
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
AZD4547 (AZD-4547; AZD 4547) is a novel and orally bioavailable FGFR (fibroblast growth factor receptor) inhibitor with potential antineoplastic activity. In cell-free assays, it inhibits FGFR1/2/3 with IC50s of 0.2 nM/2.5 nM/1.8 nM. However, its activity against other kinases, including FGFR4, VEGFR2(KDR), IGFR, CDK2, and p38, is minimal or weaker. Significant tumor-suppressive effects of AZD4547 are observed in mice with KMS11 tumors with a TGI of 53%.
| Targets |
FGFR1 (IC50 = 0.2 nM); FGFR2 (IC50 = 2.5 nM); FGFR3 (IC50 = 1.8 nM); FGFR4 (IC50 = 165 nM)
ibroblast Growth Factor Receptor (FGFR) 1 (IC50 = 1.2 nM), FGFR2 (IC50 = 2.5 nM), FGFR3 (IC50 = 1.8 nM), FGFR4 (IC50 = 8.6 nM); weak activity against VEGFR2 (IC50 = 120 nM), PDGFRβ (IC50 = 150 nM); no significant activity against EGFR, ALK, MET (IC50 > 1000 nM) [1] - ATP-competitive binding to FGFR kinase domain; no covalent interaction with target residues [1] |
|---|---|
| ln Vitro |
Fexagratinib also inhibits recombinant VEGFR2 (KDR) kinase activity with an IC50 of 24 nM. Fexagratinib exhibits a powerful inhibition of autophosphorylation of FGFR1, 2, and 3 tyrosine kinases (IC50 values of 12, 2, and 40 nM, respectively) in KG1a, Sum52-PE, MCF7, and KMS11 cell lines. However, it lessens the inhibition of FGFR4 cellular kinase activity (IC50=142 nM). Compared to cellular KDR and IGFR ligand-induced phosphorylation, significantly lower inhibitory activity is seen (IC50 values of 258 and 828 nM, respectively), indicating selectivity over cellular FGFR1 of about 20 and 70 times, respectively. Furthermore, FGFR phosphorylation and downstream signaling mediated by FRS2, PLCγ, and MAPK are potently inhibited by fexagratinib at the cellular level[1].
Inhibited recombinant FGFR kinase activity: FGFR1 (IC50 = 1.2 nM), FGFR2 (IC50 = 2.5 nM), FGFR3 (IC50 = 1.8 nM), FGFR4 (IC50 = 8.6 nM); suppressed FGFR autophosphorylation in Ba/F3-FGFR1 cells (IC50 = 4.3 nM), Ba/F3-FGFR2 cells (IC50 = 5.1 nM) [1] - Reduced viability of FGFR-amplified cancer cell lines: Lung cancer NCI-H1581 (FGFR1 amplified, IC50 = 9.7 nM), gastric cancer SNU-16 (FGFR2 amplified, IC50 = 12.3 nM), bladder cancer RT112 (FGFR3 mutated, IC50 = 10.5 nM); no activity in FGFR-negative A549 cells (IC50 > 500 nM) [1] - Suppressed FGFR downstream signaling: 100 nM Fexagratinib (AZD-4547; ADSK-091) reduced p-FGFR (Tyr653/654) by 90% in NCI-H1581 cells (2 hours), and downregulated p-ERK1/2 (Thr202/Tyr204) and p-AKT (Ser473) by >85% [1] - Induced apoptosis in SNU-16 cells: 200 nM Fexagratinib increased Annexin V-positive cells from 5% (vehicle) to 42% (48 hours); caspase-3/7 activity elevated by 3.4-fold [1] - Reduced colony formation of RT112 cells: 50 nM Fexagratinib decreased colony number by 78% compared to vehicle (14-day incubation) [1] |
| ln Vivo |
Fexagratinib is administered chronically at various well-tolerated doses to female SCID mice with KMS11 tumors. The mice are treated in a randomized manner. Tumor growth inhibition is dose-dependent when administered orally with Fexagratinib. When compared to vehicle-treated controls, the administration of Fexagratinib at a dose of 3 mg/kg twice a day results in a statistically significant tumor growth inhibition of 53% (P0.0005 by one-tailed t test), whereas doses of 12.5 mg/kg once daily and 6.25 mg/kg twice a day result in complete tumor stasis (P0.0001). Fexagratinib at a dose of 12.5 mg/kg once daily in the KG1a model was found to be 65% effective in inhibiting tumor growth (P=0.002)[1].
In nude mice bearing NCI-H1581 (FGFR1 amplified) xenografts: Oral Fexagratinib (25 mg/kg/day) for 28 days resulted in 88% tumor growth inhibition (TGI); tumor p-FGFR levels reduced by 82% (immunohistochemistry) [1] - In nude mice bearing SNU-16 (FGFR2 amplified) xenografts: Oral Fexagratinib (30 mg/kg/day) for 21 days achieved 83% TGI; median tumor doubling time extended from 6 days (vehicle) to 23 days [1] - In nude mice bearing RT112 (FGFR3 mutated) xenografts: Oral Fexagratinib (20 mg/kg/day) for 35 days improved survival (median survival: 52 days vs. 30 days for vehicle); no tumor regression but delayed progression [1] |
| Enzyme Assay |
AZD4547 is tested with ATP concentrations at or slightly below the corresponding Km to see if it can inhibit the human recombinant kinase activities of FGFR1-3).
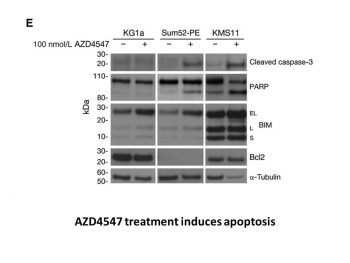
In vitro protein expression analysis and kinase inhibition studies[1] Cells were treated with AZD4547 or control for 3 hours at 37°C and then stimulated with 10 ng/mL aFGF/bFGF and 10 μg/mL heparin for 20 minutes. Western blotting was conducted with standard SDS-PAGE procedures and antibody incubation carried out overnight at 4°C. Antibodies were obtained from the following sources: FGFR1, FGFR2 and FRS2, FGFR3 proteins, α-tubulin-B512 and Bcl2, and BIM. Secondary antibodies were applied and immunoreactive proteins visualized with “SuperSignal West Dura” Chemiluminescence substrate according to the manufacturer's instructions. Inhibition of cellular receptor phosphorylation[1] For FGFR phosphorylation studies, FGFR1, 3, or 4–transfected Cos-1 cells were cultured in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 2 mmol/L l-glutamine and 3% FCS. For FGFR2, Sum52-PE cells were cultured in RPMI-1640, growth media supplemented with 2 mmol/L l-glutamine and 10% FBS. Following 1-hour incubation with AZD4547, media were removed; cells were fixed, permeabilized, and then incubated with monoclonal anti-phospho-FGFR antibody (Cell Signaling Technology; 1:1,000) for 1 hour followed by incubation with anti-mouse Alexa Fluor 594 secondary antibody (1:500) and Hoechst (1:1,000) for 1 hour. Fluorescence measurement was conducted with Arrayscan. For KDR phosphorylation studies, primary human umbilical vein endothelial cells were obtained from PromoCell and cultured according to the supplier's protocol. Cells were incubated with AZD4547 for 90 minutes and then stimulated for 5 minutes with VEGF ligand (25 ng per well). Cells were lysed with standard radioimmunoprecipitation assay buffer containing phosphatase/protease inhibitors. Lysates were analyzed with the human phospho-VEGF R2 ELISA protocol according to the manufacturer's instructions. For insulin-like growth factor-1 receptor (IGF1R) phosphorylation studies, R+ cells were derived from murine transgenic IGF1R knockouts and then stably transfected with human IGF1R. Cells cultured in DMEM supplemented with 1% heat-inactivated FCS and 1% l-glutamine were incubated with AZD4547 and then stimulated with IGF ligand, followed by fixation, blocking, and incubation with a rabbit anti-phospho IGF1R/IR antibody (1:350) for 1 hour. Secondary detection and measurement was carried out with an Acumen Explorer HTS Reader at an excitation wavelength of 488 nm and emission wavelength of 530 nm. FGFR kinase activity assay: Recombinant human FGFR1/2/3/4 kinases (50 ng/well) were incubated with Fexagratinib (0.01-100 nM) in reaction buffer (25 mM HEPES pH 7.5, 10 mM MgCl2, 1 mM DTT) at 30°C for 15 minutes. 10 μM ATP and a fluorescent peptide substrate were added, followed by 60-minute incubation at 30°C. Kinase activity was measured via homogeneous time-resolved fluorescence (HTRF; excitation 340 nm, emission 665 nm); IC50 values were calculated via nonlinear regression [1] - FGFR binding assay (SPR): FGFR1 kinase domain (2 μg/mL) was immobilized on a sensor chip. Fexagratinib (0.1-100 nM) was injected at 30 μL/min; binding affinity (KD = 0.8 nM) was determined by fitting sensorgrams to a 1:1 binding model [1] |
| Cell Assay |
For 72 hours, cell lines are incubated with AZD4547 at fixed concentrations. Cells are fixed with 70% ethanol and then incubated with a propidium iodide/RNase A labeling solution for fluorescence-activated cell sorting (FACS). CellQuest analysis software and an FACSCalibur instrument are used to evaluate cell-cycle profiles. Cells and media are carefully collected, centrifuged, and cell pellets are then washed in preparation for apoptotic analysis. Following that, the cells are prepared for propidium iodide uptake and FITC staining. A FACSCalibur device is then used to determine the percentage of cells that stain positively for Annexin V, and CellQuest analysis software is used to sort the cells into quadrants[1].
Cell proliferation assay (NCI-H1581/SNU-16/RT112): Cells were seeded in 96-well plates (5×10³ cells/well) and treated with Fexagratinib (0.1 nM-1 μM) for 72 hours. Cell viability was measured via tetrazolium-based colorimetric assay; absorbance at 570 nm was recorded, and IC50 values were determined via four-parameter logistic fitting [1] - Western blot assay (FGFR/ERK/AKT): NCI-H1581 cells were treated with Fexagratinib (10-200 nM) for 2 hours, lysed in RIPA buffer (with protease/phosphatase inhibitors). Lysates (30 μg protein) were separated by 8% SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against p-FGFR (Tyr653/654), total FGFR, p-ERK1/2, total ERK1/2, p-AKT, total AKT, and GAPDH. Signals were detected via chemiluminescence [1] - Apoptosis assay (SNU-16): Cells were treated with Fexagratinib (50-200 nM) for 48 hours, stained with Annexin V-FITC and propidium iodide, and analyzed by flow cytometry. Caspase-3/7 activity was measured via fluorometric assay with a specific substrate [1] - Colony formation assay (RT112): Cells were seeded in 6-well plates (1×10³ cells/well) and treated with Fexagratinib (10-50 nM) or vehicle. After 14 days, colonies were fixed with methanol, stained with crystal violet, and counted manually [1] |
| Animal Protocol |
Mice: The mice used are Swiss-derived nude (nu/nu) and severe combined immunodeficient (SCID). Tumor xenografts are created by injecting 0.1 mL (1×10 6 for LoVo, 1×10 7 for HCT-15, and 1×10 7 for Calu-6) or 0.2 mL (2×10 7 for KMS11 and KG1a) of tumor cells mixed 1:1 with Matrigel into the left flank, with the exception of LoVo and HCT-15, which do not include Matrigel. Tumors that are larger than 0.2 cm 3 are randomly assigned to treatment and control groups, and mice are given AZD4547 (1.5-50 mg/kg) orally once or twice a day. For the duration of the study, tumor condition, animal body weight, and tumor volume (measured using a caliper) are recorded twice a week. One computes the tumor volume.
NCI-H1581 xenograft model (nude mice): 6-week-old female nude mice were subcutaneously injected with 5×10⁶ NCI-H1581 cells. When tumors reached 100-120 mm³, mice were randomized to vehicle (0.5% methylcellulose + 0.2% Tween 80) or Fexagratinib groups (25 mg/kg/day, oral gavage). Treatments were administered once daily for 28 days; tumor volume (length × width² / 2) and body weight were measured every 3 days [1] - SNU-16 xenograft model (nude mice): Female nude mice were implanted with 1×10⁷ SNU-16 cells subcutaneously. When tumors reached 150 mm³, mice received Fexagratinib (30 mg/kg/day, oral gavage) for 21 days. Drug was dissolved in 10% DMSO + 40% PEG400 + 50% normal saline; tumor samples were collected for immunohistochemistry [1] - RT112 xenograft model (nude mice): Male nude mice were injected with 2×10⁶ RT112 cells subcutaneously. When tumors reached 100 mm³, mice received Fexagratinib (20 mg/kg/day, oral gavage) for 35 days. Drug was dissolved in 0.5% methylcellulose; survival time was recorded [1] |
| ADME/Pharmacokinetics |
In mice: the oral bioavailability of Fexagratinib was 52% (25 mg/kg); the plasma half-life (t1/2) was 4.8 hours; the peak plasma concentration (Cmax) 1.2 hours after oral administration was 4.1 μM [1] In rats: the clearance rate after intravenous administration (10 mg/kg) was 14 mL/min/kg; the steady-state volume of distribution (Vss) was 0.9 L/kg [1] In dogs: the oral bioavailability was 48% (15 mg/kg); t1/2 was 6.5 hours; the peak plasma concentration (Cmax) 2 hours after administration was 2.9 μM [1] Plasma protein binding: the binding rate to human plasma proteins was 99.3% (determined by ultrafiltration) [1]
|
| Toxicity/Toxicokinetics |
In the 28-day NCI-H1581 xenograft study (25 mg/kg/day, orally): no significant weight loss (>8%) was observed; serum ALT (29 ± 5 U/L) and BUN (18 ± 3 mg/dL) were within the normal range (ALT: 20-40 U/L, BUN: 15-25 mg/dL) [1]
- In the 21-day SNU-16 xenograft study (30 mg/kg/day, orally): 1 out of 8 mice experienced mild diarrhea (which resolved within 4 days); no histopathological changes were observed in the liver, kidneys, or stomach [1] - In the 35-day RT112 xenograft study (20 mg/kg/day, orally): no treatment-related deaths were observed; 2 out of 10 mice experienced mild alopecia (which resolved after treatment) [1] |
| References | |
| Additional Infomation |
AZD4547 belongs to the benzamide class of compounds. It is a carboxamide formed by the condensation of the carboxyl group of 4-(cis-3,5-dimethylpiperazin-1-yl)benzoic acid with the amino group of 5-[2-(3,5-dimethoxyphenyl)ethyl]-1H-pyrazole-3-amine. It is an inhibitor of fibroblast growth factor receptor (FGFR) and has antagonistic effects. AZD4547 belongs to the pyrazole, N-arylpiperazine, and benzamide classes of compounds. AZD4547 has been used in clinical trials for the treatment of various diseases, including cancer, lymphoma, gastric cancer, adenocarcinoma, and solid tumors. Fexagratinib is an orally bioavailable fibroblast growth factor receptor (FGFR) inhibitor with potential antitumor activity. Fexagratinib binds to FGFR and inhibits its activity, thereby inhibiting FGFR-related signaling pathways and thus inhibiting tumor cell proliferation and death. FGFR, an upregulated receptor tyrosine kinase in various tumor cell types, is crucial for tumor cell proliferation, differentiation, and survival. The role of the fibroblast growth factor (FGF) signaling pathway in tumorigenesis and chemotherapeutic resistance is increasingly recognized. Currently, several small-molecule FGF receptor (FGFR) kinase inhibitors are in clinical development; however, the most advanced drugs primarily target kinase insertion domain receptors (KDRs), which reduces their selectivity for FGFR. This article reports the pharmacological properties of AZD4547, a novel selective FGFR1, 2, and 3 tyrosine kinase inhibitor. AZD4547 inhibits recombinant FGFR kinase activity in vitro and suppresses FGFR signaling and growth in dysregulated FGFR-expressing tumor cell lines. In a representative FGFR-driven human tumor xenograft model, oral administration of AZD4547 was well-tolerated and exhibited potent dose-dependent antitumor activity consistent with plasma exposure and pharmacodynamic modulation of tumor FGFR. Importantly, no anti-KDR-related effects were observed at effective doses, confirming the selectivity of AZD4547 for FGFR in vivo. In summary, our results indicate that AZD4547 is a novel selective small molecule FGFR inhibitor with potent antitumor activity against FGFR-dysregulated tumors in preclinical models. AZD4547 is currently undergoing clinical trials for the treatment of FGFR-dependent tumors. [1]
Fexagratinib (AZD-4547; ADSK-091) is a selective ATP-competitive FGFR tyrosine kinase family inhibitor designed to target solid tumors (lung cancer, gastric cancer, bladder cancer) with FGFR amplification or mutation.[1] -Its selectivity for FGFR is much higher than that for other kinases (e.g., EGFR, ALK), thus reducing the risk of off-target toxicity.[1] -Preclinical data support its potential for oral administration in clinical trials, with good bioavailability and plasma stability.[1] |
| Molecular Formula |
C26H33N5O3
|
|---|---|
| Molecular Weight |
463.57
|
| Exact Mass |
463.258
|
| Elemental Analysis |
C, 67.36; H, 7.18; N, 15.11; O, 10.35
|
| CAS # |
1035270-39-3
|
| Related CAS # |
1035270-39-3;1394854-62-6;
|
| PubChem CID |
51039095
|
| Appearance |
white solid powder
|
| Density |
1.2±0.1 g/cm3
|
| Boiling Point |
621.7±55.0 °C at 760 mmHg
|
| Flash Point |
329.8±31.5 °C
|
| Vapour Pressure |
0.0±1.8 mmHg at 25°C
|
| Index of Refraction |
1.602
|
| LogP |
3.46
|
| Hydrogen Bond Donor Count |
3
|
| Hydrogen Bond Acceptor Count |
6
|
| Rotatable Bond Count |
8
|
| Heavy Atom Count |
34
|
| Complexity |
622
|
| Defined Atom Stereocenter Count |
2
|
| SMILES |
O=C(C1C([H])=C([H])C(=C([H])C=1[H])N1C([H])([H])[C@]([H])(C([H])([H])[H])N([H])[C@]([H])(C([H])([H])[H])C1([H])[H])N([H])C1C([H])=C(C([H])([H])C([H])([H])C2C([H])=C(C([H])=C(C=2[H])OC([H])([H])[H])OC([H])([H])[H])N([H])N=1
|
| InChi Key |
VRQMAABPASPXMW-HDICACEKSA-N
|
| InChi Code |
InChI=1S/C26H33N5O3/c1-17-15-31(16-18(2)27-17)22-9-6-20(7-10-22)26(32)28-25-13-21(29-30-25)8-5-19-11-23(33-3)14-24(12-19)34-4/h6-7,9-14,17-18,27H,5,8,15-16H2,1-4H3,(H2,28,29,30,32)/t17-,18+
|
| Chemical Name |
N-[5-[2-(3,5-dimethoxyphenyl)ethyl]-1H-pyrazol-3-yl]-4-[(3S,5R)-3,5-dimethylpiperazin-1-yl]benzamide
|
| Synonyms |
fexagratinib; AZD-4547; AZD 4547; fexagratinib; N-(5-(3,5-dimethoxyphenethyl)-1H-pyrazol-3-yl)-4-((3S,5R)-3,5-dimethylpiperazin-1-yl)benzamide; AZD 4547; 2167OG1EKJ; N-[5-[2-(3,5-dimethoxyphenyl)ethyl]-1H-pyrazol-3-yl]-4-[(3R,5S)-3,5-dimethylpiperazin-1-yl]benzamide; AZD4547
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.39 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.49 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (4.49 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 4% DMSO+30% PEG 300+5% Tween 80+ddH2O: 5mg/mL Solubility in Formulation 5: 3.33 mg/mL (7.18 mM) in 1% CMC-Na/saline water (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication (<60°C). Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1572 mL | 10.7859 mL | 21.5717 mL | |
| 5 mM | 0.4314 mL | 2.1572 mL | 4.3143 mL | |
| 10 mM | 0.2157 mL | 1.0786 mL | 2.1572 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02299999 | Active Recruiting |
Drug: AZD2014 Drug: AZD4547 |
Metastatic Breast Cancer | UNICANCER | April 7, 2014 | Phase 2 |
| NCT02664935 | Active Recruiting |
Drug: AZD4547 Drug: Vistusertib |
Carcinoma, Squamous Cell Adenocarcinoma |
University of Birmingham | May 2015 | Phase 2 |
| NCT02546661 | Active Recruiting |
Drug: AZD4547 Drug: MEDI4736 |
Muscle Invasive Bladder Cancer | AstraZeneca | October 3, 2016 | Phase 1 |
| NCT05775874 | Recruiting | Drug: AZD4547 Drug: Tislelizumab |
Urothelial Carcinoma | Abbisko Therapeutics Co, Ltd | September 30, 2022 | Phase 2 |
| NCT05086666 | Recruiting | Drug: AZD4547 | Urothelial Carcinoma | Abbisko Therapeutics Co, Ltd | June 3, 2021 | Phase 1 Phase 2 |
|
 |
AZD4547 exposure correlates with in vivo antitumor activity, pharmacodynamic modulation of phospho-FGFR, and reduced tumor cell proliferation. Cancer Res. 2012 Apr 15;72(8):2045-56. |