
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
GSK503 (GSK-503) is a novel, potent and specific inhibitor of Enhancer of zeste homolog 2 (EZH2) methyltransferase inhibitor with potential antitumor activity. It inhibits EZH2 with Kiapp of 3 to 27 nM. GSK503 demostrates excellent antiproliferative activity and high in vivo antitumor efficacy in a melanoma mouse model.
| Targets |
EZH2 (Enhancer of Zeste Homolog 2, catalytic subunit of PRC2 complex) (IC₅₀ = ~9.8 nM for wild-type EZH2 in recombinant PRC2 complex assay; IC₅₀ = ~15 nM for EZH2 Y641 mutant (Y641F) in the same assay) [1]
- EZH2 (IC₅₀ = ~10 nM for wild-type EZH2 in recombinant PRC2-mediated H3K27 methylation assay; no significant inhibition of Ezh1 (paralog) with IC₅₀ > 10 μM, confirming selectivity) [2] |
|---|---|
| ln Vitro |
GSK503, which shares structural similarities with GSK126 and GSK343, inhibits the methyltransferase activity of both wild type and mutant EZH2 with a similar potency (Kiapp=3-27 nM). GSK503 exhibits a selectivity of over 2000 fold over other histone methyltransferases and over 200 fold over EZH1 (Kiapp=636 nM)[1].
1. Antiproliferative activity in lymphoma cells: GSK503 potently inhibited the proliferation of EZH2-mutant (Y641F) and EZH2-overexpressing B-cell lymphoma cell lines. For SU-DHL-4 (EZH2 Y641F) cells, the IC₅₀ was ~0.3 μM; for OCI-Ly19 (EZH2-overexpressing) cells, the IC₅₀ was ~0.5 μM. It had minimal effect on EZH2-low expressing normal B cells (IC₅₀ > 10 μM) [1] 2. Epigenetic modulation in lymphoma cells: Treatment with GSK503 (0.1–2 μM for 48 h) dose-dependently reduced global H3K27me3 levels in SU-DHL-4 cells. At 1 μM, H3K27me3 was reduced by ~75% (Western blot). It also reactivated EZH2-silenced tumor suppressor genes: p16^(INK4a) mRNA increased by 3.5-fold, and p21^(CIP1) mRNA increased by 2.8-fold (qRT-PCR) [1] 3. Antiproliferative and anti-metastatic activity in melanoma cells: GSK503 suppressed the proliferation of melanoma cell lines (A375, SK-MEL-28) with IC₅₀ values of ~0.4 μM and ~0.6 μM, respectively. It also inhibited melanoma cell migration and invasion: in Transwell assays, 1 μM GSK503 reduced migration by ~60% and invasion by ~55% in A375 cells (crystal violet staining, cell counting) [2] 4. Apoptosis induction in melanoma cells: GSK503 (1 μM for 72 h) induced apoptosis in A375 cells. Annexin V-FITC/PI staining showed apoptotic rates increased from ~4% (vehicle) to ~32% (flow cytometry). This was accompanied by increased cleavage of caspase-3 and PARP (Western blot) [2] 5. Inhibition of melanomagenic pathways: GSK503 (1 μM for 48 h) downregulated genes involved in melanoma progression, including MITF (2.2-fold reduction) and SOX10 (1.8-fold reduction) (qRT-PCR), and reduced the protein levels of MMP9 (matrix metalloproteinase 9, a metastasis marker) by ~60% (Western blot) [2] |
| ln Vivo |
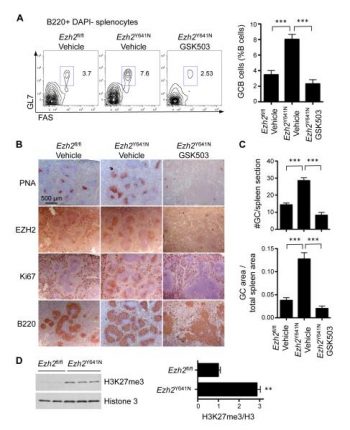
Conditional EZH2 ablation and GSK503 therapy nearly eliminate the production of metastases in a melanoma mouse model while stabilizing the illness through growth inhibition[2]. Normal melanocyte biology is not affected. In mice, GSK503 has good pharmacokinetics. After SRBC or NP-KLH immunization, GSK503 but not the vehicle suppresses the development of the germinal center, phenotyping the Ezh2 null phenotype. GC B-cell counts are decreased by flow cytometry, GC volume and quantity are decreased by immunohistochemistry, and the production of high affinity antibodies is hindered by GSK503 therapy [1].
1. Tumor growth inhibition in lymphoma xenografts: In NOD/SCID mice bearing SU-DHL-4 (EZH2 Y641F) subcutaneous xenografts, GSK503 was administered by oral gavage at 100 mg/kg once daily for 21 days. It significantly inhibited tumor growth: average tumor volume at day 21 was ~200 mm³ (GSK503 group) vs. ~850 mm³ (vehicle group), corresponding to a TGI of ~76%. Tumor weight at sacrifice was ~90 mg (GSK503) vs. ~350 mg (vehicle), a ~74% reduction [1] 2. Survival prolongation in lymphoma models: In a lethal SU-DHL-4 intravenous xenograft model (disseminated lymphoma), GSK503 (100 mg/kg, oral gavage, qd for 21 days) prolonged median survival of mice from ~22 days (vehicle) to ~35 days. At day 40, 30% of GSK503-treated mice survived, while all vehicle-treated mice died [1] 3. Tumor growth and metastasis inhibition in melanoma xenografts: In nude mice bearing A375 subcutaneous xenografts, GSK503 (50 mg/kg, intraperitoneal injection, qd for 18 days) reduced tumor volume by ~70% (day 18: ~180 mm³ vs. ~600 mm³ in vehicle). In a lung metastasis model (A375 cells injected via tail vein), GSK503 (50 mg/kg, i.p., qd for 21 days) reduced the number of lung metastatic nodules by ~80% (hematoxylin-eosin staining, nodule counting) [2] 4. Target validation in vivo: Tumor tissues from GSK503-treated mice (both lymphoma and melanoma models) showed reduced H3K27me3 levels (~65% reduction in lymphoma, ~70% in melanoma) and upregulated tumor suppressor genes (p16^(INK4a) in lymphoma, DKK1 in melanoma) via Western blot and qRT-PCR [1][2] |
| Enzyme Assay |
1. Recombinant PRC2 complex activity assay (lymphoma-focused): Recombinant human PRC2 complex (EZH2-EED-SUZ12) – either wild-type or Y641F mutant – was incubated with biotinylated H3 (1–21) peptide (substrate), S-adenosyl-L-methionine (SAM, methyl donor), and serial concentrations of GSK503 (0.1 nM–10 μM) in assay buffer (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 0.1% BSA) at 37°C for 1 h. The reaction was stopped by adding streptavidin-coated plates and anti-H3K27me3 antibody. Fluorescence intensity was measured, and IC₅₀ was calculated from dose-response curves [1]
2. Recombinant PRC2 complex activity assay (melanoma-focused): Recombinant wild-type PRC2 complex (EZH2-EED-SUZ12) was incubated with fluorescently labeled H3K27 peptide, SAM, and GSK503 (0.01 nM–10 μM) in reaction buffer at 37°C for 90 min. The amount of methylated H3K27 was quantified by measuring fluorescence polarization. The IC₅₀ for wild-type EZH2 was derived, and selectivity was tested against Ezh1-containing PRC2 (IC₅₀ > 10 μM) [2] |
| Cell Assay |
1. MTT antiproliferation assay (lymphoma): SU-DHL-4 or OCI-Ly19 cells were seeded in 96-well plates at 3×10³ cells/well and cultured overnight. Serial concentrations of GSK503 (0.01 μM–20 μM) were added, and cells were incubated for 72 h at 37°C (5% CO₂). MTT reagent was added, and after 4 h, absorbance at 570 nm was measured. IC₅₀ was calculated using nonlinear regression [1]
2. Western blot and qRT-PCR (lymphoma): SU-DHL-4 cells were treated with GSK503 (0.1–2 μM) for 48 h. Nuclear proteins were extracted for Western blot (antibodies against H3K27me3, total H3, p16^(INK4a)); total RNA was extracted for qRT-PCR (primers for p16^(INK4a), p21^(CIP1), GAPDH). Band intensities and mRNA levels were quantified relative to vehicle [1] 3. MTT and apoptosis assay (melanoma): A375 cells were seeded in 96-well plates (2×10³ cells/well) and treated with GSK503 (0.01 μM–20 μM) for 72 h; MTT assay measured proliferation (IC₅₀ calculation). For apoptosis, A375 cells were treated with 1 μM GSK503 for 72 h, stained with Annexin V-FITC/PI, and analyzed by flow cytometry [2] 4. Migration and invasion assay (melanoma): Transwell chambers (uncoated for migration, Matrigel-coated for invasion) were used. A375 cells (5×10⁴ cells/well) in serum-free medium with GSK503 (0.5–1 μM) were added to the upper chamber; medium with serum was added to the lower chamber. After 24 h (migration) or 48 h (invasion), cells on the lower membrane were fixed, stained with crystal violet, and counted. Inhibition rates were calculated relative to vehicle [2] 5. Clonogenic assay (melanoma): A375 cells were seeded in 6-well plates (200 cells/well) and treated with GSK503 (0.1–1 μM) the next day. Cells were cultured for 14 days (medium changed every 3 days), fixed with formaldehyde, stained with crystal violet, and colonies (>50 cells) were counted. Colony formation rate was reduced by ~80% at 1 μM GSK503 [2] |
| Animal Protocol |
Dissolved in 20% captisol; 150 mg/kg; i.p. injection Male SCID mice bearing SUDHL4 and SUDHL6 tumors
1. Lymphoma subcutaneous xenograft model: Female NOD/SCID mice (6–8 weeks old) were subcutaneously injected with 5×10⁶ SU-DHL-4 cells (resuspended in PBS:Matrigel = 1:1) into the right flank. When tumors reached 100–150 mm³, mice were randomized into vehicle (n=6) and GSK503 (n=6) groups. GSK503 was dissolved in DMSO:PEG400:0.9% saline (10:40:50, v/v/v) to 20 mg/mL. Mice received 100 mg/kg GSK503 via oral gavage once daily for 21 days; vehicle group received the same volume of solvent. Tumor volume (length × width² / 2) and body weight were recorded every 3 days. At the end, tumors were excised for Western blot/qRT-PCR [1] 2. Lymphoma disseminated xenograft model: Female NOD/SCID mice were intravenously injected with 2×10⁶ SU-DHL-4 cells via tail vein. Three days later, mice were randomized into vehicle (n=8) and GSK503 (n=8) groups. GSK503 was administered as above (100 mg/kg, oral gavage, qd) for 21 days. Mice were monitored for morbidity (weight loss >20%, lethargy), and survival time was recorded [1] 3. Melanoma subcutaneous xenograft model: Nude mice (6–8 weeks old) were subcutaneously injected with 2×10⁶ A375 cells (PBS:Matrigel = 1:1) into the right flank. When tumors reached 80–120 mm³, mice were divided into vehicle (n=6) and GSK503 (n=6) groups. GSK503 was dissolved in DMSO:corn oil (10:90, v/v) to 10 mg/mL. Mice received 50 mg/kg GSK503 via intraperitoneal injection once daily for 18 days. Tumor volume and body weight were measured every 2 days [2] 4. Melanoma lung metastasis model: Nude mice were intravenously injected with 1×10⁶ A375 cells via tail vein. The next day, mice were randomized into vehicle (n=8) and GSK503 (n=8) groups. GSK503 was administered as above (50 mg/kg, i.p., qd) for 21 days. Mice were euthanized, lungs were fixed with formalin, sectioned, and stained with hematoxylin-eosin. Metastatic nodules were counted [2] |
| ADME/Pharmacokinetics |
1. Oral bioavailability in mice: Female CD-1 mice were administered GSK503 orally (100 mg/kg) orally or intravenously (20 mg/kg). Blood samples were collected at 0.25, 0.5, 1, 2, 4, 8, and 24 hours post-administration. Plasma concentrations of GSK503 were determined using liquid chromatography-tandem mass spectrometry (LC-MS/MS). Oral bioavailability was calculated as ~40% (oral AUC₀₋∞ / intravenous AUC₀₋∞ × intravenous dose / oral dose × 100%) [1]
2. Plasma pharmacokinetics (oral): After oral administration of GSK503 (100 mg/kg) to CD-1 mice, the key parameters were: Cₘₐₓ = ~2.5 μM, Tₘₐₓ = ~1.5 h, t₁/₂ = ~3.2 h, AUC₀₋₂₄ₕ = ~8.5 μM·h [1] 3. Tissue distribution in melanoma model: Nude mice carrying A375 xenograft tumors were administered GSK503 (50 mg/kg, intraperitoneal injection). Tissue samples (tumor, liver, spleen, lung, kidney) were collected 1 hour after administration. The concentrations of GSK503 were approximately 1.8 μM in tumor tissue, approximately 3.0 μM in liver tissue, approximately 2.2 μM in lung tissue, approximately 1.5 μM in spleen tissue, and approximately 1.2 μM in kidney tissue (LC-MS/MS analysis) [2] |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice: Female CD-1 mice were administered GSK503 orally at doses of 150, 200, and 250 mg/kg. No deaths were observed in the 200 mg/kg group; the 250 mg/kg group resulted in the death of 2 out of 6 mice within 48 hours. The LD₅₀ was estimated to be >200 mg/kg and <250 mg/kg [1]
2. Chronic toxicity in a lymphoma model: In a 21-day oral gavage study (100 mg/kg), mice treated with GSK503 did not show significant weight loss (maximum change: 7% decrease compared to the vector group). Serum biochemical indicators (ALT, AST, creatinine, urea) were normal, and hematological indicators (white blood cells, red blood cells, platelets) were normal, indicating no liver or kidney toxicity [1] 3. Chronic toxicity in melanoma model: In an 18-day intraperitoneal injection study (50 mg/kg), mice treated with GSK503 did not show significant weight loss or discomfort. Histopathological examination of liver and kidney tissues showed no significant damage, and serum ALT/AST levels were within the normal range [2] 4. Plasma protein binding rate: GSK503 (1 μM) was incubated with mouse plasma at 37°C for 1 hour. Unbound drug was separated by ultrafiltration (30 kDa molecular weight cutoff) and measured by LC-MS/MS. The plasma protein binding rate was approximately 95% [1][2] |
| References |
|
| Additional Infomation |
GSK503 is a potent and specific EZH2 methyltransferase inhibitor. 1. Mechanism of action: GSK503 is a competitive inhibitor of EZH2. It binds to the SET domain of EZH2 and blocks PRC2-mediated H3K27 trimethylation. Reduced H3K27me3 levels can reactivate silenced tumor suppressor genes, thereby inhibiting cell proliferation, inducing apoptosis, and inhibiting metastasis [1][2]. 2. Background of lymphoma treatment: EZH2 mutations (e.g., Y641F) drive lymphocyte transformation by enhancing H3K27me3 levels and silencing tumor suppressor genes. GSK503 can target both mutant and wild-type EZH2, making it a potential therapy for EZH2-driven B-cell lymphoma [1]
3. Background for melanoma treatment: EZH2 is overexpressed in melanoma and promotes tumor growth/metastasis by silencing metastasis inhibitors (e.g., DKK1) and activating melanoma development pathways. GSK503 can reverse these effects, supporting its use in the treatment of melanoma [2] 4. Selectivity: GSK503 has much higher selectivity for EZH2 than other histone methyltransferases (e.g., G9a, SUV39H1) and kinases, thereby minimizing off-target effects [1][2] |
| Molecular Formula |
C31H38N6O2
|
|
|---|---|---|
| Molecular Weight |
526.67
|
|
| Exact Mass |
526.305
|
|
| CAS # |
1346572-63-1
|
|
| Related CAS # |
|
|
| PubChem CID |
67469117
|
|
| Appearance |
White to gray solid powder
|
|
| Density |
1.2±0.1 g/cm3
|
|
| Boiling Point |
798.6±60.0 °C at 760 mmHg
|
|
| Flash Point |
436.8±32.9 °C
|
|
| Vapour Pressure |
0.0±2.8 mmHg at 25°C
|
|
| Index of Refraction |
1.648
|
|
| LogP |
3.3
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
5
|
|
| Rotatable Bond Count |
6
|
|
| Heavy Atom Count |
39
|
|
| Complexity |
984
|
|
| Defined Atom Stereocenter Count |
0
|
|
| InChi Key |
HRDQQHUKUIKFHT-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C31H38N6O2/c1-19(2)37-18-21(4)29-25(30(38)33-17-26-20(3)13-22(5)34-31(26)39)14-24(15-27(29)37)23-7-8-28(32-16-23)36-11-9-35(6)10-12-36/h7-8,13-16,18-19H,9-12,17H2,1-6H3,(H,33,38)(H,34,39)
|
|
| Chemical Name |
N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-isopropyl-3-methyl-6-(6-(4-methylpiperazin-1-yl)pyridin-3-yl)-1H-indole-4-carboxamide
|
|
| Synonyms |
GSK 503; GSK503; GSK-503
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.75 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.75 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8987 mL | 9.4936 mL | 18.9872 mL | |
| 5 mM | 0.3797 mL | 1.8987 mL | 3.7974 mL | |
| 10 mM | 0.1899 mL | 0.9494 mL | 1.8987 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
 |
|---|