
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g | |||
| Other Sizes |
Purity: ≥98%
Ganetespib (formerly STA-9090; STA 9090; STA9090), the active metabolite of STA-1474 (a prodrug of ganetespib), is a potent and triazolone-based small-molecule HSP90 (heat shock protein 90) inhibitor with potential anticancer activity. It inhibits HSP90 with an IC50 of 4 nM in OSA 8 cells, and induces apoptosis of OSA cells while normal osteoblasts are not affected. Ganetespib is being investigated for the treatment of various cancers such as Small Cell Lung Cancer, breast cancer, Acute Myeloid Leukaemia, and Myelodysplastic Syndrome.
| Targets |
HSP90
Heat Shock Protein 90 (HSP90): Ganetespib (STA-9090) is a selective, non-geldanamycin inhibitor of HSP90, binding to the ATP-binding pocket of the active conformation of HSP90. In purified human recombinant HSP90α ATPase activity assays, its IC50 was 5 nM, ~10-fold more potent than 17-AAG (IC50=50 nM) [2]; it also inhibited HSP90β (IC50=8 nM) and GRP94 (IC50=12 nM) with high selectivity [2] - No direct binding to downstream client proteins (e.g., EGFR, Akt, JAK2, MYC), but induces their ubiquitin-dependent degradation by inhibiting HSP90 chaperone function [1][4][5] |
|---|---|
| ln Vitro |
In genomically characterized NSCLC cell lines, genesetib induces apoptosis, suppresses downstream signaling, depletes receptor tyrosine kinases, and inhibits proliferation with IC50 values ranging from 2 to 30 nM. Additionally, in isogenic Ba/F3 pro-B cells that have been made IL-3 independent through the production of EGFR and ERBB2 mutants, genetecib is around 20 times more potent[1]. In vitro, genetecib is more effective than 17-allylamino-17-demethoxygeldanamycin (17-AAG), an ansamycin inhibitor, at causing the degradation of known Hsp90 client proteins. It also shows strong cytotoxicity in a variety of solid and hematologic tumor cell lines[2]. Strong HSP90 inhibitor genesetespib has been demonstrated to destroy canine tumor cell lines in vitro[3]. In the HEL92.1.7 cells, genesetib exhibits more potent or prolonged JAK/STAT inhibitory activity compared to P6 and 17-AAG[4].
Non-Small Cell Lung Cancer (NSCLC) cells (A549, H1975, H226, literature [1]): - Antiproliferative activity: Ganetespib (1-50 nM) dose-dependently inhibited cell viability (MTT assay, 72 h): IC50=7 nM (A549), 9 nM (H1975), 8 nM (H226). At 20 nM, cell viability was reduced by 80-85% vs. control [1] - Client protein degradation: 15 nM Ganetespib for 24 h reduced HSP90 client proteins: EGFR (75%), phospho-Akt (p-Akt, 80%), phospho-ERK (p-ERK, 70%) (Western blot); no significant change in total HSP90 or HSP70 (a hallmark of HSP90 inhibition, upregulated 3.5-fold) [1] - Apoptosis induction: 20 nM Ganetespib increased apoptotic rate (Annexin V-FITC/PI staining) from 4% (control) to 42% (A549, 48 h); cleaved caspase-3/9 were upregulated 3.0-3.2-fold (Western blot) [1] - Broad-spectrum anticancer activity: - Antiproliferative activity on diverse cancer cells: Ganetespib (0.5-20 nM) inhibited viability (SRB assay, 72 h): IC50=3 nM (SK-BR-3 breast cancer), 5 nM (PC-3 prostate cancer), 6 nM (SU-DHL-4 lymphoma). At 10 nM, viability reduced by 70-75% [2] - Normal cell selectivity: IC50 for normal human fibroblasts (MRC-5) was 85 nM, ~17-fold higher than cancer cells (SK-BR-3: 3 nM) [2] - JAK/STAT signaling inhibition: - JAK2/STAT3 degradation: Ganetespib (10-30 nM) dose-dependently reduced JAK2 (70%) and phospho-STAT3 (p-STAT3, 80%) in HEL cells (JAK2V617F-mutant) after 24 h (Western blot). 20 nM inhibited STAT3 transcriptional activity by 75% (luciferase assay) [4] - Cytokine-induced STAT activation suppression: 15 nM Ganetespib blocked IL-6-induced p-STAT3 in U266 cells by 80% [4] - Rhabdomyosarcoma (RMS) cells: - MYC downregulation: Ganetespib (10-25 nM) reduced MYC protein by 65-75% (Western blot) in RD and RH30 RMS cells after 24 h. 20 nM inhibited RMS cell proliferation by 70% (BrdU assay) [5] - Client protein (Akt, IGF-1R) degradation: 20 nM reduced Akt by 70% and IGF-1R by 65% [5] |
| ln Vivo |
In NCI-H1975 xenografts, genetecib (125 mg/kg, IV) accumulates in tumors more than in normal tissues and has higher in vivo efficacy than 17-AAG without causing additional toxicity. It also slows proliferation and promotes apoptosis together with EGFR depletion[1]. (100, 125, 150 mg/kg, iv) significantly inhibits development and/or reverses tumor growth in solid and hematologic xenograft models of oncogene addiction, demonstrating strong anticancer efficacy[2].
NSCLC nude mouse xenograft model: - Animals and grouping: Female nude mice (6-8 weeks, 20-22 g, n=6/group) randomized into: Vehicle (5% DMSO/PBS, i.v.), Ganetespib 10 mg/kg (i.v.), Ganetespib 25 mg/kg (i.v.) [1] - Tumor induction and treatment: 5×10⁶ A549 cells (1:1 Matrigel/PBS) injected subcutaneously into right flank. When tumors reached ~100 mm³, drugs administered once weekly for 3 weeks (i.v. via tail vein) [1] - Efficacy outcomes: 25 mg/kg group showed 80% tumor volume inhibition (tumor volume: 210 ± 25 mm³ vs. vehicle 1050 ± 60 mm³) and 75% tumor weight reduction (0.3 ± 0.05 g vs. 1.2 ± 0.1 g). Tumor tissues had EGFR (0.25-fold) and p-Akt (0.2-fold) downregulation (Western blot) [1] - Broad-spectrum xenograft efficacy: - SK-BR-3 nude mouse model: Ganetespib 20 mg/kg (i.v., once weekly × 3) reduced tumor weight by 78% (0.22 ± 0.03 g vs. vehicle 1.0 ± 0.1 g). No significant body weight loss (21.5 ± 1.0 g vs. 22.0 ± 1.1 g) [2] - PC-3 model: 20 mg/kg reduced tumor volume by 72% (230 ± 30 mm³ vs. 820 ± 50 mm³) [2] - Phase I study in dogs with spontaneous cancer: - Animals and dosing: 12 dogs (various cancers: osteosarcoma, mammary carcinoma) dosed with Ganetespib (prodrug STA-1474, converted to Ganetespib in vivo) at 0.5, 1, 2, 5, 10 mg/kg (i.v., once every 2 weeks) [3] - Efficacy and toxicity: Maximum tolerated dose (MTD) was 5 mg/kg; 10 mg/kg caused grade 2 diarrhea. 2 dogs with mammary carcinoma showed stable disease (tumor volume unchanged for 8 weeks); 1 dog with osteosarcoma had 30% tumor reduction [3] |
| Enzyme Assay |
HSP90 binding assays[1]
Exponentially growing cells were processed in lysis buffer (20 mM HEPES, pH 7.4, 1 mM EDTA, 5 mM MgCl2, 100 mM KCl) and incubated with increasing concentrations of 17-AAG or ganetespib for 30 min at 4°C, and incubated with biotin-GM linked to Dynabeads MyOne Streptavidin T1 magnetic bead for 1 h at 4°C. Beads were washed three times in lysis buffer and heated for 5 min at 95°C in SDS–PAGE sample buffer (Invitrogen). Samples were resolved on 4-12% Bis-Tris gradient gel and Western blots were performed using an anti-HSP90 antibody. 1. Reagent preparation: Prepare 50 mM Tris-HCl buffer (pH 7.5) containing 10 mM MgCl₂, 2 mM DTT, and 0.1 mg/mL BSA. Purify human recombinant HSP90α (0.4 μg/well) and prepare [γ-³²P]-ATP (1 μCi/μL, final concentration 5 μM) [2] 2. Reaction setup: Add 80 μL buffer, 10 μL Ganetespib (0.1-50 nM) or vehicle (0.1% DMSO), and 5 μL HSP90α to a 96-well plate. Incubate at 37℃ for 15 min to allow drug-HSP90 binding [1] 3. ATP hydrolysis initiation and termination: Add 5 μL [γ-³²P]-ATP to start the reaction, incubate at 37℃ for 45 min. Terminate with 100 μL 20% trichloroacetic acid (TCA), incubate on ice for 20 min [2] 4. Detection and calculation: Centrifuge at 3500 × g for 15 min, transfer 100 μL supernatant to activated charcoal-coated plates (adsorbs unhydrolyzed ATP). Wash 3 times with 5% TCA, dry plates, and measure radioactivity (³²P-phosphate) via liquid scintillation counter. HSP90 activity = (treatment radioactivity / control radioactivity) × 100%. IC50 calculated via dose-response fitting (GraphPad Prism) [1] |
| Cell Assay |
Cell proliferation assay[1]
Cell proliferation assays were performed using the CCK-8 colorimetric assay in at least duplicate samples according to the manufacturer’s specifications. IC50 values were calculated using Kaleidagraph or Graphpad Prism. Western blots[1] Whole-cell lysates were prepared as previously described (9). Protein concentrations were determined and equivalent amounts (20 μg) were subjected to SDS-PAGE on 4-12% bis-tris gradient gels (Invitrogen). The HSP27 antibody was from Enzo Life Sciences. p23 and HSP90αantibodies were from StressMarq. Immunoprecipitation[1] 500 μg of whole cell lysate was immunoprecipitated with 2 μg of mouse anti-p23 monoclonal antibody conjugated with protein A Dynabeads. Proteins bound to p23 were resolved on 4-12% bis-tris gradient gels and Western blot was performed with an anti-HSP90 antibody. 1. Cancer Cell Proliferation Assay (MTT/SRB, literature [1][2]) 1. Cell seeding: NSCLC cells (A549, 5×10³/well) or breast cancer cells (SK-BR-3, 4×10³/well) seeded into 96-well plates, incubated at 37℃, 5% CO₂ for 24 h [1][2] 2. Drug treatment: Replace medium with fresh medium containing Ganetespib (0.1-50 nM) or vehicle. Incubate 72 h (A549: MTT assay; SK-BR-3: SRB assay) [1][2] 3. MTT detection: Add 20 μL MTT (5 mg/mL) to A549 wells, incubate 4 h. Aspirate supernatant, add 150 μL DMSO, measure OD570 nm. Viability = (treatment OD / control OD) × 100% [1] 4. SRB detection: Fix SK-BR-3 cells with 50 μL 50% TCA (4℃, 1 h), wash 5× with water, stain with 0.4% SRB (30 min). Wash 4× with 1% acetic acid, dissolve in 10 mM Tris base, measure OD515 nm. IC50 derived from dose-response curves [2] ### 2. Western Blot for Client Proteins (literature [1][4][5]) 1. Protein extraction: Cells (A549, HEL, RD) treated with Ganetespib (10-25 nM) for 24 h. Lyse in RIPA buffer (with protease/phosphatase inhibitors), centrifuge 12,000 × g, 15 min, 4℃ to collect supernatant [1][4][5] 2. Electrophoresis and transfer: Load 30 μg protein onto 10% SDS-PAGE, run 100 V for 2 h. Transfer to PVDF membrane (15 V, 30 min, semi-dry) [1][4] 3. Antibody incubation: Block membrane with 5% non-fat milk/TBST 1 h. Incubate primary antibodies (anti-EGFR, anti-p-Akt, anti-JAK2, anti-MYC, anti-GAPDH) overnight at 4℃. Wash 3× with TBST, incubate HRP-secondary antibody 1 h at room temperature [4][5] 4. Detection: ECL kit for band visualization. Quantify intensity via ImageJ (e.g., A549: EGFR intensity 0.25-fold of control at 15 nM Ganetespib) [1][5] ### 3. Apoptosis Assay (Annexin V-FITC/PI, literature [1]) 1. Cell preparation: A549 cells (2×10⁵/well) seeded into 6-well plates, incubated 24 h, treated with Ganetespib (10-25 nM) for 48 h [1] 2. Cell collection: Trypsinize (without EDTA), collect cells, wash 2× with cold PBS. Resuspend in 1× binding buffer (1×10⁶ cells/mL) [1] 3. Staining and analysis: Add 5 μL Annexin V-FITC and 5 μL PI to 100 μL suspension, incubate 15 min in dark. Add 400 μL binding buffer, analyze via flow cytometry. Apoptotic rate = Annexin V⁺/PI⁻ + Annexin V⁺/PI⁺ [1] ### 4. STAT3 Luciferase Assay 1. Cell transfection: HEL cells (2×10⁵/well, 24-well plate) transfected with STAT3-responsive luciferase plasmid and Renilla plasmid (internal control) via transfection reagent. Incubate 24 h [4] 2. Drug treatment: Replace medium with Ganetespib (5-30 nM) or vehicle. Incubate 24 h [4] 3. Luminescence detection: Lyse cells with passive lysis buffer, measure firefly (L1) and Renilla (L2) luciferase activity. Relative STAT3 activity = (L1/L2)treatment / (L1/L2)control [4] |
| Animal Protocol |
Dissolved in DMSO and diluted 1:10 with 20% Cremophor RH 40; 25 mg/kg; Tail vein injection
Female severe combined immune-deficient (SCID) mice Establishment and treatment of xenografts[1] Female 7–8-week-old C.B-17 SCID mice were maintained under pathogen-free conditions. All procedures were approved by the Synta Pharmaceutical Institutional Animal Care and Use Committee. NCI-H1975 or HCC827 cells were cultured as above and 0.5 – 1×107 cells were mixed with 50% RPMI 1640/50% Matrigel and subcutaneously injected into the flanks of SCID mice. For efficacy studies, animals with 100-200 mm3 tumors were then randomized into treatments groups of eight. Tumor volumes (V) were calculated by the equation V = 0.5236×L×W×T (Length, width, and thickness). Animals were treated by intravenous bolus tail vein injection at 10 ml/kg with ganetespib formulated in 10/18 DRD (10% DMSO, 18% Cremophor RH 40, 3.6% dextrose and 68.4% water). As a measurement of in vivo efficacy, the relative size of treated and control tumors [(%T/C) value] was determined from the change in average tumor volumes of each drug-treated group relative to the vehicle-treated group, or itself in the case of tumor regression. Body weights were monitored daily. For biomarker studies, mice bearing NCI-H1975 xenografts were treated with either a single dose of vehicle or ganetespib, or with 5 daily doses of vehicle or ganetespib, in groups of 3 or 8, and harvested at various time points. Tumors were excised and flash frozen in liquid nitrogen for preparation of protein lysates or fixed in 10% neutral buffered formalin for immunohistochemistry. Pharmacokinetic Analysis[1] Female 7–8-week-old C.B-17 SCID mice bearing NCI-H1975 xenografts received a single intravenous (i.v.) dose slightly below the highest non-severely toxic dose (HNSTD, 150 mg/kg). At time points indicated, mice (n = 3/time point) were sacrificed and plasma and tissues (tumor, liver, and lung) were harvested. Concentrations of ganetespib in plasma and tissues were determined by isocratic reversed-phase high-performance liquid chromatography with electrospray ionization mass spectrometric (HPLC/MS-MS) detection. 1. NSCLC Nude Mouse Xenograft Model 1. Animal preparation: Female nude mice (6-8 weeks, 20-22 g, n=18) housed under SPF conditions (12 h light/dark cycle, 22±2℃), free access to food/water. Acclimate 1 week [1] 2. Tumor induction: 5×10⁶ A549 cells (0.2 mL, 1:1 Matrigel/PBS) injected subcutaneously into right dorsal flank of each mouse [1] 3. Grouping and treatment: When tumors reached ~100 mm³, randomize into 3 groups (n=6/group): - Vehicle: Intravenous injection (tail vein) of 5% DMSO/PBS, once weekly for 3 weeks. - Ganetespib 10 mg/kg: IV injection of Ganetespib (10 mg/kg, dissolved in 5% DMSO/PBS), once weekly × 3. - Ganetespib 25 mg/kg: IV injection of Ganetespib (25 mg/kg, same solvent), once weekly × 3 [1] 4. Sample collection: Measure tumor volume (length×width²/2) and body weight every 3 days. Euthanize on day 21, dissect tumors to weigh; tumor tissues lysed for Western blot (EGFR, p-Akt) [1] ### 2. Phase I Study in Dogs with Spontaneous Cancer 1. Animal selection: 12 client-owned dogs (3-8 years old, 10-30 kg) with histologically confirmed cancer (4 osteosarcoma, 3 mammary carcinoma, 2 melanoma, 3 others). Screened for normal liver/kidney function [3] 2. Dosing and schedule: Dogs divided into 5 dose cohorts (0.5, 1, 2, 5, 10 mg/kg) of 2-3 dogs each. Administer Ganetespib prodrug (STA-1474) via IV infusion (30 min) once every 2 weeks. Monitor for adverse events (AE) daily for 7 days post-dose [3] 3. Efficacy and toxicity assessment: Every 4 weeks, measure tumor size via CT/ultrasound. Grade AEs per Veterinary Cooperative Oncology Group (VCOG) criteria. MTD defined as highest dose with no grade ≥3 AEs [3] |
| ADME/Pharmacokinetics |
Canine pharmacokinetics:
- Absorption:Ganetespib (derived from the prodrug STA-1474) undergoes rapid conversion after intravenous administration (conversion half-life: 0.3 hours)[3] - Distribution: Volume of distribution (Vd) = 1.2 L/kg; can penetrate into tumor tissue (tumor/plasma concentration ratio of 0.8 2 hours after administration)[3] - Elimination: Half-life (t₁/₂) = 1.2 hours; clearance (CL) = 0.8 L/kg/h; 70% is excreted in feces within 72 hours (in unchanged form)[3] |
| Toxicity/Toxicokinetics |
In vitro toxicity: - Safety in normal cells: Ganetespib (≤50 nM) showed >85% survival in normal human bronchial epithelial cells (BEAS-2B) and MRC-5 fibroblasts (MTT test, 72 hours) [1][2] - No genotoxicity: Ames test results were negative (10-1000 nM Ganetespib) [2] - In vivo toxicity: - Nude mice: Ganetespib 25 mg/kg (IV injection, 3 weeks) did not cause weight loss (21.8 ± 1.0 g vs. carrier 22.2 ± 1.1 g) or organ damage (liver/kidney HE staining: no necrosis/inflammation). Serum ALT (28 ± 4 U/L vs. 30 ± 5 U/L) and BUN (14 ± 2 mg/dL vs. 15 ± 2 mg/dL) were within the normal range [1][2]
- Dogs: Maximum tolerated dose (MTD) = 5 mg/kg (intravenous, every 2 weeks). A dose of 10 mg/kg resulted in grade 2 diarrhea (3/3 dogs) and grade 1 vomiting (2/3 dogs); no hematologic toxicity or organ damage was observed. Serum ALP (alkaline phosphatase) levels remained normal (80 ± 10 U/L vs. baseline 75 ± 8 U/L) [3] - Plasma protein binding: Ganetespib had a plasma protein binding rate of 98% in human and canine plasma (ultrafiltration) [2][3] |
| References |
|
| Additional Infomation |
Ganetespib belongs to the triazole cyclic compound class. Ganetespib is currently under investigation for the treatment of breast cancer, small cell lung cancer, acute myeloid leukemia, and myelodysplastic syndromes. Ganetespib is a synthetic small-molecule heat shock protein 90 (Hsp90) inhibitor with potential antitumor activity. Ganetespib binds to Hsp90 and inhibits its activity, leading to proteasomal degradation of oncogenic substrate proteins, inhibition of cell proliferation, and elevated levels of heat shock protein 72 (Hsp72). It may inhibit the activity of various kinases, such as c-Kit, EGFR, and Bcr-Abl, which, as substrate proteins, depend on functional Hsp90 for maintenance. Hsp90 is a 90 kDa molecular chaperone upregulated in various tumor cells, playing a crucial role in the conformational maturation, stability, and function of intracellular "client" proteins, most of which are involved in signal transduction, cell cycle regulation, and apoptosis, including kinases, transcription factors, and hormone receptors. Hsp72 has anti-apoptotic function; its upregulation can serve as a surrogate marker for Hsp90 inhibition. Ganetespib (STA-9090) is a non-galdmycin HSP90 inhibitor with a unique triazolone skeleton designed to overcome the limitations of galdmycin analogs (e.g., 17-AAG) such as poor solubility and hepatotoxicity. It has higher HSP90 inhibitory efficacy (IC50=5 nM, while 17-AAG=50 nM) and better safety[2]
- Mechanism of action: It binds to the ATP-binding pocket of HSP90, disrupts molecular chaperone function, and induces ubiquitin-dependent degradation of cancer substrate proteins (EGFR, Akt, JAK2, MYC) and pro-survival signals, thereby inhibiting cancer cell proliferation and inducing apoptosis[1][4][5] - Therapeutic potential: - Non-small cell lung cancer (NSCLC): It is effective (tumor inhibition rate >75%) and non-toxic in the A549/H1975 xenograft model, supporting its use in NSCLC[1] - Spontaneous canine cancer: Phase I clinical trials showed stable/shrinking breast cancer/osteosarcoma, validating its translational potential in human cancer treatment[3] - Rhabdomyosarcoma (RMS): Downregulates MYC (RMS (key driver factors) and IGF-1R, representing a targeted therapy for MYC-driven RMS [5] |
| Molecular Formula |
C20H20N4O3
|
|
|---|---|---|
| Molecular Weight |
364.4
|
|
| Exact Mass |
364.153
|
|
| Elemental Analysis |
C, 65.92; H, 5.53; N, 15.38; O, 13.17
|
|
| CAS # |
888216-25-9
|
|
| Related CAS # |
|
|
| PubChem CID |
135564985
|
|
| Appearance |
White to yellow solid powder
|
|
| Density |
1.4±0.1 g/cm3
|
|
| Boiling Point |
685.8±57.0 °C at 760 mmHg
|
|
| Flash Point |
368.5±32.1 °C
|
|
| Vapour Pressure |
0.0±2.2 mmHg at 25°C
|
|
| Index of Refraction |
1.698
|
|
| LogP |
5.47
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
3
|
|
| Heavy Atom Count |
27
|
|
| Complexity |
610
|
|
| Defined Atom Stereocenter Count |
0
|
|
| InChi Key |
RVAQIUULWULRNW-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C20H20N4O3/c1-11(2)14-9-15(18(26)10-17(14)25)19-21-22-20(27)24(19)13-4-5-16-12(8-13)6-7-23(16)3/h4-11,25-26H,1-3H3,(H,22,27)
|
|
| Chemical Name |
5-[2,4-dihydroxy-5-(1-methylethyl)phenyl]-4-(1-methyl-1H-indol-5-yl)-2,4-dihydro-3H- 1,2,4-triazol-3-one
|
|
| Synonyms |
STA-9090; Ganetespib; STA 9090; STA9090
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 10 mg/mL (27.44 mM) in 15% Cremophor EL + 85% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication.
Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.86 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (6.86 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: ≥ 2.5 mg/mL (6.86 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL corn oil and mix evenly. Solubility in Formulation 5: 1% DMSO+30% polyethylene glycol+1% Tween 80: 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7442 mL | 13.7212 mL | 27.4424 mL | |
| 5 mM | 0.5488 mL | 2.7442 mL | 5.4885 mL | |
| 10 mM | 0.2744 mL | 1.3721 mL | 2.7442 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02008877 | Completed Has Results | | Malignant Peripheral Nerve |
Sheath Tumors (MPNST) Sarcoma Alliance for Research |
through Collaboration December 2013 |
Phase 1 |
Phase 2 |
| NCT02192541 | Terminated Has Results | Drug: Ziv-Aflibercept Drug: Ganetespib |
Neoplasms | National Cancer Institute (NCI) | December 2, 2014 | Phase 1 |
| NCT01554969 | Completed | Drug: capecitabine + ganetespib | Rectal Cancer | Emory University | May 2012 | Phase 1 |
| NCT01485835 | Completed | Drug: Ganetespib Drug: Bortezomib |
Multiple Myeloma | Emory University | January 2012 | Phase 1 |
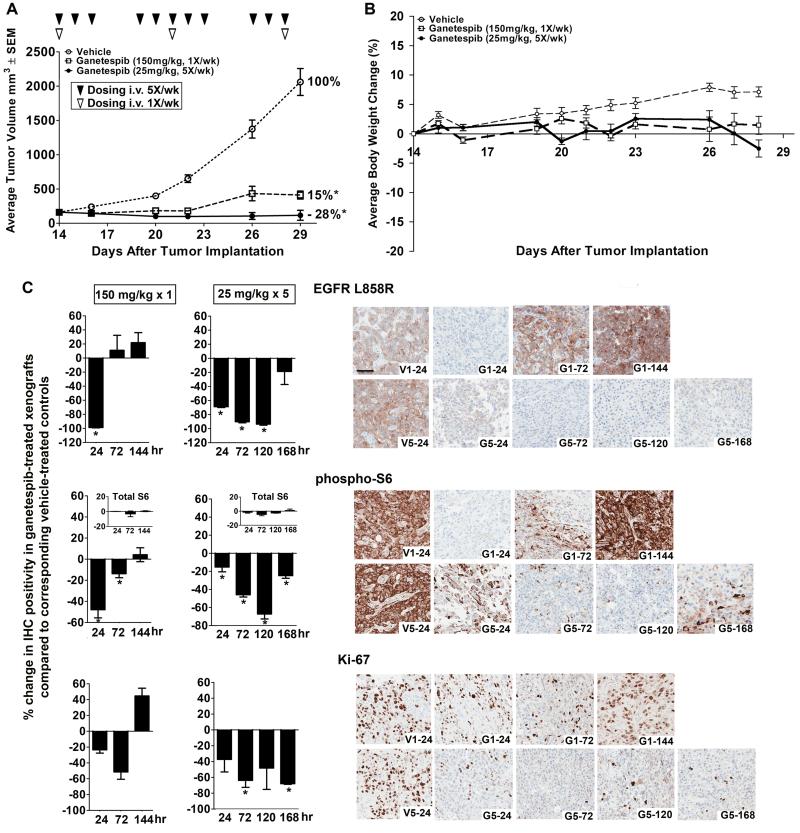 Comparison of once-weekly and five times per week dosing of ganetespib.Clin Cancer Res.2012 Sep 15;18(18):4973-85. |
|---|
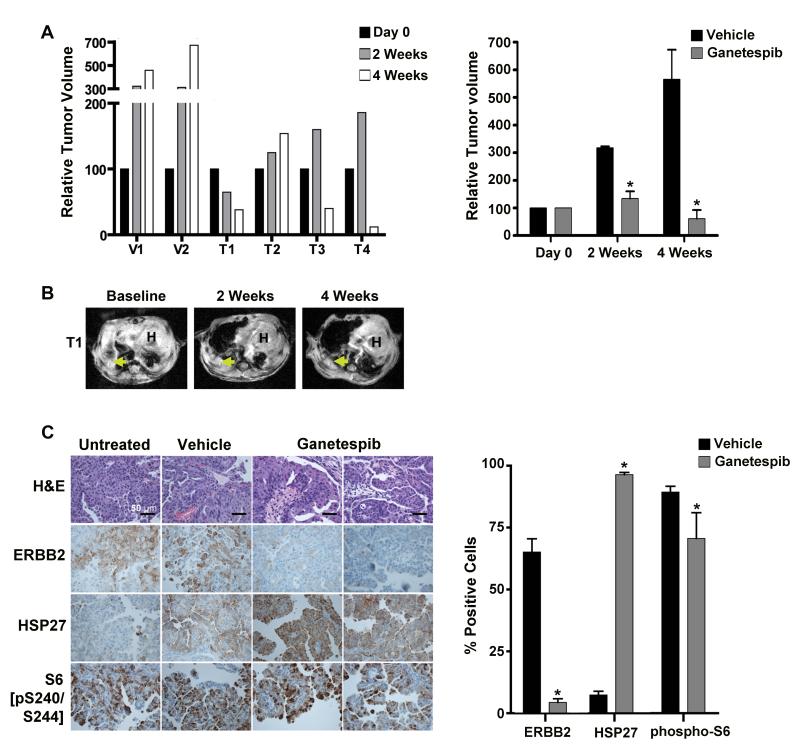 Ganetespib induces tumor regression in a mouse lung carcinoma model driven by ERRB2YVMA.Clin Cancer Res.2012 Sep 15;18(18):4973-85. |
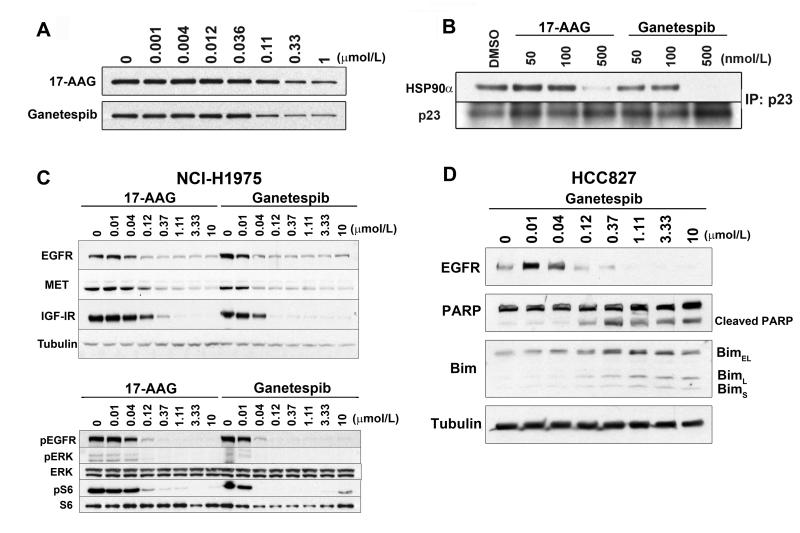 Ganetespib accumulates in tumor relative to normal tissues and displays greaterin vivoefficacy than 17-AAG.Clin Cancer Res.2012 Sep 15;18(18):4973-85. |