
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
MI-2 (MI2, Menin-MLL Inhibitor) is a novel, potent and selective inhibitor of menin-MLL interaction with antineoplastic activity. It inhibits menin-MLL interaction with an IC50 of 446 nM. MI-2 acts by binding to the wild-type menin but doesn't bind M278K and Y323K menin mutants. In HEK293 cells transfected with Flag-MLL-AF9, MI-2 effectively inhibited the menin-MLL-AF9 interaction without affecting the expression level of menin and MLL-AF9.
| Targets |
Interaction between Menin (encoded by MEN1 gene) and Mixed Lineage Leukemia (MLL, encoded by KMT2A gene) protein (Ki: ~64 nM, determined via Surface Plasmon Resonance (SPR) binding assay; IC50: ~125 nM for inhibition of Menin-MLL complex formation, measured via Homogeneous Time-Resolved Fluorescence (HTRF) assay). No significant binding to other related proteins (e.g., Menin-MLL5, Menin-JUND) at concentrations up to 10 μM, confirming selective targeting of Menin-MLL interaction [1]
|
|---|---|
| ln Vitro |
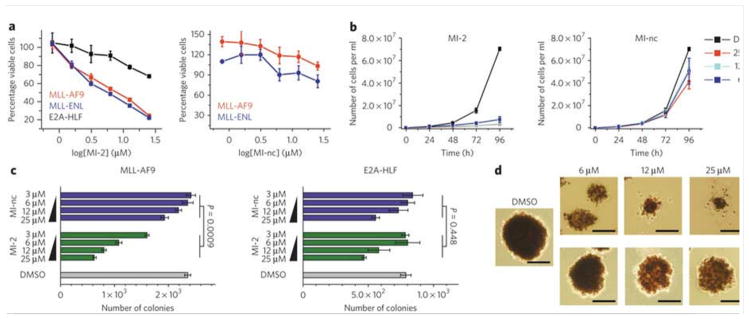
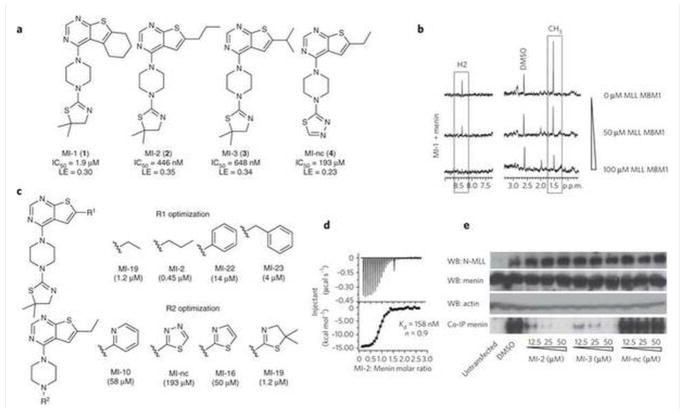
The menin-MLL inhibitor MI-2, with GI50 values of approximately 5 μM, very efficiently inhibits the proliferation of MLL-AF9 and MLL-ENL transduced BMC. Several compounds with IC50 values in the nanomolar range were developed as a result of the assessment of various hydrophobic groups at R1. These compounds include MI-2 (IC50= 446±28 nM) and MI-3 (IC50= 648±25 nM). The dissociation constants (Kd=158 nM for MI-2) for the menin-MLL inhibitors have been measured at the nanomolar range. Inhibiting the menin-MLL-AF9 connection in human cells is a highly effective way that MI-2 can access the protein target. Moreover, MI-2 exhibits minimal impact on the proliferation of BMC transduced with E2A-HLF (GI50>50 μM). This could potentially be attributed to the inhibition of menin interaction with wild-type MLL. After MI-2 treatment, GI50 values for MV4;11 (harboring MLL-AF4; GI50=9.5 μM), KOPN-8 (MLL-ENL; GI50=7.2 μM), ML-2 (MLL-AF6; GI50=8.7 μM), and MonoMac6 (MLL-AF9; GI50=18 μM) are less than 10 μM[1].
1. Antiproliferative activity in MLL-rearranged leukemia cells: MI-2 (Menin-MLL Inhibitor) inhibited proliferation of MLL-rearranged leukemia cell lines (MV4;11, MOLM-13, THP-1, RS4;11) in a dose-dependent manner. After 72-hour treatment, IC50 values were ~1.2 μM (MV4;11), ~1.8 μM (MOLM-13), ~2.5 μM (THP-1), and ~3.1 μM (RS4;11) (CellTiter-Glo assay). In contrast, non-MLL-rearranged leukemia cells (K562, HL-60) showed significantly higher IC50 (>20 μM), indicating selectivity for MLL-rearranged cells [1] 2. Inhibition of Menin-MLL downstream signaling: MV4;11 cells treated with MI-2 (Menin-MLL Inhibitor) (0.5-5 μM) for 24 hours showed dose-dependent downregulation of MLL target genes. qPCR revealed ~5-fold reduction in HOXA9 mRNA and ~4-fold reduction in MEIS1 mRNA at 2 μM. Western blot confirmed decreased HOXA9 and MEIS1 protein levels, with no changes in Menin or MLL protein expression [1] 3. Induction of apoptosis: MV4;11 cells treated with 2 μM MI-2 (Menin-MLL Inhibitor) for 48 hours showed a 4.2-fold increase in apoptotic rate (from 4.8% to 20.2%) via Annexin V-FITC/PI flow cytometry. Western blot detected upregulation of cleaved caspase-3 and cleaved PARP, and downregulation of anti-apoptotic protein BCL-2 [1] 4. Suppression of colony formation: MOLM-13 cells seeded in methylcellulose medium with MI-2 (Menin-MLL Inhibitor) (0.5-2 μM) showed dose-dependent reduction in colony number. At 2 μM, colony formation was inhibited by ~78% compared to the vehicle control, consistent with reduced leukemic cell self-renewal [1] 5. Minimal effect on normal hematopoietic cells: Human CD34+ normal hematopoietic progenitor cells treated with 5 μM MI-2 (Menin-MLL Inhibitor) for 72 hours showed no significant reduction in viability (>85% vs. control) or colony-forming capacity (>80% vs. control), indicating low toxicity to normal hematopoiesis [1] |
| ln Vivo |
MLL-AF9 transformed BMC that remained viable after 7 days of treatment with MI-2 and MI-3 showed substantial changes in morphology, indicative of monocytic differentiation, as evidenced by increased cell size, lower nuclear to cytoplasmic ratio and highly vacuolated cytoplasm. Consistent with the change in cell morphology, the expression of CD11b was substantially increased on MLL-AF9 transformed BMC after 7 days of treatment with MI-2 and MI-3.
1. MV4;11 xenograft model (NOD/SCID mice, 6-8 weeks old, female): Mice were subcutaneously injected with 2×10^6 MV4;11 cells. When tumors reached ~100 mm³, mice were randomized into two groups (n=6/group): (1) Vehicle: 10% DMSO + 90% corn oil (oral gavage, twice daily); (2) MI-2 (Menin-MLL Inhibitor): 50 mg/kg (oral gavage, twice daily). Treatment lasted 21 days. - Tumor growth inhibition: The average tumor volume in the treatment group was ~280 mm³ at day 21, compared to ~1200 mm³ in the vehicle group (tumor growth inhibition, TGI: ~77%). Tumor weight was reduced by ~75% (0.32 g vs. 1.28 g in vehicle) [1] - Mechanism validation: qPCR of tumor tissues showed ~4.5-fold downregulation of HOXA9 and ~3.8-fold downregulation of MEIS1 mRNA in the treatment group. Western blot confirmed reduced HOXA9 protein levels [1] - Animal health: No significant body weight loss (<5%) was observed in the treatment group. Serum levels of ALT, AST, BUN, and creatinine were within normal ranges, indicating no obvious肝肾 (liver/kidney) toxicity [1] |
| Enzyme Assay |
1. HTRF assay for Menin-MLL complex inhibition:
- Reaction system: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 0.01% Tween-20, 20 nM recombinant Menin protein, 10 nM His-tagged MLL peptide (residues 371-404, containing Menin-binding domain), 5 nM Eu-labeled anti-His antibody, 20 nM XL665-conjugated anti-Menin antibody, and serial concentrations of MI-2 (Menin-MLL Inhibitor) (0.01-10 μM). - Incubation and detection: The mixture was incubated at room temperature for 1 hour. Time-resolved fluorescence was measured (excitation: 337 nm, emission: 620 nm and 665 nm). The inhibition rate was calculated from the 665 nm/620 nm signal ratio, and IC50 was determined via four-parameter logistic fitting [1] 2. SPR binding assay for Ki determination: - Sensor chip preparation: Recombinant Menin protein was covalently immobilized on a CM5 sensor chip via amine coupling. - Binding analysis: Serial concentrations of MI-2 (Menin-MLL Inhibitor) (0.1-10 μM) were pre-incubated with 50 nM MLL peptide for 30 minutes, then injected over the Menin-immobilized chip at 30 μL/min. Binding responses were recorded, and Ki was calculated using a competitive binding model (BIAevaluation software) [1] 3. Selectivity assay: The HTRF assay format was used to test MI-2 (Menin-MLL Inhibitor) (0.01-10 μM) against Menin-MLL5 and Menin-JUND interactions. No significant inhibition (<10%) was observed at 10 μM, confirming selective targeting of Menin-MLL [1] |
| Cell Assay |
1. Cell proliferation (CellTiter-Glo) assay:
- Cell seeding: MLL-rearranged (MV4;11, MOLM-13) and non-MLL-rearranged (K562, HL-60) leukemia cells were seeded in 96-well plates at 5×10^3 cells/well (RPMI-1640 medium + 10% FBS + 1% penicillin-streptomycin). - Drug treatment: After 24-hour adherence (for adherent cells) or direct treatment (for suspension cells), MI-2 (Menin-MLL Inhibitor) was added at concentrations of 0.1, 0.5, 1, 5, 10, 20 μM (triplicate wells per concentration). Plates were incubated at 37°C (5% CO2) for 72 hours. - Viability detection: 100 μL CellTiter-Glo reagent was added to each well, and luminescence was measured after 10 minutes. IC50 values were calculated using GraphPad Prism [1] 2. qPCR for MLL target genes: - RNA extraction: MV4;11 cells treated with MI-2 (Menin-MLL Inhibitor) (0.5-5 μM) for 24 hours were lysed, and total RNA was isolated using an RNA extraction kit. cDNA was synthesized via reverse transcription. - Real-time PCR: qPCR was performed with SYBR Green master mix and gene-specific primers (HOXA9, MEIS1, GAPDH as internal control). Relative gene expression was calculated using the 2^(-ΔΔCt) method [1] 3. Western blot analysis: - Protein extraction: Treated MV4;11 cells were lysed in RIPA buffer (with protease/phosphatase inhibitors). Protein concentration was determined via BCA assay. - Electrophoresis and detection: 30 μg of protein was separated by 10% SDS-PAGE, transferred to PVDF membranes, and blocked with 5% non-fat milk (TBST) for 1 hour. Membranes were incubated with primary antibodies (anti-HOXA9, anti-MEIS1, anti-cleaved caspase-3, anti-BCL-2, anti-Menin, anti-MLL, anti-β-actin) overnight at 4°C, followed by HRP-conjugated secondary antibodies for 1 hour at room temperature. Bands were visualized via ECL reagent [1] 4. Colony formation assay: - MOLM-13 cells (1×10^3 cells/mL) were mixed with methylcellulose medium containing MI-2 (Menin-MLL Inhibitor) (0.5-2 μM) and plated in 6-well plates. Plates were incubated at 37°C (5% CO2) for 14 days. Colonies (>50 cells) were counted manually, and colony formation efficiency was calculated as (number of colonies in treatment group/number in control group) × 100% [1] |
| Animal Protocol |
NA NA
1. Xenograft model establishment: - Mice preparation: Female NOD/SCID mice (6-8 weeks old) were acclimated to the laboratory environment for 1 week. MV4;11 cells were harvested in the logarithmic growth phase, washed with PBS, and resuspended in PBS + 50% Matrigel at a concentration of 1×10^7 cells/mL. - Cell injection: Each mouse was subcutaneously injected with 0.2 mL of the cell suspension (2×10^6 cells) into the right flank [1] 2. Treatment protocol: - Randomization: When tumors reached ~100 mm³ (7-10 days post-injection), mice were randomized into two groups (n=6/group) to ensure equal average tumor volume. - Drug preparation: MI-2 (Menin-MLL Inhibitor) was dissolved in 10% DMSO + 90% corn oil to a concentration of 10 mg/mL (for 50 mg/kg dose, based on average mouse weight of 20 g). - Administration: The treatment group received oral gavage of MI-2 (Menin-MLL Inhibitor) (50 mg/kg) twice daily; the vehicle group received the same volume of solvent. Treatment lasted 21 days [1] 3. Monitoring and sample collection: - Tumor volume: Tumor length (L) and width (W) were measured with calipers every 2 days, and volume was calculated as V = L×W²/2. - Body weight and serum analysis: Mice were weighed weekly. At the end of treatment, blood was collected via retro-orbital bleeding, and serum ALT, AST, BUN, and creatinine levels were measured via clinical chemistry assays. - Tumor collection: Mice were euthanized by cervical dislocation. Tumors were dissected, weighed, and stored at -80°C for qPCR and Western blot analysis [1] |
| Toxicity/Toxicokinetics |
1. In vitro toxicity to normal cells: After treating human CD34+ hematopoietic progenitor cells with MI-2 (Menin-MLL inhibitor) (0.5-5 μM) for 72 hours, no significant decrease was observed in cell viability (>85% vs. control group) and colony formation ability (>80% vs. control group) [1] 2. In vivo toxicity: - Body weight: After treating mice with 50 mg/kg MI-2 (Menin-MLL inhibitor) for 21 days, no significant decrease in body weight was observed compared with the solvent control group (<5%). - Organ toxicity: Serum ALT (liver marker), AST (liver marker), BUN (kidney marker), and creatinine (kidney marker) levels were all within the normal physiological range. Histological examination of the liver, kidneys, spleen and bone marrow revealed no drug-induced lesions [1]
3. Plasma protein binding rate: The plasma protein binding rate of MI-2 (Menin-MLL inhibitor) in mouse plasma was approximately 89% (measured by ultrafiltration) [1] |
| References | |
| Additional Infomation |
4-[4-(5,5-dimethyl-4H-thiazo-2-yl)-1-piperazinyl]-6-propylthieno[2,3-d]pyrimidine is an N-arylpiperazine and thienopyrimidine compound.
1. Mechanism of action: MI-2 (Menin-MLL inhibitor) is the first small molecule inhibitor capable of disrupting the Menin-MLL protein-protein interaction. It prevents the formation of the Menin-MLL complex by binding to Menin, which is essential for maintaining the oncogenic transcriptional program (e.g., upregulation of HOXA9 and MEIS1) in MLL rearrangement leukemia [1] 2. Therapeutic potential: MLL rearrangement is common in acute leukemia in children and adults and is associated with poor prognosis. MI-2 (Menin-MLL inhibitor) has shown selective efficacy in MLL rearrangement leukemia models (in vitro and in vivo) and low toxicity to normal cells, supporting its development as a targeted therapy for MLL rearrangement leukemia [1]. 3. Research significance: MI-2 (Menin-MLL inhibitor) can be used as a chemical probe to verify the effectiveness of Menin-MLL interaction as a therapeutic target. It also provides a framework for developing more effective and clinically valuable Menin-MLL inhibitors [1]. |
| Molecular Formula |
C18H25N5S2
|
|
|---|---|---|
| Molecular Weight |
375.55
|
|
| Exact Mass |
375.155
|
|
| CAS # |
1271738-62-5
|
|
| Related CAS # |
|
|
| PubChem CID |
54765302
|
|
| Appearance |
Light yellow to yellow solid powder
|
|
| Density |
1.4±0.1 g/cm3
|
|
| Boiling Point |
538.0±60.0 °C at 760 mmHg
|
|
| Flash Point |
279.2±32.9 °C
|
|
| Vapour Pressure |
0.0±1.4 mmHg at 25°C
|
|
| Index of Refraction |
1.712
|
|
| LogP |
3.59
|
|
| Hydrogen Bond Donor Count |
0
|
|
| Hydrogen Bond Acceptor Count |
6
|
|
| Rotatable Bond Count |
4
|
|
| Heavy Atom Count |
25
|
|
| Complexity |
504
|
|
| Defined Atom Stereocenter Count |
0
|
|
| InChi Key |
SRQYLNYQAPCPIR-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C18H25N5S2/c1-4-5-13-10-14-15(20-12-21-16(14)24-13)22-6-8-23(9-7-22)17-19-11-18(2,3)25-17/h10,12H,4-9,11H2,1-3H3
|
|
| Chemical Name |
4-[4-(5,5-dimethyl-4H-1,3-thiazol-2-yl)piperazin-1-yl]-6-propylthieno[2,3-d]pyrimidine
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.66 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.66 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (6.66 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6628 mL | 13.3138 mL | 26.6276 mL | |
| 5 mM | 0.5326 mL | 2.6628 mL | 5.3255 mL | |
| 10 mM | 0.2663 mL | 1.3314 mL | 2.6628 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04856254 | Not yet recruiting | Procedure: Trial of labor after cesarean section |
Previous Cesarean Section Scar | Mansoura University | August 1, 2020 | Not Applicable |
| NCT05933993 | Recruiting | Other: Elective cesarean section | Cesarean Section Complications | Sygehus Lillebaelt | September 2023 | |
| NCT03967028 | Completed | Procedure: cesarean section | CESAREAN SECTION | Assiut University | November 5, 2019 | |
| NCT04948892 | Recruiting | Other: Prescence of the father during cesarean section in general anesthesia |
Cesarean Section | Sygehus Lillebaelt | January 1, 2022 |
|
 |
 |