
| Size | Price | Stock | Qty |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| Other Sizes |
|
Purity: ≥98%
Rilzabrutinib (PRN-1008) is a novel, highly potent, and reversible covalent inhibitor of BTK (Bruton's Tyrosine Kinase) being studied for treating rheumatoid arthritis. It inhibits BTK with an IC50 of 1.3 nM. BTK is a Tec family tyrosine kinase, is critical in immune pathways as an essential intracellular signaling element, participating in both adaptive and immune responses. PRN1008 was found to be very potent against BTK and highly selective when tested against a panel of 251 other kinases. Cysteine targeting of BTK by PRN1008 results in a slow off-rate demonstrated by retention of 79 ± 2% of binding to BTK in PBMC 18 hours after washing away the compound in vitro. PRN1008 was safe and well-tolerated following oral administration, and achieved high, sustained levels of BTK occupancy in peripheral blood mononuclear cells. PRN1008 is currently under Phase I development as a therapeutic agent for rheumatoid arthritis.
| ln Vitro |
With an IC50 of 1.3±0.5 nM, rilzabrutinib is a reversible covalent inhibitor of Bruton's tyrosine kinase (BTK). Additionally, when rilzabrutinib was evaluated against a panel of 251 additional kinases, it was shown to be extremely selective. Rilzabrutinib, which targets the cysteine of BTK, caused a delayed dissociation rate; 18 hours after the chemical was washed away in vitro, 79±2% of bound BTK was still present in PBMC. Complete reversibility of covalent cysteine binding occurs upon target denaturation. Rilzabrutinib suppressed human B cell proliferation (10% serum) produced by anti-IgM and B cell CD69 expression, with IC50 values of 5±2.4 nM and 123±38 nM, respectively [2].
|
||
|---|---|---|---|
| ln Vivo |
Following the drug's removal from the bloodstream, rilzabrutinib continues to have pharmacodynamic effects that are consistent with a prolonged target residence duration. Additionally, in a dose-dependent manner, rilzabrutinib reversed and totally suppressed collagen-induced arthritis in rats, which associated target occupancy with the alleviation of the disease [2].
|
||
| Animal Protocol |
|
||
| References |
[1]. Smith PF, et al. A phase I trial of PRN1008, a novel reversible covalent inhibitor of Bruton's tyrosine kinase, in healthy volunteers. Br J Clin Pharmacol. 2017 Nov;83(11):2367-2376.
[2]. Hill RJ, Bradshaw JM, Bisconte A, Tam D, Owens TD, Brameld KA, Smith PF, Funk JO, Goldstein DM, Nunn PA. Preclinical Characterization of PRN1008, a Novel Reversible Covalent Inhibitor of BTK that Shows Efficacy in a RAT Model of Collagen-Induced Arthritis. Annals of the Rheumatic Diseases 2015; 74(Suppl 2): 216. |
| Molecular Formula |
C36H40FN9O3
|
|
|---|---|---|
| Molecular Weight |
665.77
|
|
| CAS # |
1575596-29-0
|
|
| Related CAS # |
1575596-77-8;1575596-29-0;1575591-66-0
|
|
| SMILES |
FC1C([H])=C(C([H])=C([H])C=1C1C2=C(N([H])[H])N=C([H])N=C2N([C@]2([H])C([H])([H])C([H])([H])C([H])([H])N(C(/C(/C#N)=C(\[H])/C(C([H])([H])[H])(C([H])([H])[H])N3C([H])([H])C([H])([H])N(C([H])([H])C3([H])[H])C3([H])C([H])([H])OC3([H])[H])=O)C2([H])[H])N=1)OC1C([H])=C([H])C([H])=C([H])C=1[H]
|
|
| Chemical Name |
|
|
| Synonyms |
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.12 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.12 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (3.12 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5020 mL | 7.5101 mL | 15.0202 mL | |
| 5 mM | 0.3004 mL | 1.5020 mL | 3.0040 mL | |
| 10 mM | 0.1502 mL | 0.7510 mL | 1.5020 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
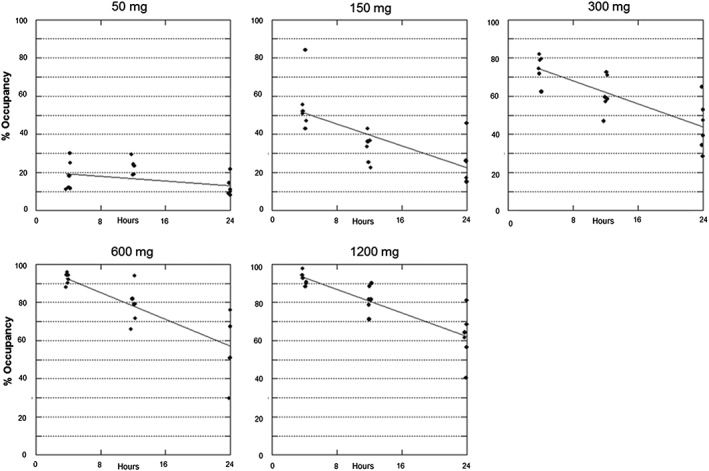 Individual BTK occupancy by PRN1008 dose level (Part A). Solid line represents fit of a linear regression model to estimate loss of occupancy over time.Br J Clin Pharmacol.2017 Nov;83(11):2367-2376. |
|---|
Duration of BTK occupancy (squares) in relation to the plasma concentration profile of PRN1008 (circles), following final dose on day 10 of a 600mg once daily dosing regimen in the multiple ascending dose study.Br J Clin Pharmacol.2017 Nov;83(11):2367-2376. |
Exposure–response relationship between 4‐hour BTK occupancy and PRN1008 maximum observed concentration (Part A).Br J Clin Pharmacol.2017 Nov;83(11):2367-2376. |