoites of Cryptosporidium microsporum and the trophozoites of Giardia lamblia are inhibited, thereby alleviating diarrhea symptoms. Interference with the PFOR enzyme-dependent electron transport reaction may only be one of many pathways by which nizoralite exerts its antiprotozoal activity. [2-[[(5-nitro-2-thiazolyl)amino]-oxymethyl]phenyl]acetic acid ester is a carboxylic acid ester belonging to the benzamide class of compounds. It is functionally related to salicylamide drugs. Nizoralite belongs to the thiazole class of drugs. Nizoralite (NTZ) is a broad-spectrum anti-infective drug that significantly modulates the survival, growth, and proliferation of various extracellular and intracellular protozoa, worms, anaerobic bacteria, microaerophiles, and viruses. This drug is effective in treating gastrointestinal infections in healthy individuals, including those caused by Cryptosporidium or Giardia lamblia. It is generally well-tolerated. Nizorazoxanil is a first-line standard of care for healthy (non-immunosuppressed) adults and children with diseases caused by Cryptosporidium or Giardia lamblia, and can also be used to treat diseases caused by other protozoa or worms. In recent years, it has been investigated as a broad-spectrum antiviral drug due to its ability to inhibit the replication of various RNA and DNA viruses. Nizorazoxanil is an antiprotozoan drug. Nizorazoxanil is an antibacterial drug effective against a variety of parasites and protozoa, primarily used in the United States for the treatment of Giardiasis and Cryptosporidium infections. It has been reported that nizorazoxanil treatment does not cause elevated serum transaminases or clinically significant liver damage. Nizorazoxanil is a synthetic benzamide drug with antiprotozoan activity. Nizorazoxane exerts its antiprotozoal activity by interfering with the pyruvate ferroredoxin/flavoprotein oxidoreductase-dependent electron transfer reaction, which is crucial for anaerobic energy metabolism. PFOR enzymes reduce nizorazoxane, thereby impairing energy metabolism. However, interfering with PFOR enzyme-dependent electron transfer may not be the only way nizorazoxane exerts its antiprotozoal activity. Nizorazoxane is effective against both Giardia lamblia and Cryptosporidium microsporidium. Nizorazoxane is a small molecule drug, with clinical trials up to Phase IV (covering all indications). It was first approved in 2002 for the treatment of amebiasis and diarrhea, and has 26 investigational indications. Thiazole drugs are a new class of anti-infectives, with the parent compound being the nitrothiazole drug nizorazoxane [2-acetoxy-N-(5-nitro-2-thiazolyl)benzamide] (NTZ). NTZ has broad-spectrum activity against a variety of worms, protozoa, and intestinal bacteria that infect animals and humans. In vivo, nitrozonide (NTZ) is rapidly deacetylated to tezolinide (TIZ), which has similar activity. We compared the effects of NTZ, TIZ, several other modified thiazole compounds, and metronidazole (MTZ) on the in vitro activity of Giardia lamblia trophozoites grown under sterile conditions and in co-culture with the human colon cancer cell line Caco2. Modifications of thiazole compounds included replacing the nitro group on the thiazole ring with bromine and altering the position of the methyl group on the benzene ring. Of the seven compounds with bromine-substituted nitro groups, only RM4820 showed moderate inhibition of Giardia lamblia proliferation under sterile conditions but no inhibition under Caco2 cell co-culture conditions, with an IC50 of 18.8 μM; in contrast, the IC50 values for nitrothiazole (NTZ) and tezolinide were 2.4 μM, and for metronidazole (MTZ) was 7.8 μM. In addition, methylation or carboxylation at the 3-position of the benzene ring significantly reduced the activity, while methylation at the 5-position completely eliminated the antiparasitic activity of nitrothiazole compounds. Trophozoites treated with nitrothiazole showed obvious lesions on the peripelvis within 2 to 3 hours after treatment, while treatment with metronidazole resulted in severe damage to the dorsal membrane at a later time. [1] Background: Japanese encephalitis virus (JEV) has a significant impact on public health. It is estimated that 3 billion people in high-risk areas are still unvaccinated, and the number of unvaccinated people in some Asian countries is increasing. Therefore, there is an urgent need to develop new therapeutic drugs for Japanese encephalitis. Nitrozonide (NTZ) is a thiazole anti-infective drug approved for the treatment of parasitic gastroenteritis. Recent studies have shown that NTZ has antiviral properties. This study evaluated the antiviral activity of NTZ against Japanese encephalitis virus (JEV) in cultured cells and mouse models. Methods: JEV-infected cells were treated with different concentrations of NTZ. The replication of Japanese encephalitis virus (JEV) in control and NTZ-treated cells was detected by viral titer assay. NTZ was administered at different time points after JEV infection to determine the stage at which NTZ affects JEV replication. Mice were infected with a lethal dose of JEV and administered NTZ by gavage starting on day 1 post-infection. This study evaluated the protective effect of NTZ against JEV infection in mice. Results showed that NTZ significantly inhibited JEV replication in cultured cells in a dose-dependent manner, with a 50% effective concentration (EC50) of 0.12 ± 0.04 μg/ml, which was non-toxic to cultured cells (50% cytotoxic concentration was 18.59 ± 0.31 μg/ml). The calculated chemotherapeutic index was 154.92. Compared with the control group, the viral yield in the NTZ-treated group was significantly reduced at 12, 24, 36, and 48 hours post-infection. NTZ exerts its anti-JEV effect in the early to mid-stages of viral infection. The anti-Japanese encephalitis virus (JEV) activity of NTZ was also confirmed in vivo. In mice that were given 100 mg/kg/day of NTZ by gavage, 90% of the mice were protected from lethal doses of JEV. Conclusion: Both in vitro and in vivo data showed that NTZ has anti-JEV activity, suggesting that NTZ may be used to treat Japanese encephalitis. [2] Nitrozonide (NTZ) is a drug currently undergoing human clinical trials to evaluate its efficacy in treating chronic cryptosporidiosis. This study evaluated the activity of NTZ in cell culture and two animal models. The inhibitory activity of NTZ was compared with that of paromomycin (PRM), a drug that is partially effective against Cryptosporidium microsporidium. NTZ at a concentration of 10 μg/ml (32 μM) reduced the growth of the parasite in cell culture by more than 90% with almost no drug-related cytotoxicity, while PRM at a concentration of 2000 μg/ml (3.2 mM) reduced it by only 80%. In contrast to in vitro efficacy, in SCID mice infected with Cryptosporidium and treated with anti-gamma interferon, neither NTZ at doses of 100 or 200 mg/kg body weight/day for 10 consecutive days effectively reduced the parasite load. The combination of NTZ and PRM was not superior to PRM alone. Finally, in a germ-free piglet diarrhea model, oral administration of NTZ at 250 mg/kg/day for 11 consecutive days partially reduced the parasite load, but a dose of 125 mg/kg/day was ineffective. However, higher doses of nitrozonide (NTZ) induced drug-related diarrhea in piglets, which may have affected its therapeutic efficacy. As we previously reported, PRM at a dose of 500 mg/kg/day significantly reduced the parasite load in piglets. Our results indicate that, among all tested models, the piglet diarrhea model best mimics the partial response of patients with chronic cryptosporidiosis to nitrozonide treatment. [3] Nizorazoxanil is a novel thiazole antiparasitic drug that exhibits excellent in vitro activity against a variety of protozoa and worms. It is administered orally, has good bioavailability and is well-tolerated, with the main side effect being mild gastrointestinal side effects. No drug interactions have been found. Nizorazoxanil is approved for the treatment of Giardia lamblia diarrhea in patients aged 1 year and older and Cryptosporidium diarrhea in children aged 1–11 years. Currently, it is seeking approval for the treatment of Cryptosporidium infection in adults and for the treatment of immunocompromised patients. It is an important addition to the antiparasitic drug library. [4] |
| Molecular Formula |
C12H9N3O5S
|
|---|---|
| Molecular Weight |
307.28
|
| Exact Mass |
307.026
|
| Elemental Analysis |
C, 46.90; H, 2.95; N, 13.67; O, 26.03; S, 10.44
|
| CAS # |
55981-09-4
|
| Related CAS # |
Nitazoxanide-d4;1246819-17-9
|
| PubChem CID |
41684
|
| Appearance |
Light yellow to yellow solid powder
|
| Density |
1.5±0.1 g/cm3
|
| Melting Point |
202ºC
|
| Index of Refraction |
1.673
|
| LogP |
1.79
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
7
|
| Rotatable Bond Count |
4
|
| Heavy Atom Count |
21
|
| Complexity |
428
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
S1C(=C([H])N=C1N([H])C(C1=C([H])C([H])=C([H])C([H])=C1OC(C([H])([H])[H])=O)=O)[N+](=O)[O-]
|
| InChi Key |
YQNQNVDNTFHQSW-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C12H9N3O5S/c1-7(16)20-9-5-3-2-4-8(9)11(17)14-12-13-6-10(21-12)15(18)19/h2-6H,1H3,(H,13,14,17)
|
| Chemical Name |
[2-[(5-nitro-1,3-thiazol-2-yl)carbamoyl]phenyl] acetate
|
| Synonyms |
NSC-697855; NTZ; NSC 697855;NSC697855; NITAZOXANIDE; 55981-09-4; Alinia; Nitazoxanida; 2-((5-nitrothiazol-2-yl)carbamoyl)phenyl acetate; Nitazoxamide; Nitazoxanidum; Daxon; Alinia, Colufase, Daxon, Nitazoxamide
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO : 61~100 mg/mL ( 198.51~325.44 mM )
|
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3.25 mg/mL (10.58 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 32.5 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 10% DMSO+40% PEG300+5% Tween-80+45% Saline: ≥ 3.25 mg/mL (10.58 mM) (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2544 mL | 16.2718 mL | 32.5436 mL | |
| 5 mM | 0.6509 mL | 3.2544 mL | 6.5087 mL | |
| 10 mM | 0.3254 mL | 1.6272 mL | 3.2544 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
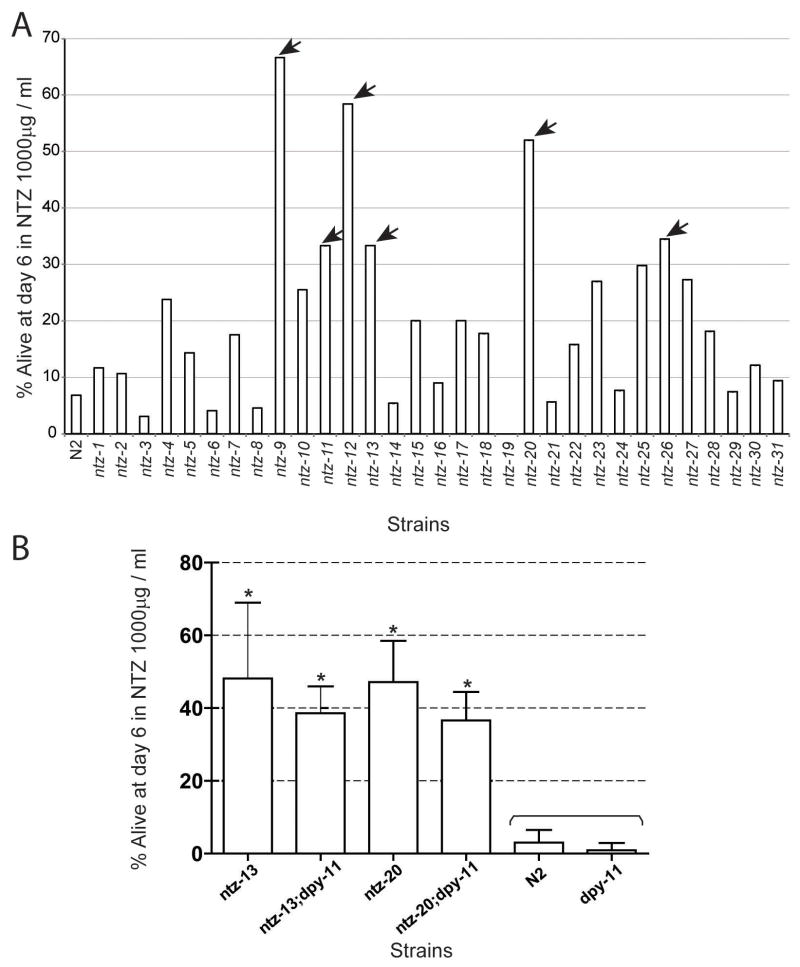 Forward genetic screen for identification of NTZ resistant worm mutants.Mol Biochem Parasitol.2014 Jan;193(1):1-8. |
|---|
Dose response curves ofC. elegansmutants resistant to other classes of drugs to NTZ in a six day lethality assay.Mol Biochem Parasitol.2014 Jan;193(1):1-8. |
Combination of NTZ with albendazole (ALB) and pyrantel (PYR).Mol Biochem Parasitol.2014 Jan;193(1):1-8. |
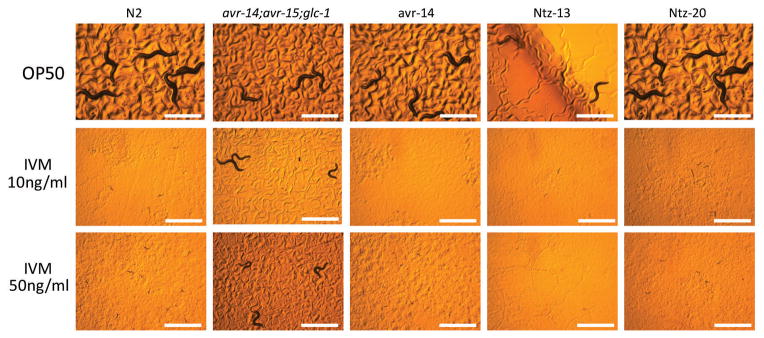 Ivermectin (IVM) susceptibility of the two NTZ resistant mutants identified in forward genetic screens at 44–45 hours.Mol Biochem Parasitol.2014 Jan;193(1):1-8. |
|---|
Effect of NTZ onC. elegansN2 wild-type nematodes.Mol Biochem Parasitol.2014 Jan;193(1):1-8. |