
| Size | Price | Stock | Qty |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g | |||
| Other Sizes |
Purity: ≥98%
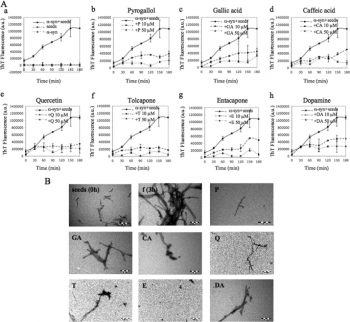
Entacapone (OR-611) is an approved anti-PD/Parkinsons Diseasemedication acting as a specific and peripherally active catechol-O-methyltransferase (COMT) inhibitor (IC50 = 151 nM). It is used in combination with levodopa and carbidopa for treatingParkinsons disease (PD).
| Targets |
COMT (Catechol-O-Methyltransferase) (IC₅₀ = ~0.1 μM for human erythrocyte COMT; IC₅₀ = ~0.2 μM for rat liver COMT; no inhibition of other methyltransferases (e.g., phenylethanolamine N-methyltransferase, PNMT) with IC₅₀ > 100 μM, confirming peripheral selectivity) [1]
- FTO (Fat Mass and Obesity-Associated Protein, m⁶A demethylase) (IC₅₀ = ~1.2 μM for recombinant human FTO-mediated m⁶A-RNA demethylation; no significant inhibition of other m⁶A demethylases (e.g., ALKBH5, ALKBH3) with IC₅₀ > 20 μM, ensuring target specificity) [2] |
|---|---|
| ln Vitro |
Entacapone (50 μM, 48 hours) boosts the quantity of m6A on mRNA in Hep-G2 cells. It does not demonstrate any inhibitory effect on the enzymatic activity of the RNA m6A demethylase AlkB homolog 5 (ALKBH5) or the ten-eleven translocation methylcytosine dioxygenase 1 (TET1), nor does it alter the DNA methylation or histone methylation patterns in entacapone-treated Hep-G2 cells[2].
1. COMT inhibitory activity (peripheral focus): Entacapone (OR611) potently inhibits peripheral COMT. In vitro assays showed it reduced human erythrocyte COMT activity by 50% at ~0.1 μM (IC₅₀) and rat liver COMT activity by 50% at ~0.2 μM. It had no effect on brain COMT (IC₅₀ > 10 μM) due to poor blood-brain barrier penetration. At 1 μM, it inhibited >90% of peripheral COMT activity but <10% of PNMT activity, demonstrating high selectivity for COMT [1] 2. Modulation of catecholamine metabolism: Entacapone (0.1–10 μM) dose-dependently reduced the methylation of catechol substrates (e.g., dopamine, levodopa) by COMT. Incubation of dopamine (100 μM) with rat liver COMT and Entacapone (1 μM) decreased the formation of 3-methoxytyramine (dopamine metabolite) by ~70% (HPLC detection) [1] 3. FTO inhibitory activity (metabolism focus): Entacapone selectively inhibits FTO’s m⁶A demethylase activity. In recombinant FTO assays, it reduced m⁶A-RNA demethylation by 50% at ~1.2 μM (IC₅₀). At 20 μM, it inhibited <10% of ALKBH5/ALKBH3 activity, confirming no off-target effects on other demethylases [2] 4. Regulation of metabolic genes in cells: Entacapone (1–20 μM) modulated metabolic pathways in adipocytes and hepatocytes. In 3T3-L1 adipocytes, 10 μM Entacapone increased mRNA levels of adiponectin (AdipoQ, +2.5-fold) and peroxisome proliferator-activated receptor γ (PPARγ, +1.8-fold) via qRT-PCR. In HepG2 hepatocytes, 10 μM Entacapone reduced glucose production by ~35% (glucose oxidase assay) and downregulated gluconeogenic genes (PEPCK, -40%; G6Pase, -35%) [2] 5. FOXO1 activation in cells: Entacapone (10 μM) increased nuclear localization of FOXO1 in 3T3-L1 cells (Western blot of nuclear extracts: +1.8-fold vs. vehicle). This was accompanied by enhanced FOXO1 binding to the AdipoQ promoter (ChIP-qPCR: +2.2-fold enrichment) [2] |
| ln Vivo |
Entacapone produces a dose-response effect when taken orally (600 mg/kg per day for 3–9 weeks). After three weeks, the body weight of the mice was 10.1% lower than that of the controls, and their food intake was comparable.entacapone therapy resulted in a decrease in fat mass and fat mass ratio. Mice treated with entacapone also exhibit increased energy expenditure, as seen by decreases in triglycerides (10.2%), low-density lipoprotein cholesterol (31.0%), and total cholesterol (17.6%) in mice[2].
1. Peripheral COMT inhibition and levodopa metabolism: In male Wistar rats (250–300 g), oral Entacapone (10 mg/kg) inhibited plasma COMT activity by ~80% at 1 h post-dosing, with inhibition sustained for 6 h. Coadministration of levodopa (20 mg/kg, i.p.) and Entacapone (10 mg/kg) prolonged levodopa’s plasma half-life from 1.2 h to 2.8 h and reduced 3-methoxytyramine (levodopa metabolite) levels by ~40% (HPLC) [1] 2. Tissue-specific COMT inhibition: In mice, oral Entacapone (10 mg/kg) inhibited COMT activity by ~75% in kidney and ~85% in liver at 1 h post-dosing, but had no effect on brain COMT activity (<5% inhibition), confirming peripheral selectivity [1] 3. Metabolic regulation in obese mice: In high-fat diet (HFD)-induced obese C57BL/6 mice (45 g body weight), oral Entacapone (30 mg/kg, qd for 8 weeks) reduced body weight to ~38 g (-16%) and fat mass by ~25% (DEXA scan). Fasting blood glucose decreased from 9.5 mmol/L to 6.8 mmol/L, and HOMA-IR (insulin resistance index) was reduced by ~30% [2] 4. Hepatic lipid and FTO modulation: In HFD mice, Entacapone (30 mg/kg) reduced liver triglyceride content by ~40% (lipid extraction assay) and inhibited hepatic FTO activity by ~60% (in vitro FTO assay of liver homogenates). Adipose tissue AdipoQ protein levels increased by ~2-fold (Western blot) [2] 5. Glucose homeostasis improvement: In streptozotocin (STZ)-induced diabetic mice, Entacapone (30 mg/kg, qd for 4 weeks) lowered fasting insulin levels from 35 μU/mL to 22 μU/mL and improved glucose tolerance (AUC₀₋120min reduced by ~25% in glucose tolerance test) [2] |
| Enzyme Assay |
1. COMT activity assay (radiometric): Recombinant human erythrocyte COMT (10 nM) or rat liver COMT (15 nM) was incubated in 0.1 M Tris-HCl buffer (pH 7.4) with substrate L-dopa (100 μM), S-adenosyl-L-methionine (SAM, 50 μM, including ³H-labeled SAM), and serial concentrations of Entacapone (0.01–10 μM) at 37°C for 30 min. The reaction was stopped with 0.1 M HCl, and methylated products (3-methoxy-L-dopa) were extracted with ethyl acetate. Radioactivity of the extract was measured by liquid scintillation counting, and IC₅₀ was calculated from dose-response curves [1]
2. FTO m⁶A demethylase assay (HPLC-based): Recombinant human FTO (5 nM) was incubated in 20 mM HEPES buffer (pH 7.5) with m⁶A-modified RNA substrate (500 ng, 5'-FAM-labeled), MgCl₂ (2 mM), and serial concentrations of Entacapone (0.1–20 μM) at 37°C for 1 h. RNase A (1 μg) was added to digest RNA, and the reaction mixture was analyzed by HPLC (detection wavelength: 260 nm) to quantify m⁶A (demethylation product). Inhibition rate was calculated relative to vehicle, and IC₅₀ was derived [2] 3. FTO selectivity assay: The above FTO assay protocol was adapted to test Entacapone (0.1–20 μM) against ALKBH5 (5 nM) and ALKBH3 (5 nM) using their respective m⁶A-RNA or m⁶A-DNA substrates. Enzyme activity was measured by HPLC, and no significant inhibition (<10% at 20 μM Entacapone) was observed for ALKBH5/ALKBH3 [2] |
| Cell Assay |
1. 3T3-L1 adipocyte differentiation and gene expression: 3T3-L1 preadipocytes were seeded in 6-well plates and cultured in DMEM (10% FBS) until confluence. Differentiation was induced with medium containing 0.5 mM IBMX, 1 μM dexamethasone, and 10 μg/mL insulin for 6 days. Cells were then treated with serial concentrations of Entacapone (1–20 μM) for 48 h. Total RNA was extracted with TRIzol, and qRT-PCR was performed to detect AdipoQ, PPARγ, and GAPDH (housekeeping) mRNA. Nuclear proteins were extracted for Western blot analysis of FOXO1 [2]
2. HepG2 hepatocyte glucose production assay: HepG2 cells were seeded in 24-well plates and cultured in DMEM (10% FBS) until confluent. Medium was replaced with glucose-free DMEM containing Entacapone (1–10 μM) or insulin (100 nM, positive control) and incubated for 16 h. Gluconeogenesis was stimulated by adding 20 mM lactate and 2 mM pyruvate, and glucose concentration in the medium was measured after 4 h using a glucose oxidase kit [2] 3. FOXO1 ChIP-qPCR in adipocytes: 3T3-L1 adipocytes treated with 10 μM Entacapone for 48 h were cross-linked with 1% formaldehyde. Chromatin was sheared by sonication and incubated with anti-FOXO1 antibody or IgG (control) overnight at 4°C. Immune complexes were pulled down with protein A/G beads, and DNA was purified. qPCR was performed using primers targeting the AdipoQ promoter to quantify FOXO1 binding [2] |
| Animal Protocol |
Animal/Disease Models: High-fat diet-induced obese (DIO) mouse model[2]
Doses: 600 mg/kg Route of Administration: Oral administration; 600 mg/kg per day; 3-9 weeks Experimental Results: Regulated the metabolic disorders in DIO mouse. 1. Rat COMT inhibition and levodopa interaction study: Male Wistar rats (n=6/group) were randomized into vehicle (0.5% CMC-Na) and Entacapone groups. Entacapone (10 mg/kg) was administered orally, and vehicle was given at the same volume. At 0.5, 1, 2, 4, 6, and 8 h post-dosing, blood was collected to measure plasma COMT activity. For levodopa interaction, rats received levodopa (20 mg/kg, i.p.) + Entacapone (10 mg/kg) or levodopa + vehicle, and plasma levodopa/metabolite levels were measured by HPLC [1] 2. Mouse tissue COMT activity assay: Male ICR mice (n=6/group) received oral Entacapone (10 mg/kg) or vehicle. At 1 h post-dosing, mice were euthanized, and liver, kidney, and brain were collected. Tissue homogenates were prepared, and COMT activity was measured using the radiometric assay described above [1] 3. HFD-induced obese mouse metabolic study: Male C57BL/6 mice (6 weeks old) were fed HFD (60% fat) for 8 weeks to induce obesity. Mice (n=8/group) were then randomized to vehicle (DMSO:PEG400:saline = 10:40:50) or Entacapone (30 mg/kg, oral gavage, qd for 8 weeks). Body weight and food intake were recorded weekly. Fasting blood glucose/insulin were measured, and glucose tolerance tests were performed. At sacrifice, liver and adipose tissue were collected for triglyceride measurement and Western blot [2] 4. STZ-induced diabetic mouse study: Male C57BL/6 mice were injected with STZ (50 mg/kg, i.p.) for 5 days to induce diabetes. Mice (n=8/group) were treated with Entacapone (30 mg/kg, oral gavage, qd for 4 weeks) or vehicle. Fasting blood glucose/insulin and glucose tolerance were assessed, and pancreatic insulin content was measured by ELISA [2] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Entacapone is rapidly absorbed (approximately 1 hour). The absolute bioavailability after oral administration is 35%. Entacapone is almost completely metabolized before excretion, with only a very small amount (0.2% of the dose) found unchanged in the urine. Since only about 10% of the entacapone dose is excreted in the urine as unchanged drug and conjugated glucuronide, biliary excretion appears to be the primary route of excretion. 20 L 850 mL/min In rats and humans, the absolute bioavailability of entacapone is dose-dependent. In rats, single doses of 10, 65, and 400 mg/kg resulted in bioavailability ranging from 20% to 55%; in humans, single doses of 5, 25, 50, 100, 200, 400, and 800 mg resulted in bioavailability ranging from 29% to 49%. In both rats and dogs, absorption of unmetabolized entacapone was quite rapid following a single oral dose. In rats, two peak plasma concentrations were observed, occurring 5–15 minutes and 3–5 hours post-administration, indicating enterohepatic circulation of entacapone; in dogs, a single peak plasma concentration occurred 3 hours post-administration. In both animal groups studied, entacapone was converted to its (Z)-isomer, with very low conversion in rats and significant conversion in dogs. /Milk/ Animal studies have shown that entacapone is secreted into the milk of mother rats. In both rats and dogs, entacapone metabolites are primarily excreted in feces (two-thirds as glucuronide or sulfate conjugates), one-third in urine, and less than 1.5% of the dose is excreted unchanged. Within the first hour post-administration, 30–45% of the dose is recovered via bile, and approximately 10% of the radioactive material is excreted via enterohepatic circulation. For more complete data on the absorption, distribution, and excretion of entacapone (8 types), please visit the HSDB record page. Metabolism/Metabolites: Entacapone is first metabolized to the cis isomer via isomerization, followed by direct glucuronidation of the parent and cis isomers. In rats and dogs, entacapone metabolites are primarily excreted in feces (two-thirds as glucuronide or sulfate conjugates), one-third in urine, and less than 1.5% of the dose is excreted unchanged. Within one hour of administration, 30-45% of the dose is excreted in bile, and approximately 10% of the radioactive material is excreted via enterohepatic circulation. Entacapone is extensively metabolized in the liver of all species, including humans, primarily via glucuronidation, sulfation, and isomerization from the (E)-isomer to the (Z)-isomer (the active metabolite). Similar metabolic pathways across species include reduction of the C=C double bond in the side chain (less important in rats and humans) and hydrolysis to 3,4-dihydroxy-5-nitrobenzaldehyde. Differences include N-dealkylation of the amide, nitro reduction, and O-methylation (only in rats), amide hydrolysis and nitrile hydrolysis (only in dogs), and oxidative hydrolysis of an ethyl group in the diacetamide group (only in humans). Entacapone is almost completely metabolized before excretion, with only a very small amount (0.2% of the dose) present unchanged in the urine. The primary metabolic pathway is isomerization to the cis isomer, followed by direct glucuronidation of the parent and cis isomers; the glucuronide conjugate is inactive. Entacapone is extensively metabolized primarily in the liver. In humans, the primary metabolic pathway of entacapone is isomerization to the cis isomer, followed by direct glucuronidation of the parent and cis isomers; the glucuronide conjugate is inactive. The known metabolites of entacapone include entacapone-3-O-glucuronide. Metabolic pathway: First, it isomerizes to the cis isomer, then the parent compound and the cis isomer undergo direct glucuronidation. Excretion pathway: Entacapone is almost completely metabolized before excretion, with only a very small amount (0.2% of the dose) remaining unchanged in the urine. Since only about 10% of the entacapone dose is excreted in the urine as unchanged drug and conjugated glucuronide, biliary excretion appears to be the main route of excretion for this drug. Half-life: 0.4–0.7 hours Biobiological half-life0.4–0.7 hours Elimination of entacapone is biphasic, with a β-phase elimination half-life of 0.4 to 0.7 hours and a γ-phase elimination half-life of 2.4 hours. The overall elimination half-life of entacapone in dogs is 30 minutes to 1 hour, and in humans it is 1.5 to 3 hours. 1. Oral bioavailability: In rats, the oral bioavailability of entacapone (10 mg/kg) is approximately 35% (calculated by comparing AUC₀₋∞ with the intravenous dose of 3 mg/kg). In mice, the oral bioavailability is approximately 32% (comparing oral 30 mg/kg with intravenous 10 mg/kg)[1][2] 2. Plasma pharmacokinetics: In rats, the Cₘₐₓ of oral entacapone (10 mg/kg) is approximately 2.5 μM, Tₘₐₓ is approximately 1 h, t₁/₂ is approximately 2.5 h, and AUC₀₋₂₄ₕ is approximately 7.8 μM·h. In mice, the oral entacapone (30 mg/kg) concentrations were Cₘₐₓ = ~3.2 μM, Tₘₐₓ = ~1.2 h, and t₁/₂ = ~3.0 h [1][2] 3. Tissue distribution: In rats, oral entacapone (10 mg/kg) was distributed in the liver (Cₘₐₓ = ~8 μM), kidney (Cₘₐₓ = ~6 μM), and intestine (Cₘₐₓ = ~5 μM), but the brain concentration was <0.1 μM (it could not cross the blood-brain barrier). In high-fat diet (HFD) mice, entacapone (orally administered 30 mg/kg) accumulated in adipose tissue (Cₘₐₓ = ~2.8 μM) and liver (Cₘₐₓ = ~4.5 μM) [1][2] 4. Metabolism and excretion: In rats, approximately 70% of orally administered entacapone (10 mg/kg) was excreted via bile within 24 hours (30% as the original drug and 40% as glucuronide metabolites). Approximately 20% was excreted via urine (mainly as metabolites). The main metabolic pathway was hepatic glucuronidation (inactive metabolites) [1] |
| Toxicity/Toxicokinetics |
Toxicity Summary
Identification and Use: Entacapone is a selective, reversible catechol-O-methyltransferase (COMT) inhibitor used in combination with levodopa and carbidopa to treat end-of-dose failure in patients with Parkinson's disease. Human Exposure and Toxicity: Post-marketing data show several cases of overdose. The highest reported dose of entacapone is at least 40,000 mg. Common acute symptoms and signs in these cases include somnolence and decreased activity, decreased level of consciousness (coma, confusion, and disorientation), and discoloration of the skin, tongue, and urine, as well as agitation, anxiety, and aggressive behavior. Entacapone treatment results in dose-dependent inhibition of catechol-O-methyltransferase (COMT). Theoretically, an overdose of entacapone could lead to 100% inhibition of catecholamine-O-methyltransferase (COMT) in the body, thereby preventing the metabolism of endogenous and exogenous catechols. Post-marketing reports also indicate that patients may experience new or worsening mental and behavioral changes, including psychotic-like behaviors, during entacapone treatment or after initiating or increasing the dose. Therefore, entacapone should generally not be used to treat patients with severe psychosis due to the risk of exacerbating their psychosis. Under metabolically activated conditions, entacapone has a chromosome-breaking effect on cultured human lymphocytes. Animal studies: Rats were administered entacapone orally at daily doses of 20, 90, or 400 mg/kg for two years. Results showed an increased incidence of renal tubular adenomas and carcinomas in male rats treated with the highest dose. Reproductive studies of entacapone at doses up to 1000 mg/kg/day and 300 mg/kg/day were conducted in rats and rabbits, respectively. In the absence of significant maternal toxicity, an increased incidence of fetal malformations was observed in offspring of rats treated with the highest dose of entacapone. In rabbit pups treated with entacapone at doses of 100 mg/kg/day or higher (maternally toxic dose), increased abortion rates, increased late/complete absorption rates, and decreased fetal weight were observed. No evidence of teratogenicity was found in these studies. In female rats treated with entacapone at doses of 160 mg/kg/day or higher before mating and in early pregnancy, without maternal toxicity, an increased incidence of fetal ocular malformations (macrophthalmia, microphthalmia, and anophthalmia) was observed. In late pregnancy and throughout lactation, administration of up to 700 mg/kg entacapone daily to female rats did not indicate impaired offspring development. Daily administration of up to 700 mg/kg entacapone did not impair fertility or general reproductive function in rats. Female rats administered 700 mg/kg entacapone daily experienced delayed mating but no impaired fertility was observed. In in vitro mouse lymphoma TK assays, entacapone exhibited mutagenicity and chromosome breakage induced regardless of metabolic activation. In in vivo mouse micronucleus assays, entacapone, alone or in combination with levodopa and carbidopa, did not show chromosome breakage inducedness; entacapone also did not show mutagenicity in the bacterial reverse mutation assay (Ames assay). The mechanism of action of entacapone is believed to be altering the plasma pharmacokinetics of levodopa by inhibiting COMT in peripheral tissues. When entacapone is used in combination with levodopa and aromatic amino acid decarboxylase inhibitors (e.g., carbidopa), levodopa plasma concentrations are higher and more sustained than when levodopa and aromatic amino acid decarboxylase inhibitors are used alone. It is believed that, at a given levodopa dosing frequency, this more sustained levodopa plasma concentration leads to more sustained dopaminergic stimulation in the brain, thereby more effectively alleviating the symptoms of Parkinson's syndrome. Hepatotoxicity Entacapone treatment was associated with elevated serum transaminases (more than 3 times the upper limit of normal) in only 0.3% to 0.5% of patients, a proportion similar to or slightly higher than the placebo group. These elevations were usually transient and asymptomatic, rarely requiring dose adjustment. No clinically significant serious liver injury with jaundice was reported in the initial clinical trials. Subsequently, the sponsor received sporadic reports of hepatotoxicity cases, with liver injury occurring 2 to 6 weeks after initiation of entacapone, manifested as mild jaundice and cholestatic liver enzyme elevations, which resolved rapidly upon discontinuation of the drug. No immune hypersensitivity or autoimmune features were observed. The clinical phenotype and associated features of liver injury have not been reported in detail. Therefore, entacapone may very rarely cause clinically significant liver injury, but is not associated with the severe hepatitis and acute liver failure commonly seen in cases of liver injury caused by tocapone. Probability Score: D (Probably, but rarely, a cause of clinically significant liver injury). Effects during pregnancy and lactation ◉ Overview of medication use during lactation There is currently no information on the use of entacapone during lactation. Especially during the nursing period for newborns or premature infants, alternative medications may be necessary. ◉ Effects on breastfed infants No relevant published information was found as of the revision date. ◉ Effects on lactation and breast milk No relevant published information was found as of the revision date. Protein binding 98% (bound to serum albumin) Interactions Because entacapone is primarily excreted via bile, caution should be exercised when taken concurrently with medications known to interfere with bile excretion, glucuronidation, and intestinal β-glucuronidase. These medications include probenecid, cholestyramine, and some antibiotics (e.g., erythromycin, rifampin, ampicillin, and chloramphenicol). Entacapone has a high protein binding rate (98%). In vitro studies have shown that entacapone does not exhibit binding displacement interactions with other highly binding drugs such as warfarin, salicylic acid, phenylbutazone, and diazepam. It may interact pharmacokinetically with drugs that interfere with bile excretion, glucuronidation, and intestinal β-glucuronidase (e.g., cholestyramine, probenecid, and certain anti-infective drugs such as ampicillin, chloramphenicol, erythromycin, and rifampin) (reducing entacapone excretion). It may interact pharmacologically with non-selective monoamine oxidase (MAO) inhibitors (e.g., phenelzine, transphenylcyclopropionate) (inhibiting catecholamine metabolism). The likelihood of pharmacological interactions with selective MAO-B inhibitors (e.g., selegiline) is very low. For more complete data on the interactions of entacapone (13 drugs in total), please visit the HSDB record page. 1. Acute toxicity: In mice, the oral LD₅₀ of entacapone was > 2000 mg/kg; in rats, the oral LD₅₀ was > 1500 mg/kg. No death or significant toxicity (e.g., somnolence, weight loss) was observed at an oral dose of 1000 mg/kg [1] 2. Subacute toxicity: In rats, no changes were observed in body weight, food intake, or serum biochemical parameters (ALT, AST, creatinine, urea nitrogen) after oral administration of entacapone (100 mg/kg, once daily for 28 days). Liver and kidney histopathological examination results were normal [1] 3. Metabolic model toxicity: In high-fat diet (HFD) mice, serum ALT/AST levels and liver and kidney histopathological examination results were normal after treatment with entacapone (30 mg/kg, once daily for 8 weeks). No inflammatory infiltration was observed in adipose tissue [2] 4. Plasma protein binding rate: In rat plasma, the protein binding rate of entacapone was approximately 98% (measured by ultrafiltration method, molecular weight cutoff of 30 kDa). In mouse plasma, the binding rate was approximately 97% [1][2] 5. Drug interactions: Entacapone (1–10 μM) does not inhibit CYP450 enzymes (CYP1A2, CYP2D6, CYP3A4) in human liver microsomes. No adverse pharmacokinetic interactions were observed when used in combination with levodopa, only the half-life of levodopa was prolonged [1] |
| References |
|
| Additional Infomation |
Therapeutic Uses
Anti-Parkinson's Disease Drugs; Enzyme Inhibitors /Clinical Trials/ ClinicalTrials.gov is a registry and results database that lists human clinical studies funded by public and private institutions worldwide. The website is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each record on ClinicalTrials.gov includes a summary of the study protocol, including: the disease or condition; the intervention (e.g., the medical product, behavior, or procedure being studied); the title, description, and design of the study; participation requirements (eligibility criteria); the location of the study; contact information for the study location; and links to relevant information from other health websites, such as the NLM's MedlinePlus (for providing patient health information) and PubMed (for providing citations and abstracts of academic articles in the medical field). Entacapone is listed in the database. Comtan is indicated as adjunctive therapy to levodopa and carbidopa for the treatment of "end-of-dose failure" symptoms in patients with Parkinson's disease. /Included in US Product Label/ Stalevo is a combination medication consisting of levodopa, carbidopa (a dopa decarboxylase inhibitor), and entacapone (a catechol-O-methyltransferase-COMT inhibitor) for the treatment of Parkinson's disease. Statalevo can be used to: replace (at equal concentrations of all three components) previously used carbidopa/levodopa and entacapone alone; or replace carbidopa/levodopa (without entacapone) when the patient experiences signs and symptoms of "end-of-dose failure" and is taking a total daily dose of levodopa of 600 mg or less, and has no motor impairment. /Included in US Product Label/ Parkinson's disease (PD) is a neurodegenerative disease characterized by a variety of motor symptoms, including gait freezing (FOG), which is a brief cessation of walking, as if the patient's feet are "stuck to the ground." Treatment of FOG remains challenging. Although the norepinephrine precursor L-threo-3,4-dihydroxybenzylserine (L-DOPS) has been marketed in Japan due to its efficacy against frozen gait (FOG), its clinical application has not yielded satisfactory results. However, the fact that some patients respond to L-DOPS prompted us to hypothesize that simultaneously using L-DOPS and a catechol-O-methyltransferase (COMT) inhibitor to increase intracranial L-DOPS concentration, thereby blocking L-DOPS metabolism in the peripheral circulation, might be beneficial for FOG. Based on our hypothesis, we conducted a preliminary study in a small number of subjects with frozen gait (FOG). Of the 16 Parkinson's disease (PD) patients with frozen gait (FOG) who completed this study, group 1 (n=6) received levodopam (L-DOPS) in combination with entacapone (a globally used anti-Parkinson's drug, belonging to the catecholamine-O-methyltransferase inhibitor class); group 2 (n=5) received entacapone alone; and group 3 (n=5) received L-DOPS alone. Results showed that only patients in group 1 experienced significant improvement in FOG symptoms. Furthermore, this beneficial effect was observed only in levodopa-resistant FOG patients. At least in patients with levodopa-resistant frozen gait (FOG), these results support our hypothesis and suggest that the combination of levodopam (L-DOPS) and entacapone may be a novel strategy for treating FOG. Drug Warning In clinical studies, 10% of patients receiving entacapone reported diarrhea, and approximately 2% required discontinuation of the drug due to diarrhea. Diarrhea is usually mild to moderate, but in rare cases, severe diarrhea requiring hospitalization may occur. Diarrhea typically occurs during the first 4–12 weeks of entacapone treatment, but can also appear as early as the first week of treatment or as late as several months after treatment begins. Diarrhea symptoms usually subside upon discontinuation of the medication. A meta-analysis conducted by the U.S. Food and Drug Administration (FDA) suggests that patients receiving combination therapy with levodopa, carbidopa, and entacapone may have a higher risk of cardiovascular events (e.g., myocardial infarction, stroke, cardiovascular death) compared to patients receiving levodopa-carbidopa monotherapy. This meta-analysis summarized the results of 15 cardiovascular-related clinical trials comparing the efficacy of combination therapy with levodopa, carbidopa, and entacapone to levodopa-carbidopa monotherapy. The results showed a slightly increased risk of cardiovascular events in patients receiving combination therapy with levodopa, carbidopa, and entacapone, but this was statistically significant (relative risk: 2.46). However, the increased risk primarily stemmed from one trial (STRIDE-PD); after removing data from that trial from the analysis, the results were no longer statistically significant. Several factors make it difficult to draw definitive conclusions from this meta-analysis. Many of the trials included in the analysis lasted less than 6 months (potentially insufficient to detect cardiovascular risk) and were not specifically designed to assess cardiovascular safety. Furthermore, most patients already had pre-existing cardiovascular risk factors. Currently, the FDA has not concluded that combination therapy with levodopa, carbidopa, and entacapone increases the risk of cardiovascular events and is continuing to review existing data related to this safety concern. Patients currently receiving entacapone as adjunctive therapy to levodopa-carbidopa (whether used alone or as a fixed-dose combination) should continue to take the medication as prescribed unless otherwise directed by their clinician. Cardiac function should be monitored regularly in these patients, especially those with a history of cardiovascular disease. Dopaminergic treatment in patients with Parkinson's disease is associated with orthostatic hypotension. Entacapone can increase the bioavailability of levodopa; therefore, an increased incidence of orthostatic hypotension is expected. In controlled studies, approximately 1.2% and 0.8% of patients in the entacapone 200 mg group and placebo group, respectively, reported at least one episode of syncope. Syncope reports were generally more common among patients with documented episodes of hypotension in both treatment groups. Post-marketing reports indicate that new or worsening changes in mental status and behavior, including psychotic-like behaviors, may occur during or after taking Comtan, or after starting or increasing the dose of Comtan. These changes can be severe. Other medications used to improve Parkinson's disease symptoms may also have similar effects on thought and behavior. Abnormal thought and behavior can lead to delusions, illusions, hallucinations, confusion, disorientation, aggression, agitation, and delirium. Psychotic-like behaviors have also been observed during the clinical development of Comtan. Patients with severe psychosis should generally not be treated with Comtan due to the risk of exacerbating their psychosis. Furthermore, some medications used to treat psychosis may worsen Parkinson's disease symptoms and may reduce the efficacy of Entacapone. For more complete (22) drug warnings regarding entacapone, please visit the HSDB records page. Pharmacodynamics Entacapone is structurally and pharmacologically related to tolcapone, but unlike tolcapone, entacapone is not hepatotoxic. Entacapone is used to treat Parkinson's disease as an adjunct to levodopa/carbidopa therapy. Entacapone selectively and reversibly inhibits catechol-O-methyltransferase (COMT). In mammals, COMT is distributed across various organs, with the highest activity in the liver and kidneys. COMT is also present in the heart, lungs, smooth and skeletal muscle, intestines, reproductive organs, various glands, adipose tissue, skin, blood cells, and nerve tissue, particularly in glial cells. COMT catalyzes the methyl transfer of S-adenosyl-L-methionine to the phenolic hydroxyl group of a substrate containing a catechol structure. The physiological substrates of COMT include dopa, catecholamines (dopamine, norepinephrine and epinephrine) and their hydroxylated metabolites. The function of COMT is to clear bioactive catecholamines and some other hydroxylated metabolites. In the presence of decarboxylase inhibitors, COMT becomes the major metabolic enzyme of levodopa, catalyzing its conversion to 3-methoxy-4-hydroxy-L-phenylalanine (3-OMD) in the brain and periphery. 1. COMT inhibition mechanism: Entacapone binds to the active site of peripheral COMT, competing with catechol substrates (such as levodopa) for SAM-dependent methylation. This reduces the degradation of levodopa in the periphery and increases its delivery to the brain for dopamine synthesis (Parkinson's disease treatment)[1] 2. FTO inhibition mechanism: Entacapone binds to the catalytic domain of FTO, blocking its m⁶A demethylase activity. Increased m⁶A levels on FOXO1 mRNA enhance nuclear localization of FOXO1, activate lipogenesis/glycemic regulation genes (e.g., AdipoQ), thereby improving metabolism [2] 3. Application in the treatment of Parkinson's disease: Entacapone is a peripherally selective COMT inhibitor that can be used as an adjunct to levodopa/carbidopa in the treatment of Parkinson's disease. It reduces motor fluctuations caused by "hypoplasia" by prolonging the efficacy of levodopa (since it does not cross the blood-brain barrier, there are no central side effects) [1] 4. Potential for the treatment of metabolic diseases: FTO overexpression is associated with obesity and type 2 diabetes. The FTO inhibitory activity (weight loss, glycemic control) shown by entacapone in preclinical models suggests its potential for treating metabolic diseases, but clinical studies are still ongoing [2] |
| Molecular Formula |
C14H15N3O5
|
|---|---|
| Molecular Weight |
305.29
|
| Exact Mass |
305.101
|
| CAS # |
130929-57-6
|
| Related CAS # |
Entacapone-d10;1185241-19-3;(Z)-Entacapone-d10;Entacapone sodium salt;1047659-02-8;(E)-Entacapone-d10
|
| PubChem CID |
5281081
|
| Appearance |
Light yellow to yellow solid powder
|
| Density |
1.4±0.1 g/cm3
|
| Boiling Point |
526.6±50.0 °C at 760 mmHg
|
| Melting Point |
162-1630C
|
| Flash Point |
272.3±30.1 °C
|
| Vapour Pressure |
0.0±1.4 mmHg at 25°C
|
| Index of Refraction |
1.642
|
| LogP |
2.38
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
6
|
| Rotatable Bond Count |
4
|
| Heavy Atom Count |
22
|
| Complexity |
500
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
CCN(CC)C(=O)/C(=C/C1=CC(=C(C(=C1)O)O)[N+](=O)[O-])/C#N
|
| InChi Key |
JRURYQJSLYLRLN-BJMVGYQFSA-N
|
| InChi Code |
InChI=1S/C14H15N3O5/c1-3-16(4-2)14(20)10(8-15)5-9-6-11(17(21)22)13(19)12(18)7-9/h5-7,18-19H,3-4H2,1-2H3/b10-5+
|
| Chemical Name |
(E)-2-cyano-3-(3,4-dihydroxy-5-nitrophenyl)-N,N-diethylacrylamide
|
| Synonyms |
OR 611; OR611; OR-611; Comtan; Entacapone; HSDB-8251; HSDB8251; HSDB 8251;
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO: 61 mg/mL (199.8 mM)
Water:<1 mg/mL
Ethanol: 2 mg/mL (6.5 mM)
|
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.19 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (8.19 mM) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (6.81 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 1 mg/mL (3.28 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2756 mL | 16.3779 mL | 32.7557 mL | |
| 5 mM | 0.6551 mL | 3.2756 mL | 6.5511 mL | |
| 10 mM | 0.3276 mL | 1.6378 mL | 3.2756 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT06236230 | Recruiting | Drug: levodopa/carbidopa/entacapone | Parkinson Disease | Second Affiliated Hospital of Soochow University |
November 15, 2023 | Phase 4 |
| NCT04006769 | Completed | Drug: Entacapone Drug: Imatinib Mesylate |
Gastrointestinal Stromal Tumor, Malignant |
Xiangya Hospital of Central South University |
October 30, 2020 | Early Phase 1 |
| NCT00373087 | Completed | Drug: entacapone Drug: l dopa versus placebo |
Parkinson's Disease | Assistance Publique - Hôpitaux de Paris |
October 2006 | Phase 4 |
| NCT00192855 | Completed | Drug: Entacapone | Schizophrenia | Rambam Health Care Campus | June 2003 | Not Applicable |
 |
|---|