
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
Purity: ≥98%
Ganetespib (formerly STA-9090; STA 9090; STA9090), the active metabolite of STA-1474 (a prodrug of ganetespib), is a potent and triazolone-based small-molecule HSP90 (heat shock protein 90) inhibitor with potential anticancer activity. It inhibits HSP90 with an IC50 of 4 nM in OSA 8 cells, and induces apoptosis of OSA cells while normal osteoblasts are not affected. Ganetespib is being investigated for the treatment of various cancers such as Small Cell Lung Cancer, breast cancer, Acute Myeloid Leukaemia, and Myelodysplastic Syndrome.
| ln Vitro |
In genomically characterized NSCLC cell lines, genesetib induces apoptosis, suppresses downstream signaling, depletes receptor tyrosine kinases, and inhibits proliferation with IC50 values ranging from 2 to 30 nM. Additionally, in isogenic Ba/F3 pro-B cells that have been made IL-3 independent through the production of EGFR and ERBB2 mutants, genetecib is around 20 times more potent[1]. In vitro, genetecib is more effective than 17-allylamino-17-demethoxygeldanamycin (17-AAG), an ansamycin inhibitor, at causing the degradation of known Hsp90 client proteins. It also shows strong cytotoxicity in a variety of solid and hematologic tumor cell lines[2]. Strong HSP90 inhibitor genesetespib has been demonstrated to destroy canine tumor cell lines in vitro[3]. In the HEL92.1.7 cells, genesetib exhibits more potent or prolonged JAK/STAT inhibitory activity compared to P6 and 17-AAG[4].
|
||
|---|---|---|---|
| ln Vivo |
In NCI-H1975 xenografts, genetecib (125 mg/kg, IV) accumulates in tumors more than in normal tissues and has higher in vivo efficacy than 17-AAG without causing additional toxicity. It also slows proliferation and promotes apoptosis together with EGFR depletion[1]. (100, 125, 150 mg/kg, iv) significantly inhibits development and/or reverses tumor growth in solid and hematologic xenograft models of oncogene addiction, demonstrating strong anticancer efficacy[2].
|
||
| Animal Protocol |
|
||
| References |
[1]. Shimamura T, et al. Ganetespib (STA-9090), a Non-Geldanamycin HSP90 Inhibitor, has Potent Antitumor Activity in In Vitro and In Vivo Models of Non-Small Cell Lung Cancer. Clin Cancer Res. 2012 Jul 17.
[2]. Ying W, et al. Ganetespib, a unique triazolone-containing Hsp90 inhibitor, exhibits potent antitumor activity and a superior safety profile for cancer therapy. Mol Cancer Ther. 2012 Feb;11(2):475-84. [3]. London CA, et al. Phase I evaluation of STA-1474, a prodrug of the novel HSP90 inhibitor ganetespib, in dogs with spontaneous cancer. PLoS One. 2011;6(11):e27018. [4]. Proia DA, et al. Multifaceted intervention by the Hsp90 inhibitor ganetespib (STA-9090) in cancer cells with activated JAK/STAT signaling. PLoS One. 2011 Apr 14;6(4):e18552. [5]. Stewart E, et al. Identification of Therapeutic Targets in Rhabdomyosarcoma through Integrated Genomic, Epigenomic, and Proteomic Analyses. Cancer Cell. 2018 Sep 10;34(3):411-426.e19 |
| Molecular Formula |
C20H20N4O3
|
|
|---|---|---|
| Molecular Weight |
364.4
|
|
| CAS # |
888216-25-9
|
|
| Related CAS # |
|
|
| SMILES |
O=C1NN=C(C2=CC(C(C)C)=C(O)C=C2O)N1C3=CC4=C(N(C)C=C4)C=C3
|
|
| InChi Key |
RVAQIUULWULRNW-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C20H20N4O3/c1-11(2)14-9-15(18(26)10-17(14)25)19-21-22-20(27)24(19)13-4-5-16-12(8-13)6-7-23(16)3/h4-11,25-26H,1-3H3,(H,22,27)
|
|
| Chemical Name |
5-[2,4-dihydroxy-5-(1-methylethyl)phenyl]-4-(1-methyl-1H-indol-5-yl)-2,4-dihydro-3H- 1,2,4-triazol-3-one
|
|
| Synonyms |
STA-9090; Ganetespib; STA 9090; STA9090
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 10 mg/mL (27.44 mM) in 15% Cremophor EL + 85% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication.
Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.86 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (6.86 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: ≥ 2.5 mg/mL (6.86 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL corn oil and mix evenly. Solubility in Formulation 5: 1% DMSO+30% polyethylene glycol+1% Tween 80: 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7442 mL | 13.7212 mL | 27.4424 mL | |
| 5 mM | 0.5488 mL | 2.7442 mL | 5.4885 mL | |
| 10 mM | 0.2744 mL | 1.3721 mL | 2.7442 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02008877 | Completed Has Results | | Malignant Peripheral Nerve |
Sheath Tumors (MPNST) Sarcoma Alliance for Research |
through Collaboration December 2013 |
Phase 1 |
Phase 2 |
| NCT02192541 | Terminated Has Results | Drug: Ziv-Aflibercept Drug: Ganetespib |
Neoplasms | National Cancer Institute (NCI) | December 2, 2014 | Phase 1 |
| NCT01554969 | Completed | Drug: capecitabine + ganetespib | Rectal Cancer | Emory University | May 2012 | Phase 1 |
| NCT01485835 | Completed | Drug: Ganetespib Drug: Bortezomib |
Multiple Myeloma | Emory University | January 2012 | Phase 1 |
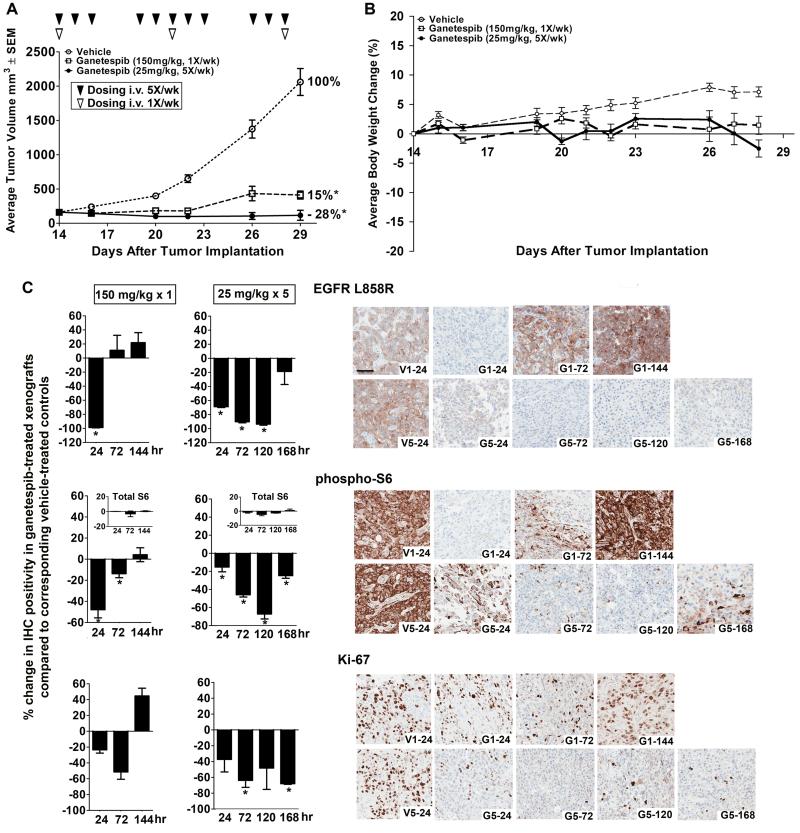 Comparison of once-weekly and five times per week dosing of ganetespib.Clin Cancer Res.2012 Sep 15;18(18):4973-85. |
|---|
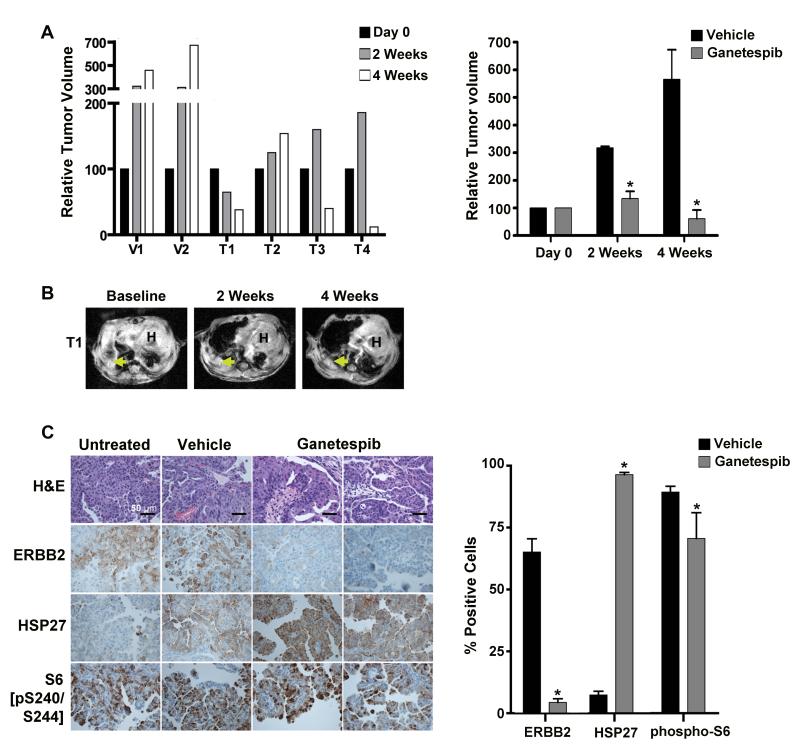 Ganetespib induces tumor regression in a mouse lung carcinoma model driven by ERRB2YVMA.Clin Cancer Res.2012 Sep 15;18(18):4973-85. |
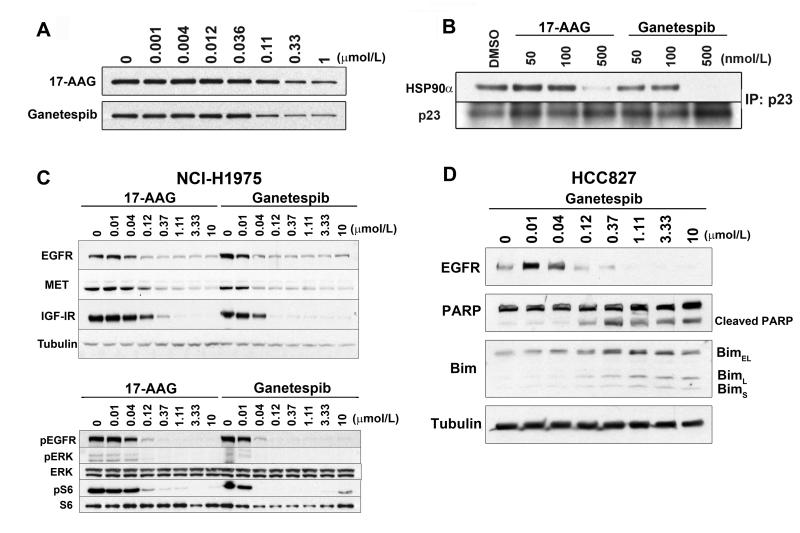 Ganetespib accumulates in tumor relative to normal tissues and displays greaterin vivoefficacy than 17-AAG.Clin Cancer Res.2012 Sep 15;18(18):4973-85. |