
| Size | Price | Stock | Qty |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg | |||
| Other Sizes |
Purity: ≥98%
Resminostat (formerly RAS-2410; 4SC-201) is a potent inhibitor of classes I and II HDACs (histone deacetylases) such as HDAC1/3/6 with potential anticancer activity. With IC50 values between 43 and 72 nmol/L, it inhibits HDAC1/3/6. Both in vivo and in vitro antitumor efficaciousness are strong points of this substance. 4SC is exploring the use of resminostat as a liver cancer treatment partner with Yakult in Japan.
| Targets |
HDAC1 ( IC50 = 42.5 nM ); HDAC3 ( IC50 = 50.1 nM ); HDAC6 ( IC50 = 71.8 nM ); HDAC8 ( IC50 = 877 nM )
Histone Deacetylases (HDACs): In multiple myeloma (MM) cell lines (U266, RPMI 8226, MM.1S), the IC50 values for Resminostat (RAS2410) -mediated proliferation inhibition (related to HDAC inhibition) were 0.5 μM (U266), 0.8 μM (RPMI 8226), and 1.2 μM (MM.1S) [1] - Histone Deacetylases (HDACs, including HDAC1, HDAC2, HDAC3, HDAC6): In head and neck squamous cell carcinoma (HNSCC) cell lines (SCC-25, CAL-27, FaDu), the IC50 for HDAC inhibition (assessed by histone H3 acetylation) was 0.3–0.7 μM; specific IC50 for HDAC1 was 0.4 μM, HDAC2 was 0.5 μM, HDAC3 was 0.6 μM, and HDAC6 was 0.8 μM [2] - Histone Deacetylases (HDACs): In human hepatocellular carcinoma (HCC) cell lines (HepG2, Huh7), the IC50 for proliferation inhibition (linked to HDAC inhibition) was 0.6 μM (HepG2) and 0.9 μM (Huh7); when combined with mTOR inhibitor (everolimus), the IC50 of Resminostat (RAS2410) decreased to 0.2 μM (HepG2) and 0.3 μM (Huh7) [3] |
|---|---|
| ln Vitro |
Resminostat (RAS2410; 4SC-201; 5 μM) causes histone acetylation. in myeloma cells. Resminostat hydrochloride has a mean Ki value of 27 nM, indicating a substrate competitive binding mode. Histone hyperacetylation is induced in myeloma cells by 5 μM of resminostat hydrochloride. Resminostat suppresses the growth of cells, triggers apoptosis, and reduces the number of MM cells. Resminostat (5 μM) also affects the expression of proteins in the bcl-2 family and prevents signaling through the Akt pathway that occurs after Akt. Combining resminostat with both established and novel anti-myeloma agents has a synergistic effect on myeloma cells[1].
Resminostat shows IC50s ranging from 0.775 μM to 1.572 μM (IC50 for SCC25: 0.775 μM, CAL27: 1.572 μM, and FaDu: 0.899 μM) in head and neck squamous cell carcinoma cell lines, indicating that it inhibits cell growth. HNSCC cell lines respond well to both irradiation and resminostat (1.25 and 2.5 μM). One can downregulate survivin by combining resminostat with cisplatin. On Mcl-1 and p-AKT expression, however, Resminostat has no effect[2]. Resminostat decreases HCC cell viability when combined with AZD-2014; IC50s range from 0.89 ± 0.12 μM to 0.07 ± 0.01 μM[3]. In MM cell lines (U266, RPMI 8226, MM.1S): Resminostat (RAS2410) inhibited cell proliferation in a dose-dependent manner (IC50: 0.5–1.2 μM). It induced apoptosis, with Annexin V-positive cells increasing from 5% (control) to 35–45% (1 μM treatment for 48 h). Western blot analysis showed increased acetylation of histone H3 and H4, upregulated expression of pro-apoptotic proteins (Bax, cleaved caspase-3/9), and downregulated anti-apoptotic protein (Bcl-2). PCR results revealed increased mRNA levels of p21WAF1/CIP1 (a cell cycle inhibitor) [1] - In HNSCC cell lines (SCC-25, CAL-27, FaDu): Resminostat (RAS2410) reduced cell viability in a time- and dose-dependent way (IC50: 0.3–0.7 μM at 72 h). It inhibited clone formation: the number of clones decreased by 60–70% at 1 μM compared to control. It also suppressed cell migration and invasion (transwell assay: migrated cells reduced by 50–60% at 0.5 μM). Western blot showed increased acetyl-histone H3, downregulated MMP-9 (matrix metalloproteinase) and cyclin D1, and upregulated p27KIP1 [2] - In HCC cell lines (HepG2, Huh7): Resminostat (RAS2410) alone inhibited proliferation (IC50: 0.6–0.9 μM) and induced mild apoptosis (15–20% Annexin V-positive cells at 1 μM). When combined with everolimus (mTOR inhibitor, 10 nM), it exerted synergistic effects: proliferation inhibition rate increased from 40% (resminostat alone) to 75–80%, and apoptosis rate increased to 45–50%. Western blot demonstrated enhanced acetylation of histone H3, reduced phosphorylation of mTOR and its downstream targets (p70S6K, 4E-BP1), and increased cleaved caspase-3 [3] |
| ln Vivo |
Resminostat taken orally at 600 mg QD every day for 1–5 days in a 14-day cycle is well tolerated. With low inter-pt variability and high bioavailability, resminostat exhibits a favorable pharmacokinetic profile. Oral resminostat's apparent t1/2 varied from 2.7 to 4.4 hours. Additional evidence of medication activity is the modulation of plasma biomarkers.
In SCID mice bearing MM xenografts (U266 cells injected subcutaneously): Mice were divided into control (saline) and Resminostat (RAS2410) groups (50 mg/kg, intraperitoneal injection, once daily for 21 days). The treatment group showed a 65–70% reduction in tumor volume (from 1000 mm³ to 300–350 mm³) and a 55–60% decrease in tumor weight (from 1.2 g to 0.5–0.55 g) compared to control. Survival time was prolonged by 25–30% (median survival: 35 days in control vs. 45–46 days in treatment group). Immunohistochemistry of tumor tissues showed increased acetyl-histone H3 and cleaved caspase-3, and decreased Ki-67 (proliferation marker) [1] |
| Enzyme Assay |
A 96-well microtitre plate is filled with 40 microliters of enzyme buffer (15 mM Tris HCl pH 8.1, 0.25 mM EDTA, 250 mM NaCl, 10% v:v glycerol) containing HDAC1, 3, 6, or 8 activity, 29 microliters of enzyme buffer, and 1 microliter of resminostat [HCl] at varying concentrations. The reaction is initiated by adding 30 microliters of substrate peptide Ac-NH-GGK(Ac)-AMC (HDAC1, 3 and 6 assays, final concentrations of 6 μM for HDAC1, 10 μM for HDAC6, and 25 μM for HDAC3/DAD) or Ac-RHK(Ac)K(Ac)-AMC (HDAC8 assay, final concentration 50 μM). The reaction is stopped by adding 25 μL of stop solution (50 mM Tris HCl pH 8, 100 mM NaCl, 0.5 mg/mL trypsin, and 2 μM trichostatin A [TSA]) after the enzymes have been incubated for 180 min (HDAC1, HDAC6, HDAC8) or 120 min (HDAC3) at 30°C. The quantification of AMC produced by tryptic cleavage of the deacetylated peptide is done using a Wallac Victor2 1420 multilabel counter (extinction 355 nm, emission 460 nm) following an additional 40 minutes of room temperature incubation. The fluorescence in wells without test compound (1% DMSO, negative control) is set to 100% enzymatic activity for the calculation of the 50% inhibitory concentration (IC50) values, while the fluorescence in wells with 2 μM TSA (positive control) is set to 0% enzymatic activity (background fluorescence substracted)[1].
HDAC Activity Assay (for MM research): Prepare a reaction mixture containing recombinant HDACs (HDAC1/2/3/6), fluorogenic substrate (trifluoroacetyl-peptide-7-amino-4-methylcoumarin), and different concentrations of Resminostat (RAS2410) (0.1–10 μM). Incubate the mixture at 37°C for 60 minutes. Add a developer solution to stop the reaction and release the fluorescent product. Measure fluorescence intensity at excitation 360 nm and emission 460 nm. Calculate the percentage of HDAC inhibition by comparing fluorescence intensity of drug-treated groups with the control (no drug). Determine IC50 values for each HDAC subtype [1] - HDAC Activity Assay (for HNSCC research): Mix purified HDAC enzymes (HDAC1–6), fluorescent substrate (peptide conjugated to 7-amino-4-trifluoromethylcoumarin), and Resminostat (RAS2410) (0.05–5 μM) in a 96-well plate. Incubate at 37°C for 45 minutes. Add a stop solution to quench the enzyme reaction. Detect fluorescence at 355 nm (excitation) and 460 nm (emission) using a microplate reader. Calculate HDAC activity as the ratio of fluorescence in drug-treated wells to control wells. Plot dose-response curves to obtain IC50 for each HDAC subtype [2] |
| Cell Assay |
The antiproliferative effect of resminostat on HNSCC cells is examined using a CCK-8 cell proliferation assay. Three × 105 cells per well are seeded into 96-well plates. Following a 24-hour growth period, the cells undergo treatment with resminostat and cisplatin, either separately or in conjunction, and are then incubated for a full 72 hours. Equal amounts of dimethylsulfoxide were used as a control group along with untreated cells that were kept in RPMI. CCK-8 is used to measure the proliferation of cells after 72 hours. Three times, each experiment is conducted in triplicate[2].
MM Cell Proliferation Assay: Seed MM cells (U266, RPMI 8226, MM.1S) in 96-well plates at 5×10³ cells/well. Treat with Resminostat (RAS2410) at concentrations of 0.1, 0.5, 1, 2, 5 μM (control: vehicle). Incubate for 24, 48, 72 hours. Add a cell viability reagent and incubate for 4 hours. Measure absorbance at 490 nm. Calculate proliferation inhibition rate and IC50 values [1] - MM Apoptosis Assay: Treat MM cells (U266) with 1 μM Resminostat (RAS2410) for 48 hours. Collect cells, wash with buffer, and stain with Annexin V-FITC and propidium iodide (PI) for 15 minutes in the dark. Analyze cells using flow cytometry. Count the percentage of Annexin V-positive/PI-negative (early apoptosis) and Annexin V-positive/PI-positive (late apoptosis) cells [1] - HNSCC Clone Formation Assay: Seed HNSCC cells (SCC-25) in 6-well plates at 200 cells/well. After 24 hours, treat with Resminostat (RAS2410) (0.1, 0.5, 1 μM; control: vehicle). Incubate for 14 days, replacing medium with fresh drug every 3 days. Fix cells with formaldehyde and stain with crystal violet. Count visible clones (≥50 cells/clone) and calculate the clone formation rate relative to control [2] - HCC Combined Treatment Assay: Seed HepG2 cells in 96-well plates at 4×10³ cells/well. After attachment, treat with Resminostat (RAS2410) (0.2, 0.5, 1 μM) alone or in combination with everolimus (10 nM). Incubate for 72 hours. Use a cell viability kit to measure absorbance at 450 nm. Calculate combination index (CI) using CalcuSyn software to assess synergism (CI < 0.9 indicates synergism) [3] |
| Animal Protocol |
MM Xenograft Mouse Model Protocol: Female SCID mice (6–8 weeks old) were injected subcutaneously with 5×10⁶ U266 MM cells into the right flank. When tumors reached 100 mm³, mice were randomly divided into 2 groups (n=6/group): control group (intraperitoneal injection of 0.9% saline, once daily) and Resminostat (RAS2410) group (intraperitoneal injection of 50 mg/kg Resminostat (RAS2410) dissolved in 0.9% saline, once daily). Treatments continued for 21 days. Measure tumor volume (using calipers, formula: volume = length × width² / 2) and mouse body weight every 3 days. At the end of treatment, sacrifice mice, excise tumors, and weigh them. Collect tumor tissues for immunohistochemistry and Western blot analysis. Monitor mouse survival for 60 days to calculate median survival time [1]
|
| Toxicity/Toxicokinetics |
In the MM xenograft mouse model: no significant weight loss (weight change <10% vs. control group) or significant toxic symptoms (e.g., somnolence, diarrhea, hair loss) were observed during a 21-day treatment period with 50 mg/kg Resminostat (RAS2410). Serum biochemical analyses (ALT, AST, BUN, creatinine) showed no significant differences between the control and treatment groups, indicating no significant hepatotoxicity or nephrotoxicity [1]. In the HNSCC cell line: Resminostat (RAS2410) at concentrations up to 2 μM did not induce significant cytotoxicity in normal oral keratinocytes (NOK), with cell viability remaining above 80% (compared to 40-50% viability in HNSCC cells at the same concentration), indicating selective cytotoxicity to cancer cells [2].
|
| References |
|
| Additional Infomation |
Resminostat is an enamide formed by the condensation of the carboxyl group of (2E)-3-[1-({4-[(dimethylamino)methyl]phenyl}sulfonyl)-1H-pyrrolo-3-yl]prop-2-enoic acid with the amino group of hydroxylamine. It is an orally bioavailable histone deacetylase inhibitor with potential antitumor activity. It functions as an EC 3.5.1.98 (histone deacetylase) inhibitor, antitumor drug, and apoptosis inducer. It belongs to the benzene, sulfonamide, pyrrole, tertiary amine, hydroxamic acid, and enamide classes. Resminostat has been used in clinical trials for various diseases, including Cezari syndrome, mycosis fungoides, Hodgkin's lymphoma, hepatocellular carcinoma, and T-cell cutaneous lymphoma. Resminostat is an orally bioavailable histone deacetylase (HDAC) inhibitor with potential antitumor activity. Resminostat binds to and inhibits the activity of HDACs, leading to the accumulation of highly acetylated histones. This may result in chromatin remodeling, transcriptional repression of tumor suppressor genes, inhibition of tumor cell division, and tumor cell apoptosis. HDACs are upregulated in many tumor types and are a class of enzymes that can deacetylate chromatin histones.
Drug indications Treatment of cutaneous T-cell lymphoma Resminostat (RAS2410) is a novel oral active histone deacetylase (HDAC) inhibitor that is more selective for class I HDACs (HDAC1–3) and class IIb HDACs (HDAC6) compared to non-selective HDAC inhibitors. In multiple myeloma (MM), its mechanism of action includes inhibiting histone deacetylase (HDAC) activity, increasing histone acetylation, regulating the expression of apoptosis-related genes and cell cycle-related genes, thereby inhibiting MM cell proliferation and inducing apoptosis[1]. In head and neck squamous cell carcinoma (HNSCC), Resminostat (RAS2410) exerts its antitumor effect by inhibiting HDAC-enhanced histone acetylation, downregulating the expression of cell proliferation-related genes (cyclin D1) and invasion-related genes (MMP-9), and upregulating the expression of cell cycle repressor (p27KIP1), thereby inhibiting the growth and metastasis of HNSCC cells [2]. In hepatocellular carcinoma (HCC), the synergistic effect of Resminostat (RAS2410) and mTOR inhibitors is attributed to the interaction between the HDAC and mTOR pathways: the inhibitory effect of Resminostat (RAS2410) on HDAC enhances the sensitivity of HCC cells to mTOR inhibitors, thereby improving the efficacy of mTOR inhibitors. It inhibits cell proliferation and induces apoptosis. This provides a potential combination therapy strategy for hepatocellular carcinoma [3]. |
| Molecular Formula |
C16H19N3O4S
|
|---|---|
| Molecular Weight |
349.4
|
| Exact Mass |
349.109
|
| Elemental Analysis |
C, 55.00; H, 5.48; N, 12.03; O, 18.32; S, 9.18
|
| CAS # |
864814-88-0
|
| Related CAS # |
1187075-34-8 (HCl); 864814-88-0(free base)
|
| PubChem CID |
11609955
|
| Appearance |
Solid powder
|
| Density |
1.3±0.1 g/cm3
|
| Index of Refraction |
1.600
|
| LogP |
1.69
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
5
|
| Rotatable Bond Count |
6
|
| Heavy Atom Count |
24
|
| Complexity |
548
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
S(C1C=CC(CN(C)C)=CC=1)(N1C=CC(/C=C/C(=O)NO)=C1)(=O)=O
|
| InChi Key |
FECGNJPYVFEKOD-VMPITWQZSA-N
|
| InChi Code |
InChI=1S/C16H19N3O4S/c1-18(2)11-13-3-6-15(7-4-13)24(22,23)19-10-9-14(12-19)5-8-16(20)17-21/h3-10,12,21H,11H2,1-2H3,(H,17,20)/b8-5+
|
| Chemical Name |
(E)-3-[1-[4-[(dimethylamino)methyl]phenyl]sulfonylpyrrol-3-yl]-N-hydroxyprop-2-enamide
|
| Synonyms |
RAS2410; RAS-2410; RAS2410; RAS 2410; 4SC201; 4SC-201; 4SC 201; Resminostat
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8620 mL | 14.3102 mL | 28.6205 mL | |
| 5 mM | 0.5724 mL | 2.8620 mL | 5.7241 mL | |
| 10 mM | 0.2862 mL | 1.4310 mL | 2.8620 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02953301 | Active Recruiting |
Drug: resminostat Drug: Placebo |
Mycosis Fungoides Sezary Syndrome |
4SC AG | November 2016 | Phase 2 |
| NCT04525729 | Recruiting | Drug: RAS 2410 Drug: Rituximab |
IgA Nephropathy | CHENNAN | July 1, 2020 | Phase 4 |
| NCT04955340 | Completed | Drug: [14C]-resminostat | Mycosis Fungoides Sezary Syndrome |
4SC AG | October 12, 2021 | Phase 1 |
| NCT01037478 | Completed | Drug: Resminostat (4SC-201) |
Hodgkin's Lymphoma | 4SC AG | December 2009 | Phase 2 |
| NCT02400788 | Completed | Drug: Resminostat Drug: Sorafenib |
Hepatocellular Carcinoma | Yakult Honsha Co., LTD | April 2013 | Phase 1 Phase 2 |
Resminostat synergizes with common and new anti‐myeloma agents.Br J Haematol. 2010 May;149(4):518-28. |
|---|
Resminostat inhibits cell growth and induces apoptosis in MM cells.Br J Haematol. 2010 May;149(4):518-28. |
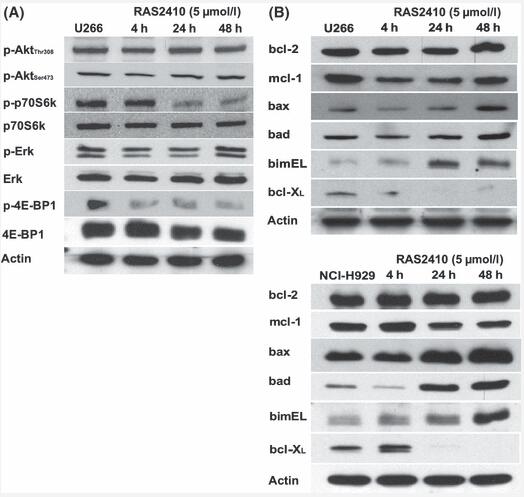 Resminostat modulates expression of bcl-2 family proteins and inhibits Akt pathway signalling downstream of Akt.Br J Haematol.2010 May;149(4):518-28. |
Resminostat induces histone acetylation in myeloma cells.Br J Haematol. 2010 May;149(4):518-28. |
|---|
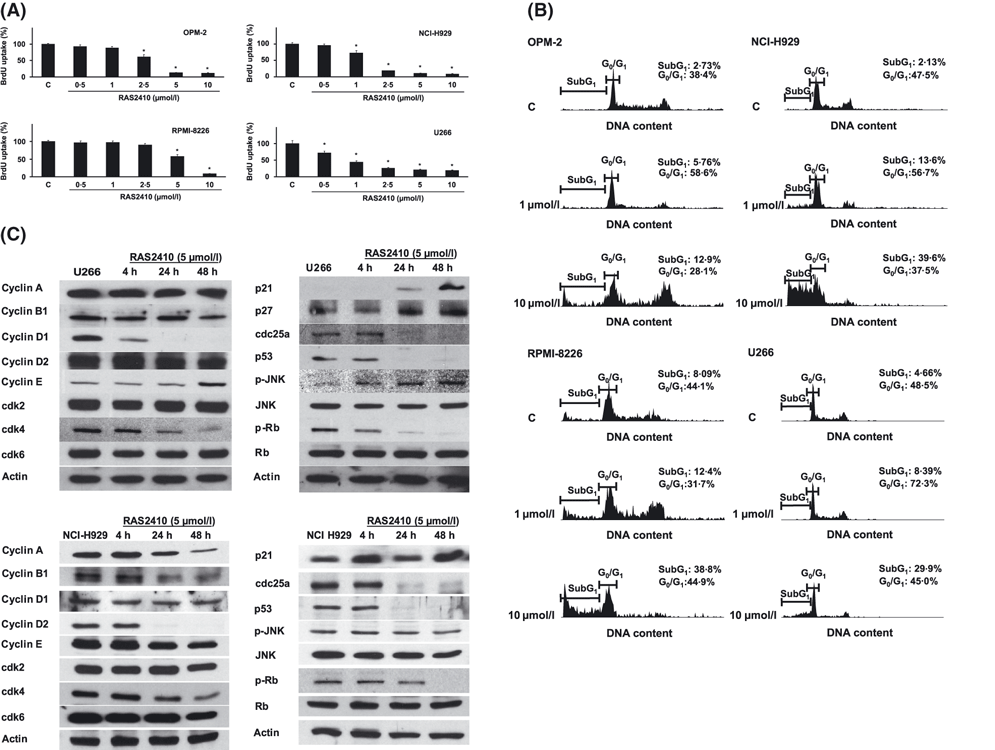 Resminostat inhibits MM cell proliferation. Br J Haematol. 2010 May;149(4):518-28.Br J Haematol. 2010 May;149(4):518-28. |